Transforming PGF2α Waste into High-Purity Alprostadil: A Commercial Scale-Up Breakthrough
Transforming PGF2α Waste into High-Purity Alprostadil: A Commercial Scale-Up Breakthrough
The pharmaceutical industry constantly seeks sustainable pathways to reduce the environmental footprint of Active Pharmaceutical Ingredient (API) manufacturing while simultaneously driving down costs. A groundbreaking approach detailed in patent CN115636776A addresses both challenges by introducing a novel method for synthesizing Alprostadil (PGE1) using byproducts from PGF2α synthesis as the primary raw material. Traditionally, the production of dinoprost (PGF2α) generates a significant quantity of 5,6-trans isomer impurities, often accounting for nearly 40% of the reaction mass, which are typically discarded as waste due to their lack of physiological activity in the target indication. This innovative technology repurposes these high-volume waste streams, transforming a liability into a valuable asset through a streamlined, four-step chemical sequence that avoids the reconstruction of chiral centers. By leveraging the inherent stereochemistry of the waste material, the process ensures high optical purity while drastically simplifying the supply chain logistics for this critical cardiovascular therapeutic agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Alprostadil has relied on complex, multi-step routes such as the Corey lactone methodology, which presents substantial hurdles for large-scale manufacturing and cost efficiency. These traditional pathways often necessitate the use of highly sensitive and hazardous reagents, including organolithium compounds like tert-butyllithium and explosive alkylating agents such as methyl iodide, which require specialized equipment and rigorous safety protocols to handle safely. Furthermore, the construction of the lower side chain in these legacy methods frequently involves Wittig-Horner reactions that demand precise temperature control and generate stoichiometric amounts of phosphine oxide waste, complicating downstream purification and increasing the overall E-factor of the process. The reliance on reconstructing chiral centers from simpler achiral starting materials also introduces significant risks of racemization, necessitating expensive chiral chromatography or resolution steps that erode profit margins and extend production lead times significantly.
The Novel Approach
In stark contrast, the method disclosed in CN115636776A circumvents these historical bottlenecks by initiating synthesis from the readily available 5,6-trans isomer byproduct of PGF2α production, effectively turning a waste management problem into a raw material sourcing solution. This strategy eliminates the need for constructing the cyclopentane ring or establishing the initial chiral configuration, as these structural features are already present and correctly configured in the starting impurity. The process employs a clever sequence of selective protection, oxidation, and hydrogenation that operates under mild conditions, avoiding the extreme temperatures and cryogenic requirements associated with organometallic reagents in prior art. By utilizing the trans-isomer impurity, which is structurally very similar to the desired cis-product but historically useless for PGF2α, the method achieves a circular economy model within the prostaglandin manufacturing sector, significantly reducing the cost of goods sold (COGS) and minimizing the environmental impact of chemical waste disposal.
Mechanistic Insights into Selective Protection and Hydrogenation
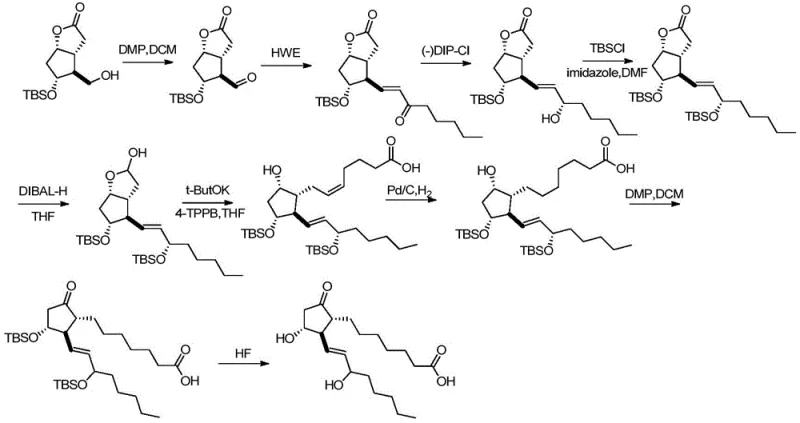
