Breakthrough Palladium-Catalyzed [7+2] Cycloaddition for Scalable Nine-Membered Ring Production
Breakthrough Palladium-Catalyzed [7+2] Cycloaddition for Scalable Nine-Membered Ring Production
The landscape of organic synthesis for medium-sized rings has long been challenged by entropic penalties and transannular strain, yet recent advancements documented in patent CN109851607B offer a transformative solution for the pharmaceutical industry. This intellectual property introduces a highly efficient synthetic methodology for constructing 6-methylene-6,7-dihydrobenzo[e,h][1,4,7]dioxathionine 13-oxide nine-membered ring compounds through a novel palladium-catalyzed [7+2] cycloaddition reaction. By utilizing a robust catalytic system comprising palladium salts and specialized diphosphine ligands, this technology enables the direct assembly of complex heterocyclic frameworks from readily available 2,2'-sulfinyl diphenols and propargyl compounds. The significance of this development cannot be overstated for R&D directors seeking reliable pathways to bioactive scaffolds, as it achieves unprecedented yields of up to 99% while maintaining exceptional stereochemical control with Z/E ratios exceeding 25:1. This report analyzes the technical merits and commercial viability of this process, positioning it as a cornerstone for next-generation intermediate manufacturing.
![General reaction scheme showing the palladium-catalyzed [7+2] cycloaddition between sulfinyl diphenol and propargyl compound](/insights/img/nine-membered-ring-synthesis-palladium-catalysis-pharma-supplier-20260309114140-05.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of nine-membered rings has been fraught with synthetic difficulties due to the inherent thermodynamic instability and kinetic barriers associated with medium-ring formation. Conventional strategies typically rely on intramolecular cyclization or ring expansion reactions, which often suffer from competing pathways that favor the formation of more stable five or six-membered by-products. These traditional approaches frequently require harsh reaction conditions, such as high temperatures or strong Lewis acids, which can degrade sensitive functional groups and complicate downstream purification processes. Furthermore, the entropic cost of bringing two ends of a linear precursor together in an intramolecular fashion often results in dilute reaction conditions and poor atom economy. For procurement managers, these inefficiencies translate into higher raw material consumption, increased waste disposal costs, and unpredictable batch-to-batch consistency, creating significant bottlenecks in the supply chain for complex API intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in the patent leverages an intermolecular [7+2] cycloaddition strategy that fundamentally alters the reaction coordinate to favor nine-membered ring closure. By employing a palladium catalyst generated in situ from precursors like Pd2(dba)3 and ligands such as 1,1'-bis(diphenylphosphino)ferrocene, the reaction proceeds smoothly at room temperature in common solvents like tetrahydrofuran. This approach eliminates the need for high-energy activation, thereby preserving the integrity of diverse functional groups on both the phenolic and propargyl substrates. The use of a mild inorganic base, specifically cesium carbonate, facilitates the deprotonation and nucleophilic attack without inducing side reactions. For supply chain heads, this translates to a process that is not only safer to operate but also significantly more robust, allowing for the use of standard stainless steel reactors without the need for specialized corrosion-resistant linings or extreme pressure ratings.
