Advanced Metal-Free Synthesis of Axial Chiral Diarylethenes for High-Value Pharmaceutical Intermediates
Advanced Metal-Free Synthesis of Axial Chiral Diarylethenes for High-Value Pharmaceutical Intermediates
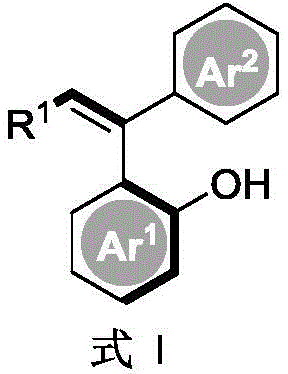
In the rapidly evolving landscape of asymmetric organic synthesis, the construction of axially chiral scaffolds remains a formidable challenge due to the low energy barriers associated with bond rotation, which often leads to racemization. Patent CN114315536A introduces a groundbreaking methodology for the synthesis of axial chiral diarylethene compounds, addressing critical limitations in current manufacturing protocols. This innovation leverages chiral phosphoric acid organocatalysis to facilitate the asymmetric nucleophilic addition of aryl nucleophiles to alkynes, achieving high yields and exceptional enantioselectivity without the need for expensive transition metals. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this technology represents a paradigm shift towards greener, more cost-effective, and highly selective chemical manufacturing processes that align with modern regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of axially chiral diarylethenes has relied heavily on transition metal-catalyzed C-H functionalization or kinetic resolution strategies, both of which present significant drawbacks for industrial scale-up. Transition metal catalysis, while effective, introduces the persistent risk of heavy metal residues such as palladium or rhodium, necessitating complex and costly purification steps to meet the stringent ppm-level limits imposed by global health authorities for pharmaceutical ingredients. Furthermore, kinetic resolution approaches are inherently inefficient, theoretically capping the maximum yield at 50% since half of the starting material is discarded as the unwanted enantiomer. These conventional methods also often suffer from narrow substrate scopes, requiring harsh reaction conditions that limit the diversity of functional groups that can be tolerated, thereby restricting the chemical space available for drug discovery and development.
The Novel Approach
The novel approach detailed in the patent utilizes a chiral phosphoric acid catalyst to drive the asymmetric hydroarylation of aryl alkynes, offering a direct and atom-economical route to the target structures. As illustrated in the general reaction scheme below, this method operates under mild inert gas atmospheres and avoids the use of toxic heavy metals entirely, simplifying the downstream processing and waste management protocols. By employing specifically designed chiral phosphoric acids with bulky substituents, the reaction achieves precise stereocontrol through a well-defined hydrogen-bonding network, resulting in products with high optical purity. This metal-free strategy not only enhances the safety profile of the manufacturing process but also drastically reduces the raw material costs associated with precious metal catalysts, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing.

Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Hydroarylation
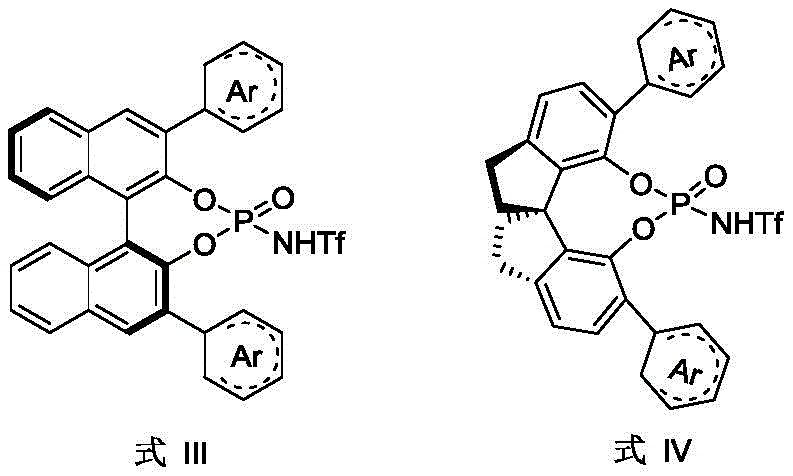
The core of this technological breakthrough lies in the unique ability of the chiral phosphoric acid catalyst to activate both the electrophilic alkyne and the nucleophilic arene simultaneously through a dual hydrogen-bonding mechanism. The catalyst, typically possessing a rigid binaphthyl or similar backbone with bulky aryl groups at the 3,3'-positions, creates a confined chiral pocket that dictates the trajectory of the incoming nucleophile. This steric environment effectively lowers the activation energy for the desired enantiomeric pathway while raising the barrier for the opposing pathway, ensuring high enantioselectivity even for substrates with low intrinsic rotational barriers. The absence of metal coordination complexes means that the reaction mechanism is driven purely by Brønsted acid catalysis, which is generally more tolerant of moisture and oxygen compared to sensitive organometallic species, thereby enhancing the robustness of the process in a production environment.
