Advanced Manufacturing of Triazine Ring Intermediates via Mixed-Base Catalysis for Global Pharma Supply Chains
The pharmaceutical industry continuously demands higher efficiency and purity in the synthesis of critical heterocyclic intermediates, particularly those serving as the backbone for broad-spectrum antibiotics. Patent CN112759558B introduces a groundbreaking methodology for the preparation of the Triazine ring, a pivotal structural motif essential for the synthesis of Ceftriaxone Sodium and various other high-value agrochemical and polymer applications. This innovative process addresses long-standing challenges in heterocyclic chemistry by replacing harsh, single-component catalytic systems with a sophisticated mixed-base approach that operates under mild thermal conditions. By leveraging a specific ratio of methanol and ethanol as a co-solvent system, the invention effectively manages reaction viscosity and heat transfer, ensuring uniform mixing that was previously unattainable with traditional sodium methoxide-only protocols. 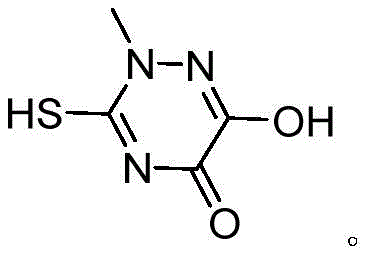 The structural integrity of the final product, as depicted in the molecular diagram, is preserved through precise pH control during the acidification and crystallization stages, resulting in a material that exhibits exceptional stability and reactivity for downstream coupling reactions. This technological leap not only enhances the chemical quality of the intermediate but also aligns with modern green chemistry principles by eliminating the need for difficult-to-recycle polar aprotic solvents.
The structural integrity of the final product, as depicted in the molecular diagram, is preserved through precise pH control during the acidification and crystallization stages, resulting in a material that exhibits exceptional stability and reactivity for downstream coupling reactions. This technological leap not only enhances the chemical quality of the intermediate but also aligns with modern green chemistry principles by eliminating the need for difficult-to-recycle polar aprotic solvents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Triazine rings has been plagued by significant operational inefficiencies and chemical limitations that hindered large-scale production capabilities. Traditional methods often relied heavily on sodium methoxide as a sole catalyst in conjunction with单一 solvent systems, which frequently resulted in highly viscous reaction mixtures that impeded effective mass transfer and heat dissipation. In these conventional setups, the strong alkalinity of sodium methoxide often led to localized zones of excessive base concentration, triggering a cascade of unwanted side reactions that degraded the yield and complicated the purification process. Furthermore, alternative synthetic routes utilizing reactive acyl chlorides, such as methyl malonyl chloride, introduced severe safety hazards and required complex multi-step procedures involving ion exchange resins for purification. These legacy processes were not only costly due to the expensive nature of the starting materials but also generated substantial waste streams that posed environmental compliance challenges for manufacturers. The reliance on solvents like DMF or DMSO in some prior art further exacerbated the problem, as these high-boiling solvents are notoriously difficult to remove and recycle, leading to increased production costs and potential contamination of the final API intermediate.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by introducing a dual-solvent system and a ternary mixed-base catalyst that fundamentally alters the reaction kinetics and thermodynamics. 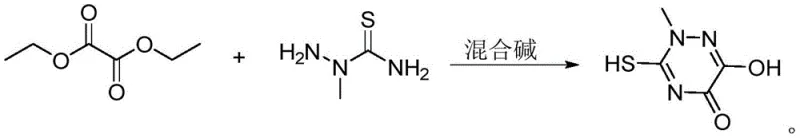 As illustrated in the reaction scheme, the condensation of 2-methyl thiosemicarbazide with diethyl oxalate proceeds smoothly in a methanol-ethanol mixture, which optimizes the polarity of the medium to keep reactants in solution while preventing the formation of viscous sludge. The strategic addition of DMAP and TMEDA alongside sodium methoxide creates a buffered basic environment that moderates the nucleophilic attack, thereby suppressing side reactions and enhancing the selectivity towards the desired triazine core. This approach allows the cyclization to occur at moderate temperatures between 45°C and 55°C, significantly reducing energy consumption compared to high-temperature reflux methods. Moreover, the use of simple alcohols as solvents facilitates straightforward recovery and recycling through distillation, drastically reducing the environmental footprint and operational expenditure associated with solvent management. The result is a streamlined, one-pot synthesis that delivers high yields without the need for complex workup procedures or hazardous reagents.
