Advanced Synthetic Route for Indeno-Indole Ketones: Scalable Manufacturing and Cost Efficiency
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways for complex nitrogen-containing heterocycles, particularly those with significant biological potential. Patent CN108503578B introduces a groundbreaking synthetic method for indeno-[1,2-b]indole-10(5H)-one compounds, a scaffold known for its pharmacological activities including anticancer and anti-Alzheimer's properties. This innovation addresses critical bottlenecks in traditional manufacturing by replacing expensive transition metal catalysts with a more accessible zinc-mediated coupling strategy. By leveraging this technology, manufacturers can achieve efficient construction of the tricyclic ketone structure while bypassing the logistical and safety challenges associated with carbon monoxide usage. The method demonstrates exceptional versatility across various substituted derivatives, ensuring broad applicability for drug discovery programs. As a reliable pharmaceutical intermediate supplier, understanding such patented advancements is crucial for maintaining a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indeno-[1,2-b]indole-10(5H)-one derivatives has relied heavily on palladium-catalyzed carbonylation reactions, which present significant operational and economic drawbacks. These conventional processes typically require the use of toxic carbon monoxide gas, necessitating specialized high-pressure equipment and rigorous safety protocols that increase capital expenditure. Furthermore, the reliance on expensive palladium acetate catalysts and specialized phosphine ligands, such as diadamantyl-n-butylphosphine, drives up the raw material costs substantially. The removal of residual heavy metals from the final product adds another layer of complexity to the downstream processing, often requiring additional purification steps that reduce overall yield. Additionally, the reaction conditions often involve high temperatures around 120°C in polar aprotic solvents, which can limit substrate scope and lead to thermal degradation of sensitive functional groups. These factors collectively hinder the cost reduction in pharmaceutical intermediate manufacturing and complicate the scale-up process for commercial quantities.
The Novel Approach
In stark contrast, the method disclosed in patent CN108503578B utilizes a zinc-mediated intramolecular coupling strategy that elegantly circumvents the need for transition metals and toxic gases. This novel approach involves the in situ generation of an indole zinc reagent from a 3-(2-bromobenzoyl)-indole precursor using alkyl lithium and zinc halides. The reaction proceeds through a controlled halogen-metal exchange that avoids the pitfalls of direct lithiation, ensuring high regioselectivity and minimizing side reactions. By operating under milder conditions and utilizing abundant reagents like n-butyllithium and zinc iodide, the process significantly lowers the barrier to entry for production facilities. The elimination of palladium not only reduces material costs but also simplifies the purification workflow, as there is no need for extensive heavy metal scavenging. This streamlined methodology represents a paradigm shift towards greener and more economically viable synthesis of complex heterocyclic intermediates.
Mechanistic Insights into Zinc-Mediated Intramolecular Coupling
The core of this synthetic breakthrough lies in the precise formation and reactivity of the organozinc intermediate, which serves as the key nucleophile in the cyclization step. The process initiates with the treatment of the bromo-substituted indole substrate with an alkyl lithium reagent at low temperatures, typically around -78°C, to generate a transient organolithium species. This species immediately undergoes transmetallation with zinc halides in the presence of lithium halides to form a stable indole zinc reagent, effectively preventing unwanted halogen-lithium exchange side reactions. The presence of lithium halides is critical as they enhance the solubility and reactivity of the organozinc species, facilitating a smooth subsequent transformation. Once formed, this zinc intermediate possesses the perfect nucleophilic character to attack the adjacent carbonyl group upon heating, driving the intramolecular cyclization forward. This mechanism ensures that the carbon-carbon bond formation occurs efficiently without the need for external oxidative additives or complex catalytic cycles.
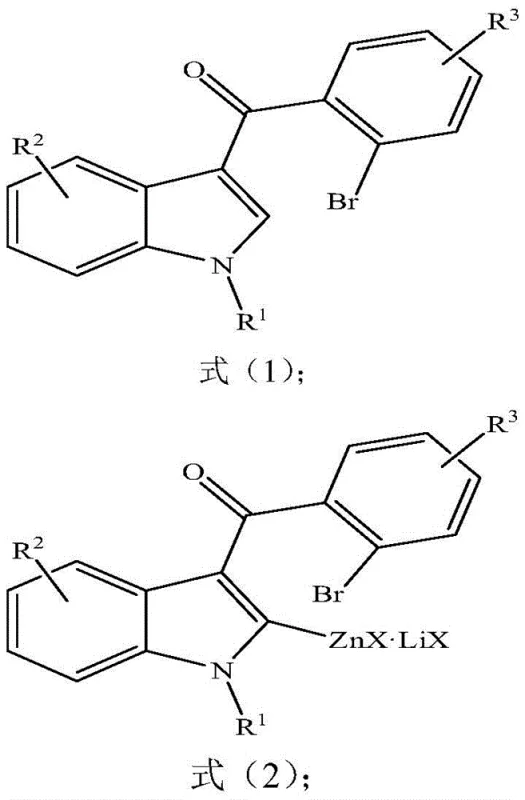
Furthermore, the impurity profile of this reaction is exceptionally clean due to the high chemoselectivity of the zinc reagent towards the ketone functionality. Unlike palladium-catalyzed methods that might suffer from homocoupling or incomplete carbonylation, this zinc-mediated pathway directs the reaction exclusively towards the desired tricyclic framework. The use of ether solvents for the initial metallation followed by the addition of aromatic hydrocarbons for the coupling step optimizes the solvation environment for each stage of the reaction. This biphasic solvent strategy helps in managing the exothermic nature of the lithiation while providing the necessary thermal energy for the cyclization step at elevated temperatures. Consequently, the resulting crude product contains fewer by-products, which translates to higher isolated yields after standard column chromatography. For R&D teams, this mechanistic clarity offers a robust platform for exploring diverse substitution patterns on the indole and benzoyl rings without compromising reaction efficiency.
