Revolutionizing Pharmaceutical Intermediate Production: Visible-Light Catalyzed Para-Trifluoromethylation of Aniline Derivatives
The Chinese patent CN113214113B introduces a groundbreaking visible-light photocatalyzed method for synthesizing aniline para-trifluoromethylated derivatives, representing a significant advancement in fluorinated compound manufacturing for pharmaceutical applications. This innovative approach addresses longstanding challenges in selective trifluoromethylation of aniline derivatives, which are critical building blocks for numerous pharmaceutical agents including leflunomide and teriflunomide. The patented process utilizes a simple photocatalytic system that operates under ambient conditions while delivering exceptional para-selectivity—a capability previously unattainable with conventional methods. By eliminating transition metal catalysts and high-temperature requirements, this methodology offers pharmaceutical manufacturers a more sustainable pathway to produce fluorinated intermediates with improved purity profiles and reduced environmental impact. The technology's commercial viability is further enhanced by its compatibility with diverse aniline substrates and straightforward scale-up potential from laboratory to industrial production volumes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to aniline trifluoromethylation have been severely constrained by multiple technical limitations that hinder their commercial viability for pharmaceutical manufacturing. Conventional methods relying on transition metal catalysis typically require elevated temperatures (90°C) and stoichiometric metal salts, resulting in low yields and extensive purification requirements to remove toxic metal residues—a critical concern for pharmaceutical applications where stringent purity standards must be met. The dehalogenative coupling strategies previously employed suffer from narrow substrate scope and necessitate pre-functionalization steps that add complexity and cost to the synthetic route. Radical-based approaches have struggled with regioselectivity issues, particularly when targeting para-substitution on substituted anilines—a challenge that has remained unresolved until now. These limitations collectively create significant barriers to producing high-purity fluorinated intermediates at commercial scale while meeting pharmaceutical industry requirements for cost-effectiveness and environmental sustainability.
The Novel Approach
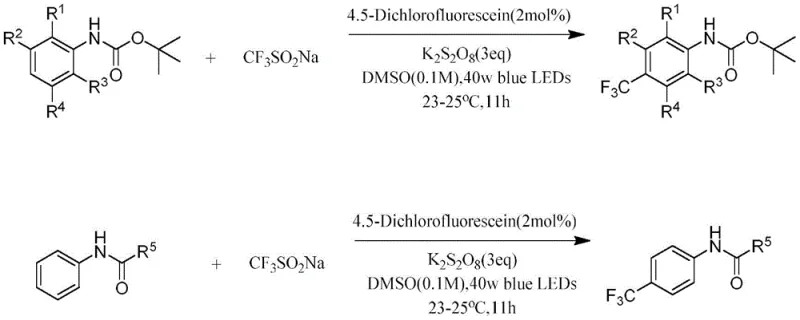
The patented methodology overcomes these longstanding challenges through an innovative visible-light photocatalytic system that operates under remarkably mild conditions—room temperature (23-25°C) with 40W blue LED illumination—while achieving unprecedented para-selectivity for trifluoromethylation of diverse aniline derivatives. By utilizing 4,5-dichlorofluorescein as a photocatalyst (2 mol%), potassium persulfate (3 equiv), and sodium trifluoromethanesulfinate in DMSO solvent (0.1M), this process eliminates transition metals entirely while maintaining excellent functional group tolerance across a wide range of substrates. The reaction proceeds efficiently over 11 hours with simple workup procedures involving aqueous quenching and ethyl acetate extraction, followed by standard column chromatography purification using petroleum ether/ethyl acetate (10:1). This approach delivers consistently high yields across numerous substrate variations while producing minimal byproducts—a critical advantage for pharmaceutical manufacturing where impurity profiles directly impact product quality and regulatory approval pathways.
Mechanistic Insights into Visible-Light Photocatalyzed Trifluoromethylation
The core innovation lies in the photoinduced electron transfer mechanism that enables selective para-trifluoromethylation without requiring transition metals or harsh conditions. Under blue LED irradiation, 4,5-dichlorofluorescein absorbs visible light to form an excited state that facilitates single-electron transfer to potassium persulfate, generating sulfate radical anions that subsequently oxidize sodium trifluoromethanesulfinate to produce trifluoromethyl radicals. These radicals then selectively attack the para-position of aniline derivatives through a well-defined electronic interaction pathway that leverages the electron-donating properties of the amino group while avoiding ortho-substitution through steric considerations. The mild reaction conditions prevent over-oxidation or decomposition pathways commonly observed in traditional methods, resulting in cleaner reaction profiles with fewer side products. This mechanistic understanding explains why the process maintains high selectivity across diverse substituted anilines where previous methods failed completely.