As illustrated in the reaction scheme, the condensation of 2-methyl thiosemicarbazide with diethyl oxalate proceeds smoothly in a methanol-ethanol mixture, which optimizes the polarity of the medium to keep reactants in solution while preventing the formation of viscous sludge. The strategic addition of DMAP and TMEDA alongside sodium methoxide creates a buffered basic environment that moderates the nucleophilic attack, thereby suppressing side reactions and enhancing the selectivity towards the desired triazine core. This approach allows the cyclization to occur at moderate temperatures between 45°C and 55°C, significantly reducing energy consumption compared to high-temperature reflux methods. Moreover, the use of simple alcohols as solvents facilitates straightforward recovery and recycling through distillation, drastically reducing the environmental footprint and operational expenditure associated with solvent management. The result is a streamlined, one-pot synthesis that delivers high yields without the need for complex workup procedures or hazardous reagents.
Mechanistic Insights into Mixed-Base Catalytic Cyclization
The success of this synthesis lies in the synergistic interaction between the three components of the mixed base catalyst: sodium methoxide, 4-Dimethylaminopyridine (DMAP), and Tetramethylethylenediamine (TMEDA). Sodium methoxide serves as the primary base to deprotonate the thiosemicarbazide, generating the nucleophilic species required for the initial attack on the diethyl oxalate ester. However, in isolation, sodium methoxide is too aggressive, leading to rapid, uncontrolled reactions; the addition of DMAP acts as a nucleophilic catalyst that accelerates the acylation step while TMEDA functions as a ligand that likely coordinates with sodium ions, effectively solvating the base and distributing its activity more evenly throughout the reaction medium. This coordination prevents the formation of localized high-pH pockets that typically cause hydrolysis of the ester groups or decomposition of the sensitive heterocyclic ring. The mechanism ensures that the cyclization proceeds through a controlled pathway where the intramolecular condensation is favored over intermolecular polymerization or degradation. By maintaining the reaction temperature strictly within the 45-55°C window, the kinetic energy is sufficient to overcome the activation barrier for ring closure without providing enough energy to drive competing decomposition pathways, thus securing the high selectivity observed in the experimental data.
Impurity control is another critical aspect where this mechanistic design excels, particularly regarding the suppression of hydrolysis byproducts and unreacted starting materials. The precise stoichiometric balance, with a slight excess of diethyl oxalate (molar ratio 1:1.05 to 1:1.1), ensures that the limiting reagent, 2-methyl thiosemicarbazide, is fully consumed, minimizing the presence of amine impurities in the crude product. The subsequent acidification step with hydrochloric acid is carefully managed to reach a pH of 0.5 to 1, which protonates the triazine nitrogen atoms just enough to induce precipitation without causing acid-catalyzed degradation of the thione or hydroxyl functionalities. The recrystallization from distilled water at controlled cooling rates (from 65-70°C down to 10-15°C) further purifies the lattice, excluding structurally similar byproducts that might have co-precipitated during the initial filtration. This rigorous control over the crystallization thermodynamics ensures that the final dry product meets the stringent purity specification of 99.5%, making it suitable for direct use in the synthesis of sensitive beta-lactam antibiotics without requiring additional chromatographic purification steps.
How to Synthesize Triazine Ring Efficiently
The operational protocol for this synthesis is designed for robustness and scalability, allowing chemical engineers to transition from laboratory benchtop experiments to multi-ton industrial reactors with minimal re-optimization. The process begins with the careful charging of raw materials into a standard glass-lined or stainless steel reactor equipped with efficient agitation and temperature control systems. The key to success lies in the rate of catalyst addition; dropping the mixed base solution over a period of 0.5 to 1.5 hours while maintaining the internal temperature between 5°C and 15°C is critical to prevent exothermic runaway and ensure the formation of the correct intermediate species. Following the addition, the reaction mixture is gently heated to promote cyclization, after which the product is isolated via simple filtration and washed to remove inorganic salts. For a comprehensive understanding of the specific parameters, including exact mass ratios and drying conditions, please refer to the standardized synthesis guide provided below.
- Charge 2-methyl thiosemicarbazide, diethyl oxalate, and a mixed methanol-ethanol solvent into a reactor, then cool the mixture to 5-15°C.
- Dropwise add a mixed base catalyst comprising sodium methoxide, DMAP, and TMEDA over 0.5-1.5 hours, then heat to 45-55°C for cyclization.
