Advanced Manufacturing of 6-Ketoestradiol: A Cost-Effective Route for Anti-Estrogen Drug Intermediates
The pharmaceutical industry's relentless pursuit of effective endocrine therapies has placed significant emphasis on the efficient production of pure anti-estrogen agents, particularly for the treatment of breast cancer. Patent CN110669089B, published in June 2022, introduces a groundbreaking synthesis method for 6-ketoestradiol, a critical intermediate in the manufacture of fulvestrant and related 7α-substituted estradiol derivatives. This technical disclosure represents a paradigm shift from traditional, environmentally hazardous pathways to a streamlined, fermentation-based approach. By leveraging advanced oxidative strategies and avoiding toxic heavy metals, this innovation addresses the dual challenges of cost-efficiency and regulatory compliance in steroid hormone manufacturing. For global supply chain leaders, this patent offers a viable roadmap to secure high-purity intermediates while mitigating the risks associated with legacy chemical processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-ketoestradiol has relied heavily on estrone as the primary starting material, a strategy fraught with economic and environmental inefficiencies. As illustrated in the traditional reaction pathways, early methods employed chromium anhydride for oxidation steps, a process notorious for its poor selectivity and generation of substantial toxic waste. The reliance on chromium not only complicates wastewater treatment due to heavy metal contamination but also results in mediocre yields, often hovering around merely 24.4%, which is economically unsustainable for large-scale operations. Furthermore, alternative routes attempting to improve selectivity through borate ester intermediates necessitate the use of highly reactive and dangerous reagents such as butyl lithium. These conventional approaches increase production risks, require stringent safety protocols, and ultimately drive up the cost of goods sold due to complex purification requirements and low overall throughput.
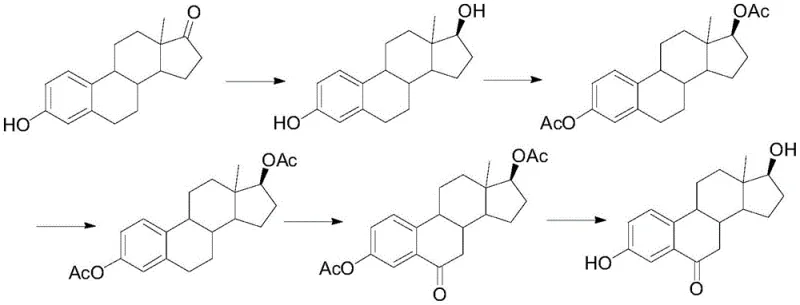
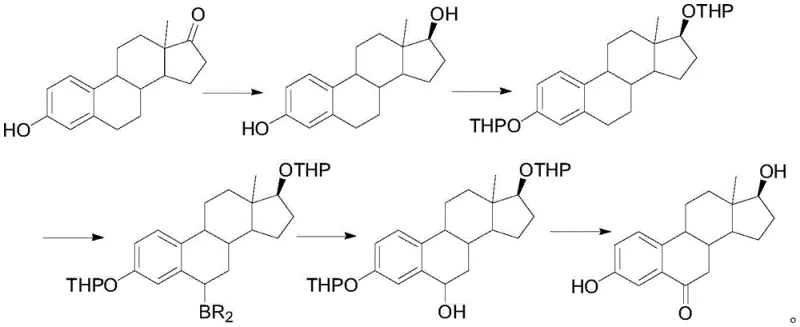
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes Compound 1, derived from 9-OH-AD obtained via phytosterol fermentation, as the foundational raw material. This strategic pivot away from expensive drug-grade estrone to fermentation-derived precursors significantly lowers the entry cost for raw materials while ensuring a stable supply chain. The new route is characterized by a logical sequence of reduction, selective oxidation, aromatization, hydrogenation, and final oxidative hydrolysis. By introducing the carbonyl group at the 6-position through a highly selective oxidation of an esterified intermediate, the process achieves superior regioselectivity. This eliminates the need for cumbersome separation of isomers and allows for milder reaction conditions that are inherently safer and more amenable to green chemistry principles, marking a substantial upgrade in process reliability.
