Scalable Metal-Free Synthesis of N-Aryl Lactams for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of N-Aryl Lactams for Advanced Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks more efficient and environmentally benign pathways for constructing complex heterocyclic scaffolds, particularly those found in bioactive alkaloids. Patent CN113292473A introduces a groundbreaking methodology for the synthesis of N-aryl substituted lactam compounds, a structural motif prevalent in therapeutic agents targeting neurological disorders such as Alzheimer's and Parkinson's disease. This innovative approach leverages a non-metallic oxyammonium salt in conjunction with tert-butyl hydroperoxide to facilitate a multi-step cascade reaction, effectively transforming N-aryl substituted saturated cyclic amines into valuable lactam structures. By eliminating the reliance on precious transition metal catalysts, this technology addresses critical pain points regarding metal residue contamination and process complexity, positioning it as a superior alternative for the manufacture of high-purity pharmaceutical intermediates.
From a strategic sourcing perspective, the adoption of this metal-free protocol offers substantial benefits for supply chain stability and cost management. The reaction operates under mild conditions using air as the oxidant source alongside common organic peroxides, drastically reducing the infrastructure requirements for specialized inert atmosphere handling. Furthermore, the broad substrate tolerance described in the patent ensures that diverse analogues can be accessed from readily available starting materials, enhancing the flexibility of drug discovery campaigns. For procurement managers and R&D directors alike, this represents a shift towards more sustainable and economically viable manufacturing processes that align with modern green chemistry principles while maintaining rigorous quality standards essential for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of N-aryl substituted lactam rings has relied heavily on methodologies that present significant operational and economic challenges for large-scale production. Traditional routes often involve the dearomatization of substituted aza-aromatic compounds or condensation reactions of functionalized chain amines, which typically require multiple synthetic steps and harsh reaction conditions. More critically, many established protocols depend on the use of transition metal catalysts to promote oxidation or epoxidation reactions. While effective on a laboratory scale, these metal-catalyzed processes introduce severe complications during the purification phase, necessitating expensive and time-consuming heavy metal scavenging procedures to meet stringent regulatory limits for residual metals in active pharmaceutical ingredients. Additionally, the preparation of specific raw materials for these older methods can be difficult and costly, limiting the overall practicality and substrate universality of the synthesis.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a transition-metal-free system driven by oxyammonium salts and organic peroxides to achieve direct oxidative cyclization. This approach streamlines the synthetic sequence into a efficient cascade reaction that constructs the lactam unit directly from saturated cyclic amine precursors. The elimination of transition metals not only simplifies the downstream processing by removing the need for metal removal resins or complex extractions but also significantly reduces the environmental footprint of the manufacturing process. The use of stable oxyammonium salts as promoters ensures high reproducibility and safety, while the compatibility with air atmosphere further lowers operational costs by removing the need for rigorous nitrogen or argon purging. This represents a paradigm shift towards simpler, greener, and more cost-effective manufacturing strategies for complex nitrogen-containing heterocycles.
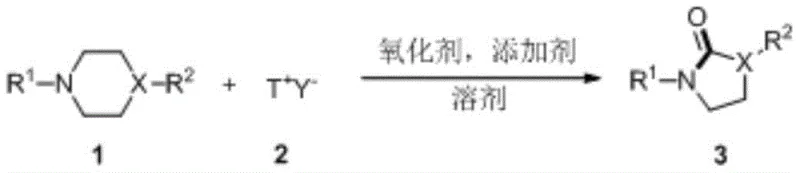
The core transformation involves the reaction of an N-aryl substituted saturated cyclic amine with an oxyammonium salt species in the presence of an oxidant and an acid additive. As illustrated in the reaction scheme, the process tolerates a wide variety of substituents on the aromatic ring, including halogens, alkyl groups, esters, and nitro groups, demonstrating exceptional versatility for medicinal chemistry applications. The ability to utilize simple solvents like toluene or acetonitrile and operate at moderate temperatures between 80°C and 120°C makes this protocol highly adaptable to existing reactor setups without requiring specialized high-pressure or cryogenic equipment. This robustness is a key factor for reliable pharmaceutical intermediate suppliers aiming to deliver consistent quality at scale.
Mechanistic Insights into Oxyammonium-Promoted Oxidative Cyclization
The mechanistic pathway of this transformation is driven by the unique redox properties of the oxyammonium salt, which acts as a potent single-electron oxidant to initiate the cascade sequence. The reaction likely proceeds through the generation of an aminium radical cation intermediate upon interaction between the tertiary amine substrate and the oxyammonium species. This radical intermediate subsequently undergoes hydrogen atom transfer or further oxidation to form an iminium ion, which is then trapped by the internal nucleophile or undergoes rearrangement to close the lactam ring. The presence of the acid additive plays a crucial role in facilitating proton transfer steps and stabilizing charged intermediates, thereby accelerating the overall reaction rate and improving yield. Understanding this radical-mediated mechanism allows chemists to fine-tune reaction parameters such as oxidant stoichiometry and acid strength to optimize outcomes for specific substrates.
