Scalable Synthesis of Alpha-Halogenated Trifluoromethyl Alkanes via Manganese-Mediated Radical Functionalization
Scalable Synthesis of Alpha-Halogenated Trifluoromethyl Alkanes via Manganese-Mediated Radical Functionalization
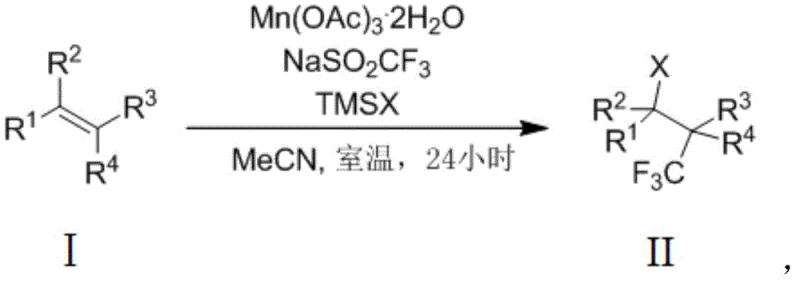
The introduction of trifluoromethyl groups into organic molecules is a cornerstone strategy in modern medicinal chemistry, profoundly influencing the lipophilicity, metabolic stability, and bioavailability of drug candidates. Patent CN111303089B presents a groundbreaking methodology for the efficient construction of alpha-halogenated trifluoromethyl substituted alkanes, utilizing a manganese-mediated radical pathway that operates under exceptionally mild conditions. This technology addresses the critical industry demand for robust, scalable routes to fluorinated building blocks, moving away from expensive hypervalent iodine reagents or complex photocatalytic setups. By leveraging manganese triacetate dihydrate as a single-electron oxidant, the process facilitates the simultaneous trifluoromethylation and halogenation of non-activated olefins at room temperature. This represents a significant leap forward for reliable alpha-halogenated trifluoromethyl substituted alkane suppliers seeking to optimize their production pipelines for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethylated compounds has relied heavily on reagents such as Togni or Umemoto reagents, which, while effective, suffer from significant drawbacks including high cost, potential instability, and the generation of stoichiometric iodine-containing waste. Furthermore, many traditional radical trifluoromethylation protocols require harsh conditions, such as high temperatures or the use of toxic radical initiators, which can compromise the integrity of sensitive functional groups present in complex drug scaffolds. Photoredox catalysis, although popular, introduces engineering challenges related to light penetration and reactor scalability, often limiting its utility in large-scale cost reduction in pharmaceutical intermediates manufacturing. Additionally, achieving regioselective halogenation alongside trifluoromethylation typically requires multi-step sequences, increasing the overall process mass intensity and extending production lead times unnecessarily.
The Novel Approach
The methodology disclosed in CN111303089B circumvents these issues by employing a straightforward, one-pot procedure that utilizes commercially available and inexpensive reagents. The use of sodium trifluoromethanesulfinate (Langlois reagent) coupled with manganese triacetate allows for the generation of trifluoromethyl radicals under ambient conditions, eliminating the need for external heating or specialized irradiation equipment. This novel approach not only simplifies the operational workflow but also enhances safety profiles by avoiding hazardous peroxides or high-pressure conditions. The direct functionalization of the carbon-carbon double bond ensures high atom economy, while the concurrent installation of a halogen atom provides a strategic synthetic handle for subsequent derivatization. This streamlined process is ideally suited for the commercial scale-up of complex fluorinated intermediates, offering a pragmatic solution for industrial applications.
Mechanistic Insights into Manganese-Mediated Radical Trifluoromethylation
The reaction mechanism proceeds through a well-defined radical cascade initiated by the oxidation of sodium trifluoromethanesulfinate by manganese(III) species. Upon single-electron transfer, a trifluoromethyl radical is generated, which subsequently undergoes regioselective addition to the electron-rich double bond of the olefin substrate. This addition creates a transient carbon-centered radical intermediate, which is pivotal to the success of the transformation. Unlike traditional methods that might terminate via hydrogen abstraction, this system leverages the oxidizing power of the manganese species to convert the carbon radical into a carbocation. This oxidative step is crucial as it activates the intermediate for nucleophilic attack by the trimethylhalosilane reagent, ultimately delivering the alpha-halogenated product with high fidelity. Understanding this mechanistic nuance is vital for high-purity alpha-halogenated trifluoromethyl substituted alkanes production, as it explains the excellent chemoselectivity observed even in the presence of other oxidizable functionalities.