The process demonstrates exceptional impurity control through its selective radical mechanism that minimizes competing side reactions typically observed in conventional trifluoromethylation approaches. By operating at room temperature without strong acids or bases, the method preserves sensitive functional groups that would otherwise decompose under traditional conditions. The absence of transition metals eliminates potential metal contamination concerns that require extensive purification steps in pharmaceutical manufacturing. The well-defined reaction pathway produces minimal byproducts—primarily unreacted starting materials that are easily separated during standard workup procedures—resulting in crude products with high purity that require minimal additional processing before meeting pharmaceutical quality standards. This inherent selectivity translates directly to improved process efficiency and reduced manufacturing costs while ensuring consistent product quality across production batches.
How to Synthesize Aniline Para-Trifluoromethylated Derivatives Efficiently
This patented methodology represents a significant advancement in the synthesis of fluorinated aniline derivatives for pharmaceutical applications, offering researchers and manufacturers a reliable pathway to produce high-value intermediates with exceptional selectivity and purity. The following standardized procedure has been developed based on extensive optimization studies documented in CN113214113B, providing a robust framework for consistent production of these critical building blocks. Detailed operational parameters have been refined to ensure maximum yield and purity while maintaining practicality for both laboratory-scale development and commercial manufacturing environments.
- Combine aniline derivative (1 equiv), 4,5-dichlorofluorescein photocatalyst (2 mol%), potassium persulfate (3 equiv), and sodium trifluoromethanesulfinate (2 equiv) in DMSO solvent (0.1M concentration).
- Irradiate the reaction mixture with 40W blue LED light at room temperature (23-25°C) while maintaining constant stirring at 500 rpm for 11 hours.
- Quench the reaction with water, extract with ethyl acetate three times, dry the combined organic layers with anhydrous sodium sulfate, and concentrate under reduced pressure before column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach directly addresses critical pain points faced by procurement and supply chain professionals in the pharmaceutical industry by delivering a more sustainable and reliable pathway to essential fluorinated intermediates. The elimination of transition metals from the synthetic route removes significant supply chain vulnerabilities associated with precious metal sourcing while reducing quality control complexities related to metal residue testing—a major regulatory concern in pharmaceutical manufacturing. The use of commercially available reagents with stable supply chains enhances production reliability while minimizing exposure to market fluctuations that commonly affect specialized chemical inputs.
- Cost Reduction in Manufacturing: The metal-free nature of this process eliminates expensive catalysts and associated purification steps required to remove metal residues from final products, resulting in substantial cost savings throughout the manufacturing workflow. The ambient temperature operation significantly reduces energy consumption compared to traditional high-temperature methods while utilizing readily available reagents that maintain stable pricing due to their widespread industrial applications across multiple sectors.
- Enhanced Supply Chain Reliability: By relying on common chemical reagents with established global supply networks rather than specialized or rare materials, this methodology provides greater supply chain resilience against market disruptions. The simplified reaction setup requires only basic laboratory equipment widely available across manufacturing facilities worldwide, eliminating dependencies on specialized instrumentation that could create production bottlenecks during equipment shortages or maintenance periods.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to commercial production volumes without requiring significant process re-engineering—a critical advantage for rapid technology transfer from development to manufacturing stages. The mild reaction conditions and aqueous workup procedures generate minimal hazardous waste streams compared to conventional methods involving strong acids or high temperatures, aligning with increasingly stringent environmental regulations while reducing waste treatment costs associated with pharmaceutical manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common concerns from technical procurement teams regarding implementation of this innovative manufacturing process for pharmaceutical intermediates. These answers are derived directly from experimental data and operational insights documented in Chinese patent CN113214113B.
Q: How does this photocatalytic method achieve high para-selectivity for trifluoromethylation of substituted anilines?
A: The visible-light photocatalysis system using 4,5-dichlorofluorescein creates a radical pathway that specifically targets the para-position of aniline derivatives through a well-defined electronic interaction mechanism, overcoming traditional limitations of regioselectivity in trifluoromethylation reactions.
Q: What are the key advantages of this method compared to conventional transition metal-catalyzed approaches?
A: This metal-free photocatalytic process eliminates expensive transition metals and associated purification steps, operates under mild room temperature conditions without specialized equipment, and demonstrates broader substrate compatibility with significantly simplified post-reaction processing.
Q: How does this method support scalable production of pharmaceutical intermediates?
A: The reaction's ambient temperature operation, use of commercially available reagents, straightforward workup procedure, and demonstrated scalability from laboratory to commercial production volumes make it ideal for manufacturing pharmaceutical intermediates with consistent quality and reduced operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aniline Para-Trifluoromethylated Derivatives Supplier
Our company stands at the forefront of advanced fluorinated intermediate manufacturing, leveraging this patented visible-light photocatalyzed technology to deliver high-purity aniline para-trifluoromethylated derivatives with exceptional consistency and reliability. As a CDMO expert specializing in complex fluorination chemistry, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical capabilities for comprehensive quality assurance.
We invite your technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements, which will demonstrate how our innovative approach can optimize your supply chain while meeting all regulatory requirements. Please contact us to obtain specific COA data and route feasibility assessments for your target compounds—our technical specialists are ready to provide detailed information on how this breakthrough technology can enhance your pharmaceutical development pipeline.
