Revolutionizing Benzylamine Production: Iron-Catalyzed Direct Amination for Commercial Scale
Revolutionizing Benzylamine Production: Iron-Catalyzed Direct Amination for Commercial Scale
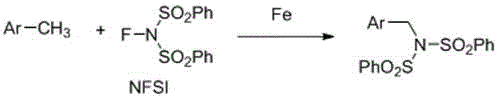
The landscape of organic synthesis is undergoing a paradigm shift towards sustainability and cost-efficiency, driven by the urgent need to replace expensive noble metal catalysts with earth-abundant alternatives. A pivotal development in this arena is documented in patent CN110003061B, which discloses a groundbreaking method for the direct amination of methyl aromatic compounds utilizing iron catalysis. This technology represents a significant leap forward in the synthesis of benzylamine derivatives, which are critical building blocks for numerous active pharmaceutical ingredients (APIs) and biologically active small molecules. By leveraging inexpensive iron compounds such as ferric oxalate hexahydrate in conjunction with N-fluorobisbenzenesulfonylimide (NFSI), this process achieves direct benzylic C-H functionalization without the necessity for external oxidants or complex ligand systems. For R&D directors and procurement managers alike, this innovation signals a move towards more robust, economical, and environmentally benign manufacturing protocols that can be seamlessly integrated into existing supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct amination of benzylic hydrocarbons has been fraught with significant technical and economic challenges that hinder large-scale adoption in the fine chemical industry. Traditional methodologies often rely on the generation of metal nitrenes, which necessitate the use of hazardous and explosive azide precursors or high-valent iodine compounds, posing severe safety risks during industrial operations. Alternatively, processes involving the additional addition of peroxides carry inherent explosion hazards, requiring specialized equipment and rigorous safety protocols that drive up capital expenditure. Furthermore, established catalytic systems frequently depend on toxic and costly copper or palladium catalysts, often requiring sophisticated and expensive phosphine ligands to achieve acceptable selectivity and yield. These legacy methods not only inflate the raw material costs but also complicate downstream processing due to the difficulty of removing trace heavy metal residues to meet stringent pharmaceutical purity standards, thereby creating bottlenecks in the production of reliable agrochemical intermediate and pharmaceutical supplies.
The Novel Approach
In stark contrast to these cumbersome traditional routes, the novel iron-catalyzed approach described in the patent data offers a streamlined and economically superior pathway for synthesizing valuable benzylamine scaffolds. This method ingeniously utilizes NFSI not merely as an amination reagent but effectively as an internal oxidant, thereby eliminating the need for any additional external oxidizing agents that could compromise safety or selectivity. The use of abundant and non-toxic iron salts, such as ferric oxalate or ferrocene derivatives, drastically reduces the catalyst cost profile while maintaining high catalytic activity across a diverse range of substrates, including substituted toluenes and 8-methylquinolines. By operating under ligand-free conditions in o-dichlorobenzene, the process simplifies the reaction mixture, facilitating easier workup and purification via standard silica gel column chromatography. This technological advancement directly addresses the pain points of cost reduction in pharmaceutical intermediate manufacturing by removing expensive metal inputs and hazardous reagents, offering a cleaner and more sustainable alternative for producing complex organic molecules.
Mechanistic Insights into Iron-Catalyzed Direct Amination
The mechanistic underpinning of this transformation relies on the unique redox properties of iron species interacting with the N-F bond of the NFSI reagent to generate reactive nitrogen-centered radicals. Upon heating in o-dichlorobenzene, the iron catalyst facilitates the homolytic cleavage of the N-F bond, initiating a radical chain process that selectively targets the benzylic C-H bond of the methyl aromatic substrate. This radical abstraction generates a stabilized benzylic radical intermediate, which subsequently couples with the nitrogen species to form the new C-N bond. The elegance of this mechanism lies in its ability to proceed without external oxidants, as the NFSI molecule itself serves the dual purpose of providing the nitrogen atom and balancing the redox cycle of the iron catalyst. This self-contained redox system minimizes side reactions and waste generation, ensuring a cleaner reaction profile that is highly desirable for the commercial scale-up of complex polymer additives and fine chemicals where impurity control is paramount.
From an impurity control perspective, the ligand-free nature of this catalytic system significantly reduces the complexity of the crude reaction mixture, thereby simplifying the purification burden on downstream processing units. In conventional transition metal catalysis, ligands often degrade or form stable complexes with the metal, creating difficult-to-remove impurities that can persist through multiple purification steps and contaminate the final API. However, the iron-catalyzed protocol described herein avoids these issues entirely, as the catalyst system consists of simple inorganic salts that can be easily separated or quenched. The broad substrate tolerance, accommodating electron-withdrawing and electron-donating groups alike, suggests a robust radical mechanism that is less sensitive to steric and electronic variations compared to concerted metalation-deprotonation pathways. This robustness ensures consistent product quality and high-purity OLED material or pharmaceutical intermediate output, reducing the risk of batch failures and enhancing overall process reliability for supply chain heads managing global distribution networks.