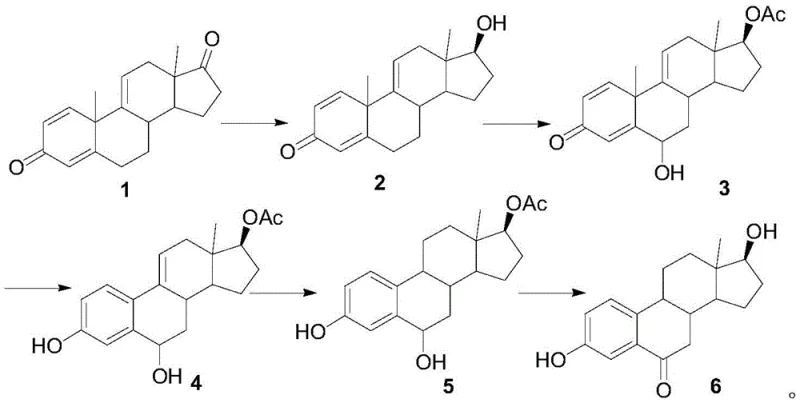
Mechanistic Insights into Selective Oxidation and Aromatization
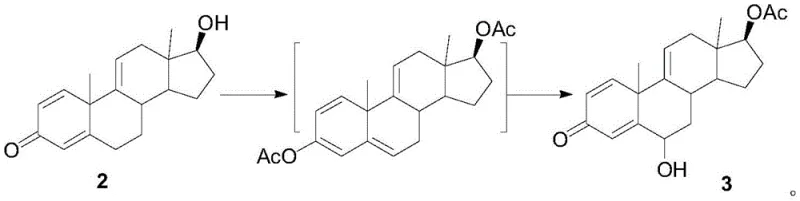
The core technical breakthrough of this synthesis lies in the precise control of oxidation states and the strategic protection of functional groups. In the second step, Compound 2 undergoes esterification followed by oxidation using meta-chloroperoxybenzoic acid (m-CPBA). This specific choice of oxidant is critical; unlike harsh chromium reagents, m-CPBA facilitates a controlled epoxidation or hydroxylation at the desired position without degrading the sensitive steroid backbone. The subsequent aromatization step utilizes activated zinc powder in a high-boiling solvent like diethylene glycol monoethyl ether at approximately 120°C. This reductive aromatization effectively removes the oxygen functionality introduced in previous steps to establish the aromatic A-ring, a structural prerequisite for estrogenic activity. The mechanism ensures that the 6-keto functionality remains intact while the rest of the molecule is stabilized, demonstrating a sophisticated understanding of steroid reactivity patterns.
Furthermore, the final transformation involves the use of 2-iodoxybenzoic acid (IBX) in dimethyl sulfoxide (DMSO), a modern oxidation system known for its tolerance of various functional groups. This step converts the intermediate alcohol to the ketone with high fidelity. The subsequent hydrolysis under basic conditions cleaves the acetate protecting group to reveal the final 6-ketoestradiol structure. The entire sequence is designed to minimize side reactions; for instance, the hydrogenation step specifically targets the 9,11-double bond using palladium on carbon, ensuring that the newly formed aromatic ring and the 6-keto group are not inadvertently reduced. This level of chemoselectivity is paramount for achieving the high purity levels (>98%) required for pharmaceutical intermediates intended for oncology applications.
How to Synthesize 6-Ketoestradiol Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing temperature control and reagent stoichiometry to maximize yield. The process begins with the reduction of the starting ketone using sodium borohydride at low temperatures (0-10°C) to prevent over-reduction or epimerization. Following isolation, the intermediate is subjected to the critical oxidation sequence using isopropenyl acetate and m-CPBA, where maintaining the temperature between 0-10°C during oxidant addition is crucial for safety and selectivity. The detailed standardized synthesis steps below outline the specific workup procedures, including quenching with sodium bisulfite and crystallization techniques, which are essential for removing impurities and achieving the target purity specifications.
