Scalable Synthesis of High-Purity Beta-Benzo Amino Acid Intermediates via Chiral Resolution
Scalable Synthesis of High-Purity Beta-Benzo Amino Acid Intermediates via Chiral Resolution
The pharmaceutical industry's relentless pursuit of novel therapeutic agents has placed beta-amino acids at the forefront of medicinal chemistry research, particularly due to their unique ability to form stable secondary structures like alpha-helices and beta-turns that differ significantly from their alpha-amino acid counterparts. Patent CN111333552B discloses a groundbreaking synthesis method for beta-benzo amino acid compounds that addresses the critical bottlenecks of cost, complexity, and scalability inherent in traditional chiral resolution techniques. This technology enables the production of high-purity enantiomers without the need for expensive chiral chromatography, utilizing a clever chemical resolution strategy with aspartic acid that simplifies the downstream processing workflow. For R&D directors and procurement managers alike, this represents a paradigm shift towards more economical and robust manufacturing processes for key pharmaceutical intermediates used in antibiotics, antiviral drugs, and metabolic disorder treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of optically pure beta-amino acids has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional chemical resolution methods often involve multi-step derivatization processes to create diastereomers that can be separated, followed by tedious de-protection steps that lower overall yield and increase waste generation. Furthermore, reliance on chiral ligand exchange chromatography or preparative HPLC introduces substantial capital expenditure (CAPEX) for specialized columns and equipment, while also limiting batch sizes due to column loading capacity constraints. These conventional approaches frequently suffer from low yields, often requiring three to four rounds of recrystallization to achieve acceptable optical purity, which drastically inflates the cost of goods sold (COGS) and extends production lead times beyond acceptable limits for fast-paced drug development pipelines.
The Novel Approach
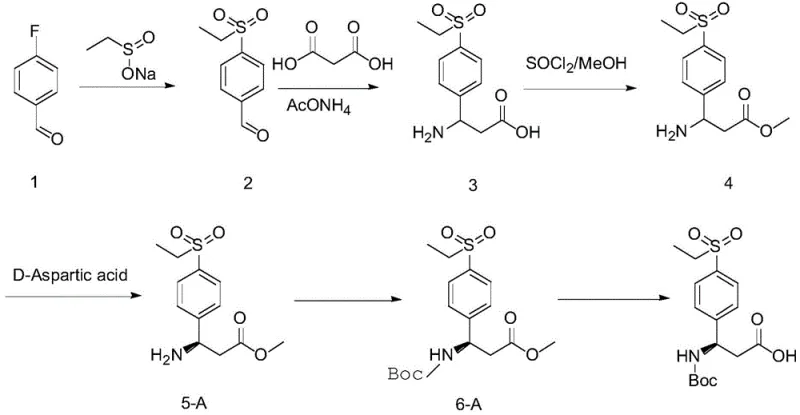
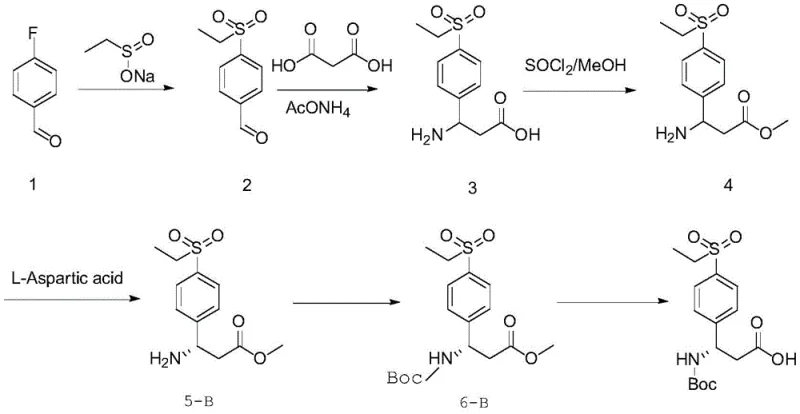
In stark contrast, the novel approach detailed in the patent leverages the specific stereochemical recognition properties of naturally occurring aspartic acid to resolve racemic beta-benzo amino acid precursors directly. By reacting the racemate (Formula I) with either L-aspartic acid or D-aspartic acid in a optimized methanol-water solvent system, the process facilitates the selective crystallization of the desired diastereomeric salt with high efficiency. This method eliminates the need for transition metal catalysts or expensive enzymatic resolutions, relying instead on simple thermal control (dissolving at 55-60°C and cooling) and filtration to achieve optical purity levels exceeding 98% ee. The simplicity of this operation allows for seamless integration into existing reactor infrastructure, offering a reliable pharmaceutical intermediate supplier pathway that drastically reduces both operational complexity and environmental footprint compared to legacy technologies.
Mechanistic Insights into Aspartic Acid-Mediated Chiral Resolution
The core mechanistic advantage of this synthesis lies in the formation of diastereomeric salts between the racemic beta-amino acid ester and the chiral resolving agent, aspartic acid, which exhibit distinct solubility profiles in the chosen solvent matrix. When the racemate is dissolved in a mixture of methanol and water at elevated temperatures (55-60°C) and then cooled, the less soluble diastereomeric salt precipitates out of the solution while the unwanted enantiomer remains in the mother liquor. The specific molar ratio of the racemate to aspartic acid, optimized between 1:0.5 and 1:1, is critical for maximizing the yield of the precipitate while maintaining high chiral purity, effectively acting as a thermodynamic filter that enriches the solid phase with the target enantiomer. This precise control over crystallization kinetics ensures that the product obtained after simple filtration and methanol washing possesses an ee value of greater than 98%, bypassing the need for further purification steps that typically erode yield in conventional resolution processes.