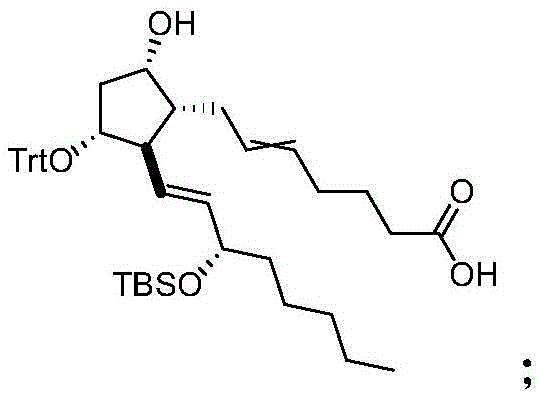
The core chemical innovation of this process lies in the exquisite control of regioselectivity during the protection and deprotection phases, driven by the subtle differences in steric hindrance between the hydroxyl groups on the cyclopentane ring and the side chains. In the initial step (S100), the protocol exploits the higher reactivity of the primary hydroxyl group at the C15 position to install a silicon-based protecting group, such as a tert-butyldiphenylsilyl (TBDPS) ether, while leaving the secondary hydroxyls available for subsequent differentiation. Following this, the secondary hydroxyl at the C11 position is selectively protected with a bulky trityl (triphenylmethyl) group; this selectivity is achieved because the C11 hydroxyl is less sterically hindered compared to the C9 hydroxyl, which is flanked by the long upper side chain at C8 in a cis-configuration, creating a crowded environment that impedes the approach of the bulky trityl chloride reagent.
Following the establishment of the orthogonal protection pattern, the synthesis proceeds through a strategic oxidation and hydrogenation sequence that capitalizes on the stability of the installed protecting groups. The oxidation of the C9 hydroxyl to a ketone (S200) is performed using mild oxidants like Dess-Martin periodinane, which preserves the acid-sensitive trityl group and the double bonds in the side chains. The subsequent hydrogenation step (S300) is particularly elegant, as it achieves two transformations simultaneously: the saturation of the C5-C6 double bond and the removal of the C11 trityl protecting group. This dual functionality is enabled by the use of a palladium on carbon catalyst under controlled hydrogen pressure, where the steric bulk of the C15 silyl ether prevents the hydrogenation of the C13-C14 double bond, ensuring that only the desired 5,6-unsaturation is reduced. This chemoselectivity is critical for maintaining the biological activity of the final Alprostadil molecule, as the C13-C14 unsaturation is essential for its pharmacological profile.
How to Synthesize Alprostadil Efficiently
The implementation of this synthesis route requires careful attention to reaction conditions, particularly regarding temperature control and reagent stoichiometry, to maximize the yield of the intermediate compounds and the final API. The process begins with the dissolution of the PGF2α byproduct mixture in a polar aprotic solvent like DMF, followed by the sequential addition of imidazole and the silylating agent at low temperatures to ensure exclusive mono-protection at the C15 position. Once the first protection is complete, the trityl chloride is introduced to the same pot, leveraging the crude reaction mixture to minimize solvent swaps and handling losses, a technique that significantly enhances the overall throughput of the manufacturing line. The subsequent oxidation and hydrogenation steps are designed to be robust and scalable, utilizing common industrial catalysts and reagents that do not require exotic handling facilities, making this route highly attractive for contract development and manufacturing organizations (CDMOs) looking to optimize their prostaglandin portfolios.
- Perform selective silicon-based protection on the 15-hydroxyl group and trityl protection on the 11-hydroxyl group of the PGF2α byproduct starting material.
- Oxidize the 9-position hydroxyl group of the protected intermediate to a carbonyl group using Dess-Martin periodinane or PCC.
- Execute a simultaneous hydrogenation of the 5,6-double bond and removal of the 11-position trityl protecting group using a palladium catalyst.