- Perform reduction of Compound 1 using sodium borohydride in THF/methanol at 0-10°C to obtain Compound 2.
- Execute oxidation and esterification of Compound 2 using isopropenyl acetate and m-CPBA to generate Compound 3.
- Conduct aromatization of Compound 3 using zinc powder in pyridine or diethylene glycol monoethyl ether at elevated temperatures.
- Hydrogenate the 9,11-double bond of Compound 4 using Pd/C catalyst to form Compound 5.
- Finalize synthesis via oxidative hydrolysis of Compound 5 using IBX and DMSO followed by base hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling strategic benefits that extend beyond simple unit cost reductions. By shifting the raw material base from estrone to fermentation-derived compounds, manufacturers can decouple their supply chains from the volatility of the hormone extraction market. This diversification ensures greater continuity of supply and protects against price spikes associated with animal-sourced starting materials. Additionally, the elimination of chromium-based reagents simplifies the environmental compliance landscape, reducing the burden of hazardous waste disposal and lowering the operational costs associated with effluent treatment facilities. These factors collectively contribute to a more resilient and cost-effective manufacturing ecosystem.
- Cost Reduction in Manufacturing: The substitution of expensive estrone with widely available fermentation products fundamentally alters the cost structure of the synthesis. Moreover, the high selectivity of the m-CPBA oxidation step drastically reduces the formation of by-products, which minimizes the loss of valuable material during purification. The avoidance of transition metal catalysts that require complex removal steps, such as chromium, further streamlines the downstream processing, leading to substantial savings in both time and resources without compromising the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: Sourcing raw materials from fermentation processes offers a distinct advantage in terms of scalability and consistency compared to extraction from natural sources. The reagents used in the new pathway, such as zinc powder and sodium borohydride, are commodity chemicals with stable global supply lines, reducing the risk of production stoppages due to reagent shortages. This reliability is critical for meeting the rigorous delivery schedules demanded by multinational pharmaceutical companies developing life-saving oncology treatments.
- Scalability and Environmental Compliance: The process conditions described, such as the use of ethanol and THF as solvents and the avoidance of pyrophoric reagents like butyl lithium, make this route inherently safer for scale-up to multi-ton production. The green chemistry profile, characterized by the absence of heavy metal contaminants, aligns perfectly with the increasingly stringent environmental regulations governing pharmaceutical manufacturing. This compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived directly from the comparative data and experimental examples provided in the patent documentation, offering clarity on yield expectations, safety profiles, and purity standards. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the primary advantages of the new synthesis route over traditional estrone-based methods?
A: The new route utilizes fermentation-derived starting materials which are significantly cheaper and more abundant than estrone. Furthermore, it eliminates the use of toxic chromium oxidants, resulting in higher selectivity, improved yields, and a much greener environmental profile suitable for industrial scale-up.
Q: How does the process ensure high selectivity at the 6-position?
A: By optimizing the reaction sequence, specifically through the use of m-CPBA oxidation on the esterified intermediate, the 6-position becomes the sole active site for oxidation. This strategic modification drastically reduces the formation of by-products compared to non-selective chromium oxidation.
Q: Is this synthesis method scalable for commercial API production?
A: Yes, the patent explicitly designs the method for industrial production. It employs robust reagents like zinc powder for aromatization and standard hydrogenation catalysts (Pd/C), avoiding hazardous reagents like butyl lithium, thereby ensuring safety and feasibility for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Ketoestradiol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation endocrine therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering 6-ketoestradiol with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the exacting standards required for clinical and commercial API synthesis. Our capability to implement complex steroid chemistry allows us to offer a reliable supply of this vital intermediate to the global market.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this fermentation-based pathway. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of innovation, quality, and reliability.
