Advanced Ruthenium Catalysis for Scalable Polysubstituted 1-Naphthoic Acid Production
Advanced Ruthenium Catalysis for Scalable Polysubstituted 1-Naphthoic Acid Production
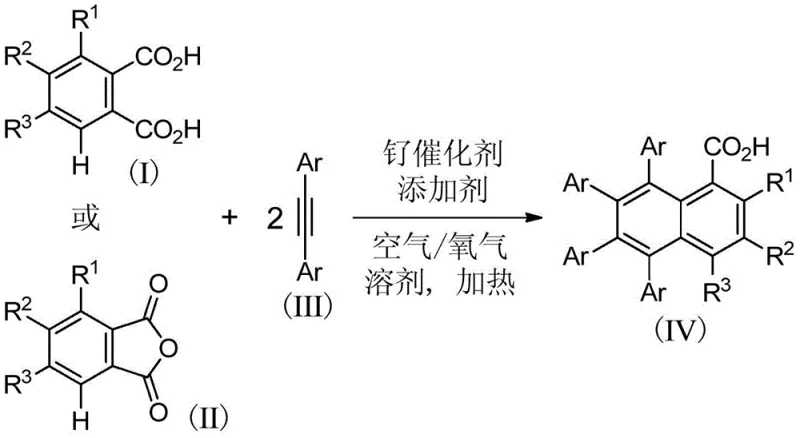
The landscape of organic synthesis for complex carbocyclic compounds is undergoing a significant transformation driven by the urgent need for greener, more atom-economical processes. Patent CN109665958B introduces a groundbreaking methodology for the preparation of polysubstituted 1-naphthoic acid compounds, utilizing a highly efficient ruthenium catalytic system. This technology addresses critical bottlenecks in the production of bioactive scaffolds found in anticancer agents like neoazinomatin and bile acid receptor agonists. By leveraging the unique reactivity of ruthenium complexes, the process enables the direct coupling of readily available phthalic acid or phthalic anhydride derivatives with symmetrical diaryl alkynes. The reaction proceeds under mild aerobic conditions, utilizing air or oxygen as the terminal oxidant, which fundamentally alters the environmental footprint of synthesizing these valuable pharmaceutical intermediates.
For procurement specialists and supply chain managers, the implications of this patent extend far beyond the laboratory bench. The shift away from stoichiometric metal oxidants and hazardous reagents towards a catalytic system driven by atmospheric oxygen represents a substantial opportunity for cost reduction in pharmaceutical intermediate manufacturing. The reliance on commodity chemicals like phthalic acid ensures a robust and continuous supply chain, mitigating risks associated with exotic starting materials. Furthermore, the operational simplicity of the reaction, which tolerates a wide range of functional groups without the need for protecting groups or complex substrate pre-functionalization, streamlines the path from discovery to commercial production. This report analyzes the technical depth of this innovation and its strategic value for global chemical sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted naphthoic acid derivatives has been plagued by significant inefficiencies and environmental concerns inherent to transition metal catalysis. Traditional approaches often rely on rhodium catalysts which, while effective, necessitate the use of equivalent amounts of copper acetate as a stoichiometric oxidant. This requirement generates substantial quantities of heavy metal waste, complicating downstream purification and increasing waste disposal costs dramatically. Alternative strategies employing palladium catalysis typically demand ortho-brominated benzoate substrates, which are not only more expensive to source but also limit the structural diversity of the final product due to the strict requirement for halogenation at specific positions. Additionally, nickel-catalyzed routes frequently involve the tedious installation and subsequent removal of bidentate directing groups, adding at least two extra synthetic steps that erode overall yield and increase process time. These legacy methods suffer from poor atom economy and pose significant challenges for scaling up to industrial volumes due to safety hazards associated with strong chemical oxidants.