How to Synthesize Benzylamine Derivatives Efficiently
The practical implementation of this synthesis route involves a straightforward procedure that is amenable to both laboratory optimization and industrial replication. The process begins by charging a reaction vessel with the methyl aromatic substrate, a catalytic amount of an iron salt such as ferric oxalate hexahydrate, and o-dichlorobenzene as the solvent medium. Subsequently, N-fluorobisbenzenesulfonylimide (NFSI) is added in a stoichiometric excess to drive the reaction to completion, typically requiring a molar ratio of substrate to NFSI between 1:1 and 1:3. The detailed standardized synthesis steps, including precise temperature controls, stirring rates, and specific workup procedures for maximizing yield, are outlined in the guide below for technical teams seeking to adopt this methodology.
- Charge a reaction vessel with methyl aromatic compound (e.g., toluene derivative), iron catalyst (e.g., ferric oxalate hexahydrate), and o-dichlorobenzene solvent.
- Add N-fluorobisbenzenesulfonylimide (NFSI) to the mixture at a molar ratio of 1: 1.5 relative to the substrate.
- Heat the mixture to reflux for 4-28 hours, then purify the resulting benzylamine derivative via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iron-catalyzed technology translates into tangible strategic advantages that extend far beyond simple reagent substitution. The elimination of precious metals like palladium and toxic copper removes a major source of price volatility and supply risk, as iron salts are globally abundant and subject to minimal geopolitical supply constraints. Furthermore, the removal of hazardous oxidants and explosive azides from the process inventory significantly lowers insurance premiums and regulatory compliance costs associated with handling dangerous goods. This shift towards safer chemistry not only protects the workforce but also streamlines logistics and storage requirements, allowing for more flexible and resilient supply chain operations that can withstand market fluctuations without compromising production schedules.
- Cost Reduction in Manufacturing: The economic impact of switching to this iron-catalyzed system is profound, primarily driven by the drastic reduction in raw material costs associated with catalysts and reagents. By replacing expensive noble metals and complex ligands with commodity iron salts, manufacturers can achieve substantial cost savings on the bill of materials, which directly improves gross margins for high-volume products. Additionally, the simplified workup procedure, which avoids the need for specialized scavengers to remove heavy metal residues, reduces the consumption of auxiliary chemicals and shortens the overall cycle time per batch. These efficiencies compound over large-scale production runs, delivering significant financial benefits that enhance competitiveness in the global market for fine chemical intermediates without sacrificing product quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is critically dependent on the availability and stability of key raw materials, and this technology secures that foundation by utilizing universally available iron compounds. Unlike specialized catalysts that may have long lead times or single-source suppliers, iron salts are produced by multiple vendors worldwide, ensuring a redundant and robust supply network that mitigates the risk of shortages. The stability of the reagents also allows for longer shelf life and simpler storage conditions, reducing waste from expired materials and enabling just-in-time inventory strategies. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug manufacturers receive their materials on schedule to maintain their own production timelines.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process aligns perfectly with modern green chemistry principles and increasingly stringent regulatory frameworks. The absence of toxic heavy metals simplifies wastewater treatment and waste disposal, lowering the environmental footprint of the manufacturing facility and reducing fees associated with hazardous waste management. The reaction conditions are mild and do not require extreme pressures or temperatures, making the process inherently safer and easier to scale from pilot plant to multi-ton commercial production. This ease of scale-up ensures that the technology can meet growing market demand for specialty chemicals while maintaining compliance with environmental regulations, positioning the manufacturer as a responsible and sustainable partner in the global value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed amination technology, based on the specific data and embodiments provided in the patent literature. These insights are designed to clarify the operational parameters and potential benefits for stakeholders evaluating this route for their specific product portfolios. Understanding these nuances is crucial for making informed decisions about process adoption and supply chain integration.
Q: What are the primary advantages of using iron catalysts over copper or palladium for benzyl amination?
A: Iron catalysts are significantly cheaper, non-toxic, and environmentally friendly compared to expensive noble metals like palladium or toxic copper salts. Furthermore, this specific iron-catalyzed method eliminates the need for additional oxidants or complex ligands, simplifying the purification process and reducing overall production costs.
Q: What types of substrates are compatible with this direct amination method?
A: The method demonstrates broad substrate scope, successfully converting various toluene derivatives (including those with halogen, cyano, carbonyl, and nitro groups) as well as 8-methylquinoline derivatives into their corresponding benzylamine products with yields ranging from 47% to 80%.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process uses common solvents like o-dichlorobenzene and stable iron salts, avoiding explosive azides or peroxides. The absence of sensitive ligands and the robustness of the iron catalyst make it highly suitable for commercial scale-up in fine chemical and pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzylamine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed direct amination technology and possess the technical expertise to bring it from patent literature to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of benzylamine derivatives meets the highest international standards. Our commitment to quality and process excellence makes us the ideal partner for pharmaceutical and agrochemical companies seeking to optimize their supply chains with cutting-edge synthetic methodologies.
We invite you to collaborate with us to explore how this innovative synthesis route can be tailored to your specific production needs, offering a pathway to reduced costs and enhanced sustainability. Please contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your target molecules. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals and secure your supply of critical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