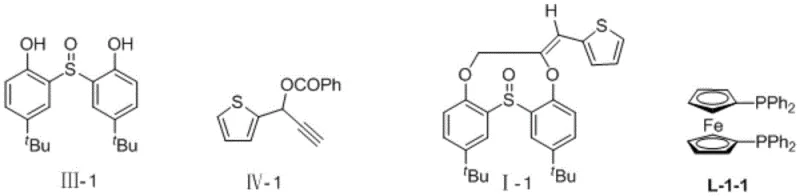
Mechanistic Insights into Palladium-Catalyzed [7+2] Cycloaddition
The catalytic cycle underpinning this transformation involves a sophisticated interplay between the palladium center and the bidentate phosphine ligand to orchestrate bond formation with high precision. Initially, the palladium(0) species undergoes oxidative addition with the propargyl ester substrate, generating a cationic pi-allyl palladium intermediate that is highly electrophilic. Concurrently, the 2,2'-sulfinyl diphenol, activated by the base, coordinates to the metal center, positioning the nucleophilic oxygen atoms for attack. The unique geometry imposed by the bulky diphosphine ligand, such as the ferrocene-based L-1 or binaphthyl-based L-2 derivatives, creates a chiral environment that directs the subsequent cyclization steps. This steric guidance is crucial for overcoming the entropic barrier of forming the nine-membered ring, effectively templating the transition state to favor the desired macrocyclization over oligomerization or smaller ring closure. The result is a highly selective process that minimizes the formation of regioisomers and simplifies the impurity profile, a critical factor for regulatory compliance in pharmaceutical manufacturing.
Furthermore, the mechanism ensures exceptional control over the stereochemistry of the exocyclic double bond formed during the elimination step. The data indicates a strong preference for the Z-isomer, with ratios consistently greater than 25:1 across a wide range of substrates. This high stereoselectivity is attributed to the specific conformational constraints within the palladium complex during the reductive elimination phase. For R&D teams, this means that the synthetic route delivers the target molecule with the correct spatial arrangement required for biological activity, reducing or eliminating the need for difficult chromatographic separations of geometric isomers. The ability to tolerate various substituents on the aromatic rings, including electron-withdrawing groups like halogens and electron-donating groups like alkyls, demonstrates the versatility of the electronic properties of the catalytic system. This broad substrate scope allows medicinal chemists to rapidly generate libraries of analogues for structure-activity relationship studies without redesigning the core synthetic pathway.
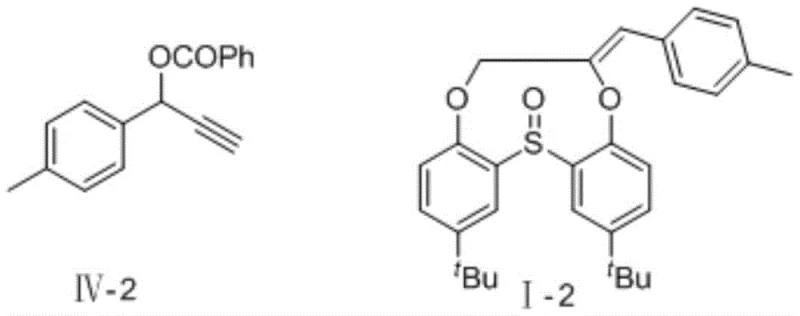
How to Synthesize 6-methylene-6,7-dihydrobenzo[e,h][1,4,7]dioxathionine 13-oxide Efficiently
Implementing this synthesis requires careful attention to the preparation of the active catalytic species and the stoichiometry of the reagents to ensure maximum conversion. The process begins with the generation of the catalyst under an inert atmosphere to prevent oxidation of the palladium(0) species, followed by the sequential addition of substrates and base. The reaction is remarkably forgiving, proceeding effectively at ambient temperature which reduces energy consumption and operational complexity. Detailed standard operating procedures regarding specific molar ratios, solvent drying techniques, and workup protocols are essential for reproducibility on a larger scale. The following guide outlines the critical operational parameters derived from the patent examples to assist process engineers in technology transfer.
- Prepare the chiral palladium catalyst by stirring palladium salt and diphosphine ligand in THF under nitrogen protection.
- Dissolve the propargyl compound, 2,2'-sulfinyl diphenol, and cesium carbonate base in anhydrous tetrahydrofuran.