- Remove the remaining silicon-based protecting group at the 15-position using tetrabutylammonium fluoride to yield the final Alprostadil product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this waste-recycling synthesis route offers profound strategic advantages that extend far beyond simple unit cost reductions. By sourcing raw materials from the byproduct stream of PGF2α production, manufacturers can decouple their Alprostadil supply from the volatility of global markets for specialty chiral building blocks, which are often subject to long lead times and geopolitical supply constraints. The ability to utilize a material that was previously classified as hazardous waste not only eliminates the disposal costs associated with that waste but also creates a negative-cost raw material input, fundamentally altering the cost structure of the final API. Furthermore, the simplified reaction sequence reduces the number of unit operations required, which directly translates to lower capital expenditure (CAPEX) for reactor time and reduced consumption of solvents and energy, contributing to a more sustainable and economically resilient manufacturing operation.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the valorization of waste, meaning the primary feedstock is essentially a credit rather than a debit on the balance sheet. By eliminating the need for expensive chiral pool starting materials like Corey lactone and avoiding the use of precious metal catalysts that require complex recovery systems, the variable costs of production are drastically lowered. The process avoids the use of cryogenic reagents and pyrophoric substances, which reduces the need for specialized containment infrastructure and lowers insurance and safety compliance costs. Additionally, the high atom economy of the transformation, where most of the carbon skeleton of the starting material is retained in the final product, minimizes raw material waste and maximizes the output per kilogram of input, leading to substantial overall cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: Relying on internal byproduct streams or established PGF2α production lines creates a closed-loop supply chain that is inherently more stable than relying on external vendors for complex intermediates. Since the starting material is a direct congener of the target molecule, the supply risk associated with multi-step upstream synthesis is mitigated, ensuring a consistent flow of feedstock for Alprostadil production. The robustness of the chemical steps, which tolerate minor variations in starting material quality without compromising the final purity, further enhances supply continuity by reducing the rate of batch failures and reprocessing requirements. This reliability is crucial for meeting the stringent delivery schedules of global pharmaceutical partners who require just-in-time inventory management for their finished dosage forms.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard reaction vessels and workup procedures that can be easily transferred from pilot plant to commercial scale without significant re-engineering. The absence of toxic heavy metals and explosive reagents simplifies the regulatory filing process and reduces the burden of environmental monitoring and waste treatment, aligning perfectly with modern green chemistry principles. The reduction in solvent usage and the elimination of hazardous waste streams contribute to a lower environmental footprint, which is increasingly becoming a key criterion for supplier selection by major multinational pharmaceutical companies committed to sustainability goals. This alignment with environmental, social, and governance (ESG) criteria adds intangible value to the supply partnership, positioning the manufacturer as a forward-thinking and responsible partner in the global healthcare ecosystem.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Alprostadil synthesis technology, providing clarity on its operational feasibility and regulatory standing. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a realistic view of the process capabilities and limitations for potential adopters. Understanding these nuances is essential for R&D teams evaluating the technology for technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: How does this method address the waste issue in PGF2α production?
A: Traditional PGF2α synthesis generates approximately 40% unusable 5,6-trans isomer byproduct. This patented method utilizes that specific waste stream as the primary raw material, converting a disposal cost into a value-added product without requiring new chiral centers.
Q: What are the safety advantages over conventional Alprostadil synthesis routes?
A: Unlike prior art methods that rely on air-sensitive and explosive reagents like tert-butyllithium or methyl iodide, this process operates under mild conditions using stable reagents like imidazole and palladium on carbon, significantly enhancing production safety and equipment compatibility.
Q: Is the optical purity of the recycled Alprostadil comparable to standard synthesis?
A: Yes, the method leverages the existing chirality of the PGF2α byproduct. Since no new chiral centers need to be constructed or reconstructed, the optical purity remains high, meeting stringent pharmacopoeia standards without complex chiral resolution steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alprostadil Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of waste-recycling technologies in the fine chemical sector and have invested heavily in mastering complex prostaglandin synthesis pathways. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Alprostadil meets the highest international pharmacopoeia standards. Our commitment to quality is matched by our dedication to process safety and environmental stewardship, making us an ideal partner for pharmaceutical companies seeking a sustainable and reliable source of this critical cardiovascular API.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements and regional logistics. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance metrics. Let us collaborate to build a more resilient and cost-effective supply chain for Alprostadil, leveraging cutting-edge chemistry to deliver value to patients and stakeholders alike.
