Advanced Cobalt-Catalyzed Synthesis of Chiral 1,5-Diboron Intermediates for Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex chiral scaffolds with high efficiency and stereocontrol. Patent CN114957304A introduces a groundbreaking advancement in this domain by disclosing a novel synthesis method for chiral 1,5-diborane compounds, specifically utilizing an asymmetric double hydroboration strategy. This technology addresses a significant gap in organic synthesis, where the direct construction of 1,5-difunctionalized chiral skeletons has historically been challenging due to issues with regioselectivity and the thermodynamic stability of intermediates. By employing a specialized chiral cobalt catalyst system, this invention enables the transformation of readily available vinylcyclopropane derivatives into valuable chiral 1,5-diboryl alkanes. The significance of this development extends beyond academic interest, offering a practical route for generating high-purity pharmaceutical intermediates that were previously difficult to procure at scale.
Furthermore, the versatility of the resulting 1,5-diboron products allows for diverse downstream functionalization, including oxidation to 1,5-diols and amination to 1,5-diamines. This modularity is crucial for process chemists aiming to streamline synthetic routes for active pharmaceutical ingredients (APIs). The patent explicitly highlights the successful application of this methodology in the synthesis of (-)-Preclamol, a known dopamine agonist, demonstrating the tangible utility of these intermediates in medicinal chemistry. For organizations focused on cost reduction in pharmaceutical intermediate manufacturing, this cobalt-catalyzed approach presents a compelling alternative to traditional precious metal methods, leveraging earth-abundant metals to drive down raw material costs while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral diboron compounds has been largely confined to 1,1-, 1,2-, 1,3-, and 1,4-positional isomers, with very few reports detailing the efficient preparation of 1,5-diboron alkanes with high enantioselectivity. Conventional strategies often rely on multi-step sequences involving protecting group manipulations or stoichiometric chiral auxiliaries, which inherently increase the environmental footprint and production costs. A major technical hurdle in accessing 1,5-diboron systems directly from unsaturated hydrocarbons lies in the control of chemoselectivity and regioselectivity. Vinylcyclopropanes, while attractive starting materials due to their ring strain energy, pose significant challenges because they possess both an unsaturated double bond and a strained cyclopropane ring. In traditional catalytic systems, competing reactions such as simple hydrogenation or non-selective ring opening often dominate, leading to complex mixtures of side products rather than the desired doubly hydroborated species. Additionally, the formation of thermodynamically stable trisubstituted alkene intermediates during the reaction sequence can stall the process, preventing the second hydroboration event required to install the distal boron moiety.
The Novel Approach
The methodology described in patent CN114957304A overcomes these intrinsic barriers through the deployment of a tailored chiral CoX2-TIP complex catalyst. This system uniquely facilitates a tandem isomerization and hydroboration sequence that effectively navigates the energetic landscape of the vinylcyclopropane substrate. Unlike previous methods that struggle with the thermodynamic stability of intermediates, this cobalt-catalyzed process promotes the necessary isomerization to generate the reactive disubstituted alkene species in situ, allowing for the subsequent installation of the second boron group. The result is a highly efficient transformation that proceeds under mild conditions, typically at 0°C in diethyl ether, with excellent atom economy. 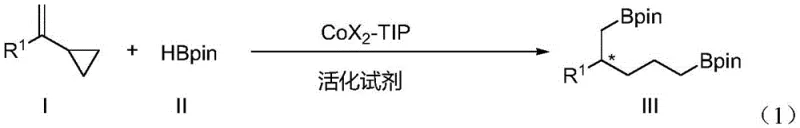 This novel approach not only simplifies the synthetic workflow by reducing the number of unit operations but also ensures high levels of stereochemical fidelity, with enantiomeric excess values reaching up to 96% ee. For supply chain managers, this translates to a more reliable source of complex chiral building blocks, reducing the dependency on lengthy, low-yielding synthetic routes that are prone to bottlenecks.