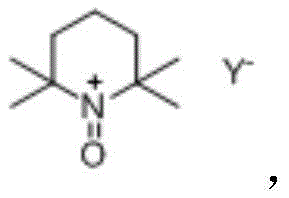
Impurity control in this metal-free system is inherently superior compared to transition-metal catalyzed routes due to the absence of metal-associated side reactions and degradation products. The primary byproducts are typically derived from the reduction of the oxidant or minor over-oxidation species, which are generally easier to separate from the target lactam than organometallic complexes. The use of well-defined oxyammonium salts, such as the TEMPO derivative shown above, ensures a consistent impurity profile that is easier to characterize and validate during the regulatory filing process. For R&D teams, this predictability reduces the risk of late-stage failures due to unexpected genotoxic impurities or difficult-to-remove metal traces, thereby accelerating the timeline from process development to commercial launch. The clean reaction profile supports the production of high-purity intermediates essential for sensitive neurological drug formulations.
How to Synthesize N-Aryl Substituted Lactam Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and temperature controls to maximize conversion while minimizing byproduct formation. The patent specifies a molar ratio of substrate to oxyammonium salt to oxidant ranging from 1:1-2:2-4, with an acid additive loading of 0.5 to 1 equivalent. Detailed standard operating procedures for mixing sequences, addition rates, and quenching protocols are critical for ensuring batch-to-batch consistency. The following guide outlines the generalized workflow derived from the patent examples, serving as a foundational reference for process engineers scaling this technology.
- Dissolve the N-aryl substituted saturated cyclic amine substrate and the oxyammonium salt catalyst in a suitable solvent such as toluene or acetonitrile.
- Add the oxidant (tert-butyl hydroperoxide) and an acid additive (such as trifluoroacetic acid) to the reaction mixture under an air atmosphere.
- Heat the reaction system to 80-120°C for approximately 4 hours, then quench with saturated sodium chloride and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this metal-free synthesis route offers compelling economic and logistical advantages that directly impact the bottom line. By removing the dependency on scarce and volatile transition metal catalysts like palladium or rhodium, manufacturers can insulate their supply chains from geopolitical disruptions and price fluctuations associated with precious metals. Furthermore, the simplified purification workflow eliminates the capital expenditure and operational costs linked to metal scavenging technologies, resulting in a leaner and more efficient production cycle. The use of commodity chemicals such as tert-butyl hydroperoxide and common organic acids ensures a stable and diversified supply base, reducing the risk of raw material shortages that could halt production lines.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated removal steps leads to significant cost savings in raw material consumption and waste disposal. Without the need for specialized metal scavengers or complex filtration systems, the overall cost of goods sold is drastically reduced, allowing for more competitive pricing in the global market. Additionally, the higher atom economy and simplified workup reduce solvent usage and energy consumption, contributing to a more sustainable and cost-efficient manufacturing model that aligns with corporate ESG goals.
- Enhanced Supply Chain Reliability: Sourcing oxyammonium salts and organic peroxides is far less risky than securing supply chains for specialized organometallic complexes, which often have limited suppliers and long lead times. The robustness of the reaction under air atmosphere means that production does not rely on continuous supplies of high-purity inert gases, further simplifying facility requirements. This reliability ensures consistent delivery schedules for downstream customers, strengthening partnerships and fostering trust in the supplier's ability to meet demanding production timelines without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic nature being manageable with standard cooling systems and its tolerance to oxygen, which removes the explosion risks associated with some anaerobic metal-catalyzed reactions. From an environmental standpoint, the absence of heavy metals simplifies wastewater treatment and solid waste disposal, ensuring compliance with increasingly strict environmental regulations. This ease of scale-up allows manufacturers to rapidly increase capacity from pilot plant to commercial tonnage without extensive re-engineering of the process, facilitating faster time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing manufacturing portfolios.
Q: What are the primary advantages of this metal-free lactam synthesis over traditional transition-metal catalyzed methods?
A: The primary advantage is the complete elimination of transition metal catalysts, which removes the need for expensive and complex heavy metal removal steps during purification. This significantly lowers production costs and simplifies the regulatory compliance process for pharmaceutical intermediates, ensuring higher purity profiles without metal residues.
Q: Is this synthesis method scalable for industrial production of N-aryl lactams?
A: Yes, the method is highly suitable for industrial scale-up. It utilizes commercially available reagents like TEMPO salts and TBHP, operates under air atmosphere without requiring inert gas protection, and employs simple workup procedures involving standard extraction and chromatography, making it robust for large-scale manufacturing.
Q: What is the substrate scope for this oxidative cyclization reaction?
A: The reaction demonstrates excellent substrate universality, tolerating a wide range of substituents on the N-aryl ring including halogens (F, Cl, Br, I), electron-withdrawing groups (nitro, cyano, ester), and electron-donating groups (alkyl, alkoxy). It is also applicable to heteroaromatic systems and naphthyl derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Aryl Substituted Lactam Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in advancing the production of high-value pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our capability to handle complex oxidative transformations safely and efficiently makes us an ideal partner for your next project.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific drug development needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this metal-free route can optimize your budget. Please contact us today to request specific COA data for our lactam intermediates and discuss route feasibility assessments that will accelerate your path to clinical success. Let us be your trusted partner in navigating the complexities of modern pharmaceutical manufacturing.