Furthermore, the subsequent protection and transesterification steps are designed to preserve this high optical integrity while converting the intermediate into a stable, protected form suitable for long-term storage or further coupling reactions. The introduction of a tert-butoxycarbonyl (Boc) protecting group on the amine functionality prevents racemization during the final hydrolysis or ester exchange reactions, ensuring that the stereochemical information established during the resolution step is locked in. This robustness against racemization is vital for maintaining the biological activity of the final drug substance, as even trace amounts of the wrong enantiomer can lead to toxicity or reduced efficacy in the final pharmaceutical product, making this mechanistic stability a key selling point for high-purity OLED material or API manufacturing contexts.
How to Synthesize Beta-Benzo Amino Acid Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible pathway for generating both (R) and (S) enantiomers of 3-((tert-butoxycarbonyl)amino)-3-(4-(ethylsulfo)phenyl)propanoic acid, a critical fragment for various bioactive molecules. The process begins with the nucleophilic substitution of para-fluorobenzaldehyde with sodium ethyl sulfinate to introduce the sulfonyl group, followed by a one-pot condensation with malonic acid and ammonium acetate to build the beta-amino acid backbone. The detailed standardized synthesis steps below outline the precise conditions for resolution, protection, and de-esterification required to achieve commercial-grade quality.
- Synthesize the racemic beta-benzo amino acid ester (Formula I) via substitution of para-halogen benzaldehyde followed by condensation with malonic acid and ammonium acetate.
- Perform chiral resolution by reacting the racemate with L-aspartic acid or D-aspartic acid in a methanol/water solvent system at 55-60°C, followed by cooling crystallization.
- Protect the amine group with a Boc group and perform transesterification/hydrolysis to obtain the final optically pure beta-benzo amino acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic advantages regarding cost stability, supply continuity, and regulatory compliance. By shifting away from enzyme-dependent or chromatography-heavy processes, manufacturers can mitigate the risks associated with the supply volatility of biocatalysts and the high maintenance costs of separation equipment. The use of commodity chemicals like aspartic acid, malonic acid, and para-halogen benzaldehydes ensures a resilient raw material base that is less susceptible to geopolitical disruptions or single-source bottlenecks, thereby enhancing the overall reliability of the supply chain for critical drug intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive chiral chromatography columns and transition metal catalysts results in substantial cost savings in pharmaceutical intermediates manufacturing. The process relies on inexpensive resolving agents and standard solvent systems, removing the need for high-CAPEX equipment and reducing the consumption of high-purity reagents. Additionally, the high yield of the resolution step (approximately 43% theoretical yield with >98% ee in a single crystallization) minimizes material loss compared to multi-recrystallization methods, leading to a significantly lower cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as para-fluorobenzaldehyde and aspartic acid ensures a stable and continuous supply of raw materials, reducing lead time for high-purity pharmaceutical intermediates. The robustness of the chemical steps, which do not require stringent anhydrous conditions or inert atmospheres for the resolution phase, allows for flexible production scheduling across multiple manufacturing sites. This flexibility is crucial for meeting sudden spikes in demand for antiviral or antibiotic therapies without the long lead times associated with sourcing specialized chiral catalysts.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory bench scale to pilot plant operations involving tens of kilograms of material. The simplified work-up procedures, primarily involving filtration and washing, generate less hazardous waste compared to extraction-heavy enzymatic processes, facilitating easier compliance with increasingly strict environmental regulations. This ease of scale-up supports the commercial scale-up of complex polymer additives or drug substances from 100 kgs to 100 MT annual production volumes with minimal process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-benzo amino acid synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on yield expectations, purity standards, and process adaptability for potential licensees or contract manufacturing partners.
Q: How does this method improve upon traditional chiral chromatography?
A: This method replaces expensive chiral chromatographic columns and complex derivatization steps with a direct chemical resolution using inexpensive L- or D-aspartic acid. It achieves high optical purity (ee > 98%) through simple crystallization and washing, significantly reducing operational complexity and capital expenditure.
Q: What are the key advantages for large-scale manufacturing?
A: The process utilizes readily available starting materials like para-halogen benzaldehyde and avoids sensitive enzymatic catalysts. The resolution step relies on standard filtration and washing protocols rather than specialized chromatography equipment, making it highly adaptable for multi-ton commercial production with consistent quality.
Q: Can both enantiomers be produced using this route?
A: Yes, the method is versatile. By selecting either L-aspartic acid or D-aspartic acid as the resolving agent in the second step, manufacturers can selectively produce either the (S)-configured or (R)-configured beta-benzo amino acid enantiomer to meet specific drug synthesis requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Benzo Amino Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route in delivering high-value chiral intermediates to the global pharmaceutical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee ee values exceeding 98%, providing our clients with the confidence needed for regulatory filings and clinical trial material supply.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and scalable technology for their pipeline projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time-to-market with our reliable beta-benzo amino acid supplier capabilities and commitment to technical excellence.