How to Synthesize Indeno-Indole Ketones Efficiently
Implementing this synthetic route requires careful attention to reagent stoichiometry and temperature control to maximize the formation of the active zinc species. The protocol begins with the dissolution of the 3-(2-bromobenzoyl)-indole substrate in an anhydrous ether solvent, followed by the dropwise addition of n-butyllithium at cryogenic temperatures to ensure controlled deprotonation and metallation. Subsequent addition of zinc iodide and lithium iodide solutions allows for the rapid transmetallation to the organozinc intermediate, which is then stirred at room temperature to ensure complete conversion before the cyclization phase. The detailed standardized synthesis steps are provided below to guide process chemists in replicating these high-yielding results in their own laboratories.
- React 3-(2-bromobenzoyl)-indole with alkyl lithium, zinc halide, and lithium halide in ether solvent at low temperature to form the indole zinc reagent.
- Add aromatic hydrocarbon solvent and heat the mixture to facilitate intramolecular coupling.
- Quench the reaction, extract with organic solvent, and purify via column chromatography to obtain the target ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zinc-mediated synthesis offers tangible benefits that extend far beyond the laboratory bench. The primary advantage lies in the drastic simplification of the raw material portfolio, replacing scarce and volatile palladium catalysts with commodity chemicals that are readily available in bulk quantities. This shift mitigates the risk of supply disruptions caused by geopolitical instability affecting precious metal markets, thereby enhancing supply chain reliability for long-term production contracts. Moreover, the absence of toxic carbon monoxide gas removes the need for specialized gas handling infrastructure and reduces regulatory compliance burdens related to hazardous air pollutants. These operational improvements collectively contribute to substantial cost savings in pharmaceutical intermediate manufacturing without sacrificing product quality or throughput.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and specialized phosphine ligands directly lowers the bill of materials for every batch produced. By removing the necessity for heavy metal removal resins and complex purification trains, the downstream processing costs are also significantly reduced, leading to a more favorable cost of goods sold. The use of common solvents like tetrahydrofuran and toluene further aligns the process with standard industrial capabilities, avoiding the need for exotic or high-boiling solvents that are difficult to recover. This economic efficiency makes the production of high-purity indeno-indole ketones commercially viable even at smaller scales, enabling faster market entry for new drug candidates.
- Enhanced Supply Chain Reliability: Relying on abundant reagents such as zinc halides and alkyl lithiums ensures a stable supply base that is less susceptible to market fluctuations compared to precious metals. The simplified reaction setup reduces the dependency on specialized equipment vendors, allowing for greater flexibility in choosing contract manufacturing organizations. This resilience is critical for maintaining continuous production schedules and meeting tight delivery deadlines for downstream API synthesis. Additionally, the robustness of the reaction conditions minimizes the risk of batch failures, ensuring consistent output quality and quantity for partners relying on reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex heterocyclic intermediates, as it avoids the safety hazards associated with high-pressure carbon monoxide reactions. The waste stream is easier to manage since it does not contain heavy metal residues, simplifying wastewater treatment and disposal procedures in accordance with environmental regulations. The high atom economy of the intramolecular coupling means less waste generation per kilogram of product, supporting sustainability goals and green chemistry initiatives. This environmental compatibility facilitates smoother regulatory approvals and enhances the corporate social responsibility profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthetic method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide accurate guidance for potential adopters. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: What are the advantages of this zinc-mediated method over palladium-catalyzed routes?
A: This method eliminates the need for expensive palladium catalysts and toxic carbon monoxide gas, significantly reducing raw material costs and simplifying safety protocols while maintaining high yields.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses readily available reagents like n-butyllithium and zinc iodide, avoids complex ligand systems, and features simple post-processing steps, making it highly scalable for commercial manufacturing.
Q: What is the typical purity profile of the resulting indeno-indole ketones?
A: The method achieves high conversion rates with minimal side reactions due to the controlled formation of the organozinc intermediate, allowing for the production of high-purity intermediates suitable for pharmaceutical applications after standard purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indeno-Indole Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN108503578B for developing next-generation therapeutic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from gram-scale research to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of indeno-indole ketone intermediates meets the highest industry standards. Our commitment to technical excellence allows us to navigate complex chemical landscapes and deliver solutions that optimize both performance and cost.
We invite you to contact our technical procurement team to discuss how we can support your specific requirements with a Customized Cost-Saving Analysis tailored to your project needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that will accelerate your development timeline. Let us help you leverage this innovative chemistry to secure a reliable supply of critical intermediates for your pharmaceutical pipeline.