- Add the substrate solution to the catalyst, stir at room temperature for 24 hours, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages that directly impact the bottom line and supply chain resilience for fine chemical manufacturers. The reliance on commodity chemicals such as substituted phenols and propargyl alcohols as starting materials ensures a stable and cost-effective supply base, mitigating the risks associated with sourcing exotic or proprietary reagents. The elimination of cryogenic conditions or high-pressure equipment drastically reduces capital expenditure requirements for plant setup, allowing existing multipurpose facilities to adopt this technology with minimal retrofitting. Moreover, the high atom economy and near-quantitative yields observed in the patent examples imply a substantial reduction in raw material waste, aligning with modern green chemistry principles and lowering disposal costs. For procurement managers, these factors combine to create a compelling value proposition characterized by lower cost of goods sold and improved margin potential for the final active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The operational simplicity of running reactions at room temperature in standard solvents like THF leads to significant energy savings compared to traditional high-temperature cyclizations. By avoiding the use of expensive transition metals beyond catalytic amounts and utilizing inexpensive inorganic bases, the overall reagent cost per kilogram of product is drastically optimized. The high selectivity of the reaction minimizes the loss of valuable intermediates to by-products, ensuring that the majority of input mass is converted into saleable product. This efficiency translates directly into a more competitive pricing structure for the final intermediate, providing a strategic advantage in tender negotiations with global pharmaceutical clients.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against variations in substrate electronics means that supply disruptions for specific substituted phenols can often be managed by switching to alternative analogues without re-optimizing the entire process. The use of stable palladium precursors and ligands that are commercially available in bulk quantities ensures that catalyst supply remains consistent even during market fluctuations. Additionally, the short reaction times and simple workup procedures involving standard silica gel chromatography allow for faster batch turnover rates. This agility enables manufacturers to respond more quickly to changes in demand, reducing lead times and improving service levels for downstream customers who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: Scaling this reaction from gram to multi-ton scale is facilitated by the absence of hazardous reagents and the exothermic nature of the reaction being manageable under ambient conditions. The process generates minimal hazardous waste streams, primarily consisting of aqueous salt solutions and spent silica, which are easier to treat and dispose of in compliance with stringent environmental regulations. The high purity of the crude product reduces the burden on purification units, lowering solvent consumption and the associated carbon footprint of the manufacturing process. These environmental benefits not only reduce compliance costs but also enhance the corporate sustainability profile, which is increasingly important for securing contracts with major multinational corporations committed to responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nine-membered ring synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential licensees or manufacturing partners. Understanding these nuances is critical for assessing the fit of this technology within existing production portfolios and R&D pipelines.
Q: What represents the primary advantage of this [7+2] cycloaddition over traditional ring expansion methods?
A: Traditional methods often suffer from thermodynamic and kinetic mismatches leading to stable small-ring by-products. This novel intermolecular [7+2] strategy bypasses those limitations, achieving yields up to 99% with exceptional Z/E selectivity (>25:1) under mild room temperature conditions.
Q: Which ligands are most effective for maximizing yield in this palladium catalytic system?
A: The patent data indicates that bidentate phosphine ligands such as 1,1'-bis(diphenylphosphino)ferrocene (dppf) and its derivatives (L-1, L-2 series) provide superior performance compared to monodentate ligands, ensuring high catalytic activity and stereocontrol.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the process utilizes easily obtainable raw materials, operates at ambient pressure and room temperature, and avoids hazardous reagents. These factors significantly simplify process engineering and safety protocols, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-methylene-6,7-dihydrobenzo[e,h][1,4,7]dioxathionine 13-oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of mastering complex ring-forming reactions to stay ahead in the competitive pharmaceutical intermediate market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to guarantee stringent purity specifications for every batch of nine-membered ring compounds we produce. Our commitment to quality assurance means that every shipment meets the exacting standards required for GMP-compliant API synthesis, giving our partners confidence in the consistency and reliability of their supply chain.
We invite forward-thinking pharmaceutical companies and contract research organizations to collaborate with us to leverage this cutting-edge synthetic technology for their drug development programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules. Let us help you accelerate your timeline to market with a supply partner dedicated to innovation, quality, and long-term mutual success.