- Acidify the reaction mixture with hydrochloric acid to pH 0.5-1, cool to 0-5°C for filtration, and recrystallize the crude product from distilled water.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented process offers substantial advantages by decoupling production costs from volatile specialty chemical markets and simplifying the supply chain logistics. The shift away from expensive and hazardous acyl chloride intermediates to stable, commodity-grade diethyl oxalate significantly lowers the raw material cost basis, providing a buffer against price fluctuations in the fine chemical sector. Furthermore, the elimination of difficult-to-remove solvents like DMF and DMSO reduces the burden on waste treatment facilities and lowers the cost of solvent recovery, directly impacting the bottom line through reduced utility and disposal expenses. The high selectivity of the reaction means that less raw material is wasted on byproduct formation, effectively increasing the atom economy and reducing the overall material intensity required per kilogram of finished product. These factors combine to create a manufacturing route that is not only economically superior but also more resilient to regulatory changes regarding environmental emissions and worker safety.
- Cost Reduction in Manufacturing: The implementation of this mixed-base catalytic system eliminates the need for costly ion exchange resin purification steps and expensive acyl chloride reagents, leading to a drastic simplification of the production workflow. By utilizing a mixed alcohol solvent system that is easily recoverable through standard distillation, the process avoids the high energy costs associated with stripping high-boiling polar aprotic solvents, resulting in significant operational expenditure savings. The high yield achieved through improved selectivity means that less feedstock is required to produce the same amount of active intermediate, further driving down the unit cost of production. Additionally, the mild reaction conditions reduce wear and tear on reactor equipment and lower energy consumption for heating and cooling, contributing to a leaner and more cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the primary raw materials, 2-methyl thiosemicarbazide and diethyl oxalate, are widely available commodity chemicals with established global supply chains, unlike specialized acyl chlorides which may have limited suppliers. The robustness of the process against minor variations in reaction conditions ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results that require reprocessing. The simplified workup procedure, which relies on aqueous crystallization rather than complex chromatographic separations, shortens the overall cycle time from raw material intake to finished goods, allowing for faster turnaround on customer orders. This reliability makes the supplier a more dependable partner for long-term contracts, ensuring continuity of supply for critical antibiotic production lines.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of low-viscosity solvents that facilitate efficient heat transfer and mixing even in large-volume reactors, mitigating the risks associated with scaling up exothermic reactions. The absence of halogenated solvents and heavy metal catalysts simplifies the environmental compliance landscape, making it easier to obtain necessary operating permits and adhere to strict international environmental standards. Waste streams generated are primarily aqueous and alcoholic, which are easier to treat biologically or recover compared to the complex organic waste generated by traditional methods. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening regulations but also enhances the corporate sustainability profile for downstream pharmaceutical customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced Triazine ring synthesis technology. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on yield expectations, purity profiles, and process safety. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing supply chains or for R&D departments looking to optimize their current synthetic routes for cephalosporin antibiotics.
Q: How does the mixed solvent system improve the Triazine ring synthesis yield?
A: The use of a mixed methanol and ethanol solvent system significantly reduces the viscosity of the reaction medium compared to single-solvent systems. This reduction in viscosity prevents local concentration gradients and avoids 'local over-alkali' conditions during catalyst addition, which minimizes side reactions and boosts the overall yield to over 92%.
Q: What are the purity specifications achievable with this novel catalytic process?
A: By utilizing a synergistic mixed base catalyst system (Sodium Methoxide/DMAP/TMEDA) and optimizing the crystallization temperature, the process consistently achieves a product purity of 99.5% or higher, meeting the stringent requirements for cephalosporin antibiotic intermediates.
Q: Why is this method superior to traditional acyl chloride routes for Triazine ring production?
A: Unlike traditional routes that rely on hazardous and expensive methyl malonyl chloride requiring complex ion exchange purification, this method uses stable diethyl oxalate and eliminates the need for toxic polar aprotic solvents like DMF, resulting in a safer, more cost-effective, and environmentally compliant manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazine Ring Supplier
At NINGBO INNO PHARMCHEM, we recognize that the consistent supply of high-quality heterocyclic intermediates is the cornerstone of reliable antibiotic manufacturing. Our technical team has extensively analyzed the potential of this mixed-base catalytic route and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to the global market. We are committed to maintaining stringent purity specifications through our rigorous QC labs, ensuring that every batch of Triazine ring we deliver meets the exacting standards required for GMP pharmaceutical synthesis. Our facility is equipped to handle the specific solvent recovery and crystallization needs of this process, guaranteeing a product that is free from residual solvents and inorganic impurities.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. By partnering with us, you gain access to a supply chain that prioritizes both economic efficiency and technical excellence. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term production goals.