From an impurity control perspective, this organocatalytic system offers distinct advantages by eliminating the formation of metal-complexed byproducts that are notoriously difficult to separate. The reaction proceeds cleanly to form the C-C bond with the generation of the axial chirality, and the catalyst itself can often be recovered or easily separated due to its polarity differences from the non-polar diarylethene products. The patent data highlights that varying the electronic and steric properties of the catalyst, such as using those with anthracenyl or substituted phenyl groups as shown in the structural diagrams, allows for fine-tuning the selectivity for different substrate classes. This level of mechanistic control ensures that the final product meets the rigorous purity specifications required for high-purity pharmaceutical intermediates, minimizing the risk of genotoxic impurities often associated with metal catalysis.
How to Synthesize Axial Chiral Diarylethene Efficiently
The synthesis protocol described in the patent provides a straightforward and reproducible method for generating these valuable chiral building blocks. The process begins with the careful selection of the aryl acetylene and substituted aromatic hydrocarbon substrates, which are combined with a catalytic amount of the chiral phosphoric acid in a suitable organic solvent. The reaction is conducted under a nitrogen atmosphere to prevent oxidation, with temperature control playing a critical role in optimizing both yield and enantiomeric excess. Detailed standard operating procedures for scaling this reaction from gram to kilogram quantities involve precise monitoring of reaction progress and optimized workup techniques to ensure maximum recovery of the valuable chiral product. The detailed standardized synthesis steps are outlined in the guide below.
- Under an inert gas atmosphere such as nitrogen, sequentially add aryl acetylene, substituted aromatic hydrocarbon, and chiral phosphoric acid catalyst into a dry reactor.
- Add the appropriate reaction solvent (e.g., 1,2-dichloroethane or dichloromethane) and stir the mixture at a controlled temperature ranging from -30°C to 30°C for 12 to 48 hours.
- Upon completion, remove the solvent under reduced pressure and purify the crude product via column chromatography using a petroleum ether and ethyl acetate mixture to isolate the pure axial chiral compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthetic route offers transformative benefits that extend far beyond the laboratory bench. The elimination of precious metal catalysts directly translates to a significant reduction in raw material expenditure, as the cost of chiral phosphoric acids is generally lower and more stable than that of palladium or rhodium complexes. Moreover, the simplified purification process reduces the consumption of silica gel and solvents during chromatography, further driving down the operational expenses associated with manufacturing. This efficiency gain allows for a more competitive pricing structure for the final intermediates, providing a strategic advantage in cost-sensitive markets where margin compression is a constant challenge.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts eliminates the need for expensive metal scavengers and extensive analytical testing for residual metals, which are major cost drivers in traditional API synthesis. By switching to an organocatalytic system, manufacturers can streamline their quality control workflows and reduce the overall cost of goods sold, enabling substantial cost savings that can be passed down the supply chain or reinvested into R&D initiatives.
- Enhanced Supply Chain Reliability: The substrates required for this reaction, such as aryl alkynes and simple aromatic hydrocarbons, are commodity chemicals with robust and established global supply chains. Unlike specialized metal ligands that may face supply bottlenecks, these starting materials are readily available from multiple vendors, ensuring continuity of supply and reducing the risk of production delays. This reliability is crucial for maintaining just-in-time manufacturing schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The reaction conditions are mild, typically ranging from -30°C to 30°C, which reduces the energy consumption associated with heating or deep cooling compared to harsher traditional methods. Furthermore, the absence of heavy metals simplifies waste disposal and environmental compliance, as the effluent does not require specialized treatment for metal removal. This green chemistry profile facilitates easier regulatory approval and supports corporate sustainability goals, making the commercial scale-up of complex pharmaceutical intermediates more feasible and environmentally responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production pipelines.
Q: What are the primary advantages of this chiral phosphoric acid method over transition metal catalysis?
A: The primary advantage is the complete elimination of transition metal catalysts, which removes the risk of heavy metal contamination in the final API and significantly reduces the cost and complexity of purification processes required to meet strict regulatory limits.
Q: What kind of enantioselectivity can be achieved with this synthetic route?
A: According to the patent data, this method achieves excellent enantioselectivity, with specific examples demonstrating ee values as high as 95%, which is critical for producing high-purity single-enantiomer pharmaceutical intermediates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process operates under mild conditions (-30°C to 30°C) and uses readily available solvents and substrates. The simple workup procedure involving solvent removal and column chromatography indicates strong potential for scalability in industrial settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Diarylethene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications and utilize our rigorous QC labs to verify every batch, guaranteeing that our axial chiral diarylethene compounds meet the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced metal-free technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this innovative route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of high-purity intermediates for your critical drug development programs.