This novel approach not only simplifies the synthetic workflow by reducing the number of unit operations but also ensures high levels of stereochemical fidelity, with enantiomeric excess values reaching up to 96% ee. For supply chain managers, this translates to a more reliable source of complex chiral building blocks, reducing the dependency on lengthy, low-yielding synthetic routes that are prone to bottlenecks.
Mechanistic Insights into Cobalt-Catalyzed Asymmetric Double Hydroboration
The core of this technological breakthrough lies in the specific design of the chiral ligand and the resulting cobalt coordination environment. The catalyst employs a thiazoline-imine-pyridine (TIP) ligand framework, which creates a chiral pocket around the cobalt center essential for inducing asymmetry. Mechanistically, the reaction is believed to initiate with the formation of a cobalt-hydride species upon activation with reagents like sodium triethylborohydride. This active hydride species undergoes migratory insertion into the terminal alkene of the vinylcyclopropane, followed by a regioselective ring-opening of the cyclopropane moiety. This ring-opening step is critical as it extends the carbon chain to the requisite five-carbon backbone while establishing the initial stereocenter. The chiral environment of the TIP ligand dictates the facial selectivity of these insertion events, ensuring that the resulting organocobalt intermediate possesses the desired configuration. Subsequent reaction with pinacolborane (HBpin) releases the mono-borated intermediate, which then undergoes a second cycle of isomerization and hydroboration to install the second boron group at the 5-position.
Controlling the impurity profile is paramount for any reliable pharmaceutical intermediate supplier, and this catalytic system demonstrates remarkable specificity. The patent data indicates that side reactions such as simple hydrogenation of the alkene or incomplete ring opening are minimized through precise tuning of the catalyst loading and reaction temperature. The use of specific activators like sodium triethylborohydride is crucial for generating the active cobalt species without promoting non-productive decomposition pathways. Furthermore, the tolerance of the catalyst system towards various functional groups on the aryl ring of the substrate—such as halogens, ethers, and esters—suggests a robust mechanism that does not easily succumb to poisoning by heteroatoms. This mechanistic resilience ensures that the final product, the chiral 1,5-diboron compound, is obtained with high chemical purity, reducing the burden on downstream purification processes and enhancing the overall viability of the route for commercial scale-up.
How to Synthesize Chiral 1,5-Diboron Compounds Efficiently
The practical implementation of this synthesis involves a straightforward protocol that balances reaction efficiency with operational simplicity. The process begins with the preparation of the chiral cobalt catalyst, which can be synthesized from commercially available ligands and cobalt salts. In a typical run, the catalyst is combined with the vinylcyclopropane substrate and pinacolborane in anhydrous diethyl ether under an inert atmosphere. The reaction is cooled to 0°C, and the activating reagent is added slowly to control the exotherm and ensure uniform catalyst activation. Following a reaction period of approximately 36 hours, the mixture is worked up by filtration through silica gel to remove metal residues, followed by solvent removal and chromatographic purification. For those interested in further derivatization, the patent describes a convenient one-pot procedure where the crude diboron intermediate is directly subjected to oxidative workup with hydrogen peroxide and sodium hydroxide to yield the corresponding chiral 1,5-diol without isolation of the boron species.
- Prepare the reaction system under inert gas by combining the chiral CoI2-TIP catalyst, alkenyl cyclopropane substrate, and pinacolborane in anhydrous diethyl ether.
- Cool the mixture to 0°C and add sodium triethylborohydride as the activating reagent, stirring for 36 hours to complete the asymmetric double hydroboration.
- Perform post-treatment by filtering through silica gel and purifying via column chromatography, or proceed directly to oxidation for one-pot diol synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this cobalt-catalyzed technology offers distinct strategic advantages over traditional precious metal-catalyzed routes. The most immediate benefit is the substantial cost savings associated with replacing expensive noble metals like palladium, rhodium, or iridium with earth-abundant cobalt. This shift not only lowers the direct material cost of the catalyst but also mitigates the supply risk associated with the volatile markets of precious metals. Moreover, the mild reaction conditions (0°C to room temperature) reduce energy consumption compared to processes requiring cryogenic temperatures or high-pressure hydrogenation, contributing to a lower overall carbon footprint and operational expenditure. The high atom economy of the double hydroboration reaction means that a greater proportion of the starting materials end up in the final product, minimizing waste generation and disposal costs. These factors collectively enhance the economic feasibility of producing complex chiral intermediates at a commercial scale.