From an impurity control perspective, the mildness of the reaction conditions plays a decisive role in minimizing side reactions such as polymerization or over-oxidation of the substrate. The use of acetonitrile as a polar aprotic solvent stabilizes the ionic intermediates involved in the final trapping step, ensuring clean conversion to the desired halide. Furthermore, the stoichiometry of the oxidant is carefully balanced to prevent the degradation of the trifluoromethyl group or the olefin backbone. The resulting products feature a halogen atom at the alpha-position relative to the trifluoromethyl group, a structural motif that is highly valued for its ability to undergo diverse cross-coupling or substitution reactions. This mechanistic robustness ensures that the process can be reliably transferred from laboratory benchtop to pilot plant scales without significant loss in yield or purity.
How to Synthesize Alpha-Halogenated Trifluoromethyl Substituted Alkanes Efficiently
The practical implementation of this synthesis involves a simple charging sequence where the olefin, oxidant, trifluoromethyl source, and halogen source are combined in acetonitrile. The reaction proceeds spontaneously at room temperature over a 24-hour period, requiring minimal operator intervention. Following the reaction, a standard aqueous workup involving sodium thiosulfate quenching effectively removes residual oxidants and manganese salts. The detailed standardized synthesis steps, including specific molar ratios and purification parameters, are outlined below to ensure reproducibility and compliance with GMP standards for reducing lead time for high-purity fluorinated building blocks.
- Combine olefin substrate, manganese triacetate dihydrate oxidant, sodium trifluoromethanesulfinate, and trimethylhalosilane in acetonitrile solvent.
- Stir the reaction mixture at room temperature for 24 hours to allow radical addition and subsequent halogen capture.
- Quench with aqueous sodium thiosulfate, extract with ethyl acetate, wash, dry, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this manganese-mediated protocol offers tangible benefits regarding raw material sourcing and inventory management. The reliance on commodity chemicals such as manganese triacetate and sodium trifluoromethanesulfinate eliminates the supply chain bottlenecks often associated with proprietary or custom-synthesized fluorinating agents. This shift towards widely available reagents significantly de-risks the procurement process, ensuring continuity of supply even during market fluctuations. Moreover, the elimination of expensive transition metal catalysts or specialized ligands drastically reduces the bill of materials, directly contributing to lower COGS (Cost of Goods Sold) for the final API intermediate.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily driven by the replacement of costly hypervalent iodine reagents with inexpensive manganese salts. By operating at room temperature, the process also eliminates the energy costs associated with heating or cooling reactors, leading to substantial utility savings. The simplified workup procedure, which avoids complex extraction or distillation steps required for removing volatile fluorinating agents, further reduces processing time and labor costs. These factors collectively enable a more competitive pricing structure for trifluoromethylated intermediates without compromising on quality or yield.
- Enhanced Supply Chain Reliability: The stability and shelf-life of the key reagents, particularly sodium trifluoromethanesulfinate and trimethylhalosilane, allow for bulk purchasing and long-term storage, mitigating the risk of stockouts. Since the reaction does not depend on sensitive photocatalysts that may degrade upon exposure to light or air, the logistics of transporting and storing reaction kits are simplified. This robustness ensures that manufacturing schedules can be maintained consistently, providing downstream partners with reliable delivery timelines for critical drug substance precursors.
- Scalability and Environmental Compliance: Scaling this reaction is straightforward due to the absence of exothermic hazards typically associated with radical initiators, allowing for safe operation in larger vessel sizes. The use of acetonitrile, a solvent with well-established recovery and recycling protocols, aligns with green chemistry principles and facilitates waste stream management. Additionally, the manganese byproducts are generally less toxic and easier to treat than heavy metal residues from palladium or rhodium catalysis, simplifying environmental compliance and reducing waste disposal fees.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process capabilities and product specifications. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using manganese triacetate over photoredox catalysts for trifluoromethylation?
A: Manganese triacetate operates under mild thermal conditions without requiring specialized light equipment, significantly simplifying reactor setup and reducing energy costs compared to photoredox systems.
Q: Can this method be applied to non-activated alkenes?
A: Yes, the patent specifically highlights the efficiency of this method for non-activated alkenes, generating alpha-halogenated trifluoromethyl products with good yields under standard conditions.
Q: How does the presence of the alpha-halogen benefit downstream synthesis?
A: The alpha-halogen serves as a versatile handle for further nucleophilic substitution, allowing for the rapid diversification of the trifluoromethylated scaffold into complex drug candidates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Halogenated Trifluoromethyl Substituted Alkane Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of fluorinated intermediates in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-halogenated trifluoromethyl alkane meets the exacting standards required by global regulatory bodies. Our commitment to quality assurance ensures that our clients receive materials that are ready for immediate use in sensitive downstream coupling reactions.
We invite you to collaborate with us to leverage this innovative manganese-mediated technology for your specific project needs. Our experts are prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your overall budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your drug development timeline with superior fluorinated building blocks.