The Novel Approach
The methodology disclosed in CN109665958B offers a transformative solution by employing a ruthenium-catalyzed double alkyne cyclization coupled with decarboxylation. This novel approach eliminates the need for stoichiometric metal oxidants by utilizing molecular oxygen from air, a truly green and inexhaustible resource. The reaction directly activates the carbon-hydrogen bond adjacent to the carboxyl group in phthalic acid derivatives, facilitating a cascade of cyclization and decarboxylation events in a single pot. This telescoped process significantly reduces the number of unit operations required, leading to a drastic simplification of the workflow. The use of inexpensive and widely available phthalic acid or phthalic anhydride as the starting scaffold ensures that raw material costs remain low and supply is secure. Moreover, the reaction conditions are remarkably mild, typically operating at temperatures between 90°C and 120°C, which reduces energy consumption compared to high-temperature pyrolysis methods. This combination of green chemistry principles and operational efficiency makes the new route superior for sustainable manufacturing.
Mechanistic Insights into Ruthenium-Catalyzed Oxidative Cyclization
At the heart of this technological breakthrough lies a sophisticated catalytic cycle driven by the unique electronic properties of the ruthenium center. The mechanism initiates with the coordination of the ruthenium catalyst to the carboxyl group of the phthalic acid derivative, which acts as a weak directing group to facilitate ortho-carbon-hydrogen bond activation. This activation step generates a reactive ruthenacycle intermediate that is poised for alkyne insertion. Unlike other systems that might stall or require harsh conditions, the ruthenium species efficiently inserts two molecules of the symmetrical diaryl alkyne into the metal-carbon bond. This sequential insertion builds the carbon framework of the naphthalene ring system with high regioselectivity. The presence of the second carboxyl group in the phthalic acid substrate plays a crucial dual role: it enhances the acidity of the substrate to promote initial coordination and participates in the stabilization of intermediates, ensuring the reaction proceeds smoothly without the need for external strong bases or aggressive activators.
Following the double alkyne insertion, the catalytic cycle culminates in an oxidative cyclization and decarboxylation sequence that releases the final polysubstituted 1-naphthoic acid product. The use of air or oxygen as the oxidant regenerates the active ruthenium species, closing the catalytic loop without generating stoichiometric waste. This mechanistic pathway is highly tolerant of diverse electronic environments, allowing for the incorporation of electron-donating groups like methoxy and alkyl chains, as well as electron-withdrawing groups such as halogens, trifluoromethyl, and nitro groups. The versatility extends to heterocyclic alkynes, as evidenced by the successful synthesis of thienyl-substituted derivatives. This broad substrate scope is critical for medicinal chemists who require rapid access to diverse analog libraries for structure-activity relationship studies. The ability to control impurity profiles through precise tuning of the catalyst loading and additive selection further underscores the robustness of this mechanistic design.
How to Synthesize Polysubstituted 1-Naphthoic Acid Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol utilizes standard laboratory glassware and does not require specialized high-pressure reactors, as the reaction can be conducted under an ambient air atmosphere or with a simple oxygen balloon. The solvent system, preferably a mixture of gamma-valerolactone and DMF, provides an optimal balance of polarity and boiling point to maintain catalyst stability and substrate solubility throughout the extended heating period. Workup procedures are straightforward, involving simple aqueous extraction and standard silica gel chromatography, which facilitates the isolation of high-purity products suitable for downstream applications. For detailed operational parameters and specific stoichiometric ratios optimized for different substrate classes, please refer to the standardized synthesis guide below.
- Combine phthalic acid or phthalic anhydride with symmetrical diaryl alkynes, a ruthenium catalyst such as p-cymene ruthenium dichloride dimer, and a bromide additive in a polar aprotic solvent mixture.
- Heat the reaction mixture to temperatures between 90°C and 120°C under an air or oxygen atmosphere for 6 to 30 hours to facilitate oxidative cyclization and decarboxylation.