- Cost Reduction in Manufacturing: The utilization of a cobalt-based catalyst system significantly reduces the reliance on costly precious metals, which are subject to price fluctuations and supply constraints. By employing an earth-abundant metal, manufacturers can achieve a more stable and predictable cost structure for their raw materials. Additionally, the high yields reported (ranging from 55% to 98%) mean that less starting material is wasted, further driving down the cost per kilogram of the final intermediate. The ability to perform telescoped one-pot reactions, such as the direct conversion to 1,5-diols, eliminates the need for intermediate isolation and purification steps, thereby reducing solvent usage, labor hours, and equipment occupancy time. This streamlined process flow translates into tangible financial efficiencies that improve the gross margin of the final API or fine chemical product.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for exotic catalysts or complex chiral ligands can create vulnerabilities in the supply chain. The ligands and cobalt salts used in this process are relatively accessible, and the synthesis of the catalyst itself is described in the patent as straightforward, allowing for potential in-house preparation or sourcing from multiple vendors. The robustness of the reaction conditions, which tolerate a wide range of functional groups and do not require extreme temperatures or pressures, ensures consistent batch-to-batch reproducibility. This reliability is critical for maintaining continuous production schedules and meeting the strict delivery timelines demanded by pharmaceutical clients. Furthermore, the versatility of the 1,5-diboron intermediate allows it to serve as a common precursor for multiple downstream targets, enabling a more flexible and responsive inventory management strategy.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often reveals hidden challenges related to heat transfer, mixing, and safety. The mild nature of this hydroboration reaction, operating near ambient temperatures in common solvents like diethyl ether, presents fewer engineering hurdles for scale-up compared to highly exothermic or high-pressure reactions. The use of cobalt, while requiring appropriate handling, avoids the stringent regulatory burdens and toxicity concerns associated with some heavy metals, simplifying waste treatment and environmental compliance. The high selectivity of the reaction minimizes the formation of difficult-to-separate byproducts, reducing the load on wastewater treatment facilities and lowering the environmental impact of the manufacturing process. These attributes make the technology well-suited for commercial scale-up of complex pharmaceutical intermediates, ensuring that production can be ramped up efficiently to meet market demand without compromising on safety or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing pipelines. The answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What enantioselectivity can be achieved with this cobalt-catalyzed method?
A: The patented process demonstrates excellent stereocontrol, achieving enantioselectivity values ranging from 74% to 96% ee across various substrates, ensuring high optical purity for downstream API synthesis.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the reaction utilizes earth-abundant cobalt catalysts and mild conditions (0°C to room temperature), which significantly lowers operational costs and safety risks compared to precious metal catalysis, facilitating scalable manufacturing.
Q: What are the primary applications of the resulting chiral 1,5-diboron compounds?
A: These versatile intermediates serve as precursors for chiral 1,5-diols, diamines, and diarylalkanes. Notably, they enable the efficient total synthesis of the dopamine agonist (-)-Preclamol and other complex bioactive molecules.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 1,5-Diboron Compounds Supplier
The technological potential of chiral 1,5-diboron compounds as versatile building blocks for drug discovery and material science is immense, yet realizing this potential requires a partner with deep expertise in process development and manufacturing. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle sensitive organometallic reactions under strict inert atmospheres, ensuring that the stringent purity specifications required for pharmaceutical intermediates are consistently met. We utilize rigorous QC labs to verify enantiomeric excess and chemical purity at every stage of production, providing our clients with the confidence that their supply chain is secure and compliant with global regulatory standards.
We invite you to explore how our capabilities can accelerate your project timelines and optimize your manufacturing costs. Our technical team is ready to collaborate with you to evaluate the feasibility of this cobalt-catalyzed route for your specific target molecules. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how partnering with us can deliver high-quality chiral intermediates efficiently and reliably.