- Upon completion, cool the mixture, extract with ethyl acetate, wash with water, and purify the crude product via silica gel column chromatography to isolate the target naphthoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ruthenium-catalyzed technology offers compelling advantages that directly impact the bottom line and supply chain resilience. The elimination of expensive stoichiometric oxidants like copper acetate removes a significant cost driver and simplifies waste management protocols, leading to substantial cost savings in pharmaceutical intermediate manufacturing. The reliance on commodity feedstocks such as phthalic acid and commercially available alkynes ensures that production is not bottlenecked by the availability of custom-synthesized precursors. This stability is crucial for long-term supply agreements and helps mitigate price volatility in the raw material market. Furthermore, the high catalytic activity allows for lower catalyst loadings relative to the substrate, optimizing the usage of the precious metal and reducing the burden on metal scavenging processes during purification.
- Cost Reduction in Manufacturing: The transition to an aerobic oxidation system fundamentally changes the cost structure of producing polysubstituted 1-naphthoic acids. By replacing costly and hazardous chemical oxidants with free atmospheric oxygen, the process eliminates the purchase cost of these reagents and the associated disposal fees for heavy metal waste. Additionally, the avoidance of directing group installation and removal steps, which are common in competing nickel-catalyzed methods, reduces the total number of synthetic steps. Fewer steps translate directly to higher overall yields, reduced solvent consumption, and lower labor costs per kilogram of product. The use of relatively inexpensive ruthenium catalysts compared to rhodium or palladium alternatives further contributes to a more favorable economic profile for large-scale production runs.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for pharmaceutical manufacturers, and this process excels by utilizing widely sourced starting materials. Phthalic acid and its anhydride are produced on a massive global scale for the plastics and resin industries, ensuring a stable and abundant supply that is unlikely to face shortages. Similarly, the symmetrical diaryl alkynes required for the reaction are accessible through established supply chains or can be synthesized via standard coupling reactions. The robustness of the reaction conditions, which tolerate moisture and air to a significant degree, reduces the need for stringent inert atmosphere controls, thereby simplifying facility requirements and reducing the risk of batch failures due to environmental excursions. This reliability makes the process ideal for securing long-term contracts for critical drug intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but this ruthenium-catalyzed method has demonstrated successful gram-scale synthesis with consistent yields, indicating strong potential for tonnage production. The use of green solvents like gamma-valerolactone, which is derived from renewable biomass, aligns with increasingly strict environmental regulations and corporate sustainability goals. The absence of toxic heavy metal waste streams simplifies the environmental permitting process and reduces the liability associated with hazardous waste disposal. The mild reaction temperatures and atmospheric pressure operation minimize energy consumption and safety risks, making the process inherently safer and easier to scale in existing multipurpose chemical plants without requiring major capital investment in specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this technology into your current manufacturing portfolio.
Q: What are the primary advantages of this ruthenium-catalyzed method over traditional rhodium or palladium systems?
A: Unlike rhodium systems that require stoichiometric copper oxidants or palladium methods needing ortho-halogenated substrates, this ruthenium protocol utilizes ambient air as a green oxidant and readily available phthalic acid starting materials, significantly reducing heavy metal waste and raw material costs.
Q: Does this synthesis require the installation and removal of directing groups?
A: No, a key innovation of this patent is that it avoids the extra synthetic steps associated with installing bidentate directing groups required in nickel-catalyzed approaches, thereby improving overall atom economy and process efficiency.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent demonstrates successful gram-scale synthesis with high yields and utilizes commercially available solvents and catalysts, indicating strong potential for commercial scale-up without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted 1-Naphthoic Acid Supplier
As the demand for complex aromatic scaffolds continues to rise in the pharmaceutical and agrochemical sectors, having a partner with deep technical expertise in advanced catalytic processes is essential. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of ruthenium-catalyzed reactions, ensuring that stringent purity specifications are met for every batch. With our rigorous QC labs and commitment to green chemistry principles, we are uniquely positioned to deliver high-quality polysubstituted 1-naphthoic acid derivatives that meet the exacting standards of global regulatory bodies.
We invite you to explore how this cutting-edge synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. By collaborating with us, you gain access to a reliable partner dedicated to driving efficiency and innovation in your chemical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
