Advanced One-Step Synthesis of Indole Naphthoquinone Derivatives for Commercial Scale-Up
Advanced One-Step Synthesis of Indole Naphthoquinone Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for complex heterocyclic scaffolds, particularly those exhibiting potent biological activities. Patent CN112979529A introduces a groundbreaking methodology for the preparation of aromatic amine indole naphthoquinone derivatives, a class of compounds with significant potential in anticancer and antimicrobial applications. This technology leverages a facile one-step condensation between indole naphthoquinone compounds and aromatic amines, bypassing the multi-step sequences often required in conventional syntheses. By utilizing a base-catalyzed Michael addition followed by an aerobic oxidation cascade, this process achieves high structural diversity while maintaining operational simplicity. For R&D directors and procurement specialists, this represents a pivotal shift towards more efficient pharmaceutical intermediate manufacturing, offering a pathway to reduce both material costs and environmental footprint without compromising on the purity or complexity of the final molecular architecture.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 2-substituted amino-1,4-naphthoquinone derivatives has relied heavily on classical nucleophilic substitution reactions involving halogenated naphthoquinone precursors. These traditional pathways often necessitate the prior synthesis of specific halogenated starting materials, which can be costly, unstable, and hazardous to handle on a large scale. Furthermore, alternative strategies involving direct 1,4-addition reactions frequently suffer from issues related to regioselectivity and the formation of unwanted side products due to the high reactivity of electron-deficient quinones. The requirement for harsh reaction conditions, expensive transition metal catalysts, or inert atmospheres in many existing protocols further exacerbates the operational complexity, leading to lower overall yields and increased production costs. These inefficiencies create significant bottlenecks in the commercial scale-up of complex pharmaceutical intermediates, limiting the ability of manufacturers to respond rapidly to market demands for novel therapeutic agents.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN112979529A offers a streamlined, atom-economical solution that directly couples indole naphthoquinones with aromatic amines. As illustrated in the general reaction scheme below, this novel approach eliminates the need for pre-functionalized halogen leaving groups, relying instead on the intrinsic reactivity of the quinone system under basic conditions.
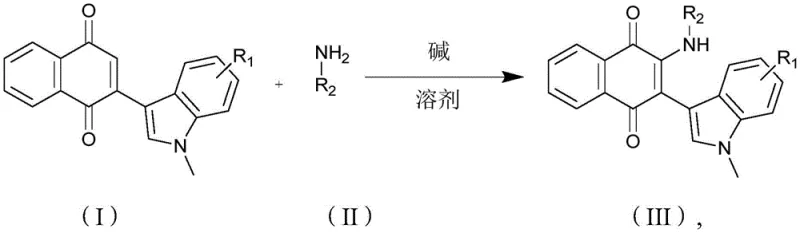
The process operates effectively at room temperature in common polar aprotic solvents like DMF, using inexpensive inorganic or organic bases such as potassium tert-butoxide. This drastic simplification of reaction parameters not only enhances safety profiles by removing the need for specialized high-pressure or high-temperature equipment but also facilitates easier purification workflows. By enabling the rapid assembly of diverse molecular libraries through simple variation of the amine component, this method provides a powerful tool for cost reduction in API manufacturing, allowing producers to access valuable chemical space with unprecedented efficiency and speed.
Mechanistic Insights into Base-Catalyzed Michael Addition and Oxidation
The success of this synthetic strategy lies in its elegant mechanistic pathway, which combines a nucleophilic attack with a spontaneous oxidative aromatization. In the presence of a base, the aromatic amine acts as a nucleophile, attacking the electron-deficient double bond of the indole naphthoquinone substrate via a Michael addition. This initial step generates a hydroquinone-like intermediate, which is inherently unstable under the reaction conditions. The subsequent restoration of the quinone system is driven by molecular oxygen from the air or by the naphthoquinone substrate itself acting as an oxidant, a feature that distinguishes this method from anaerobic processes.
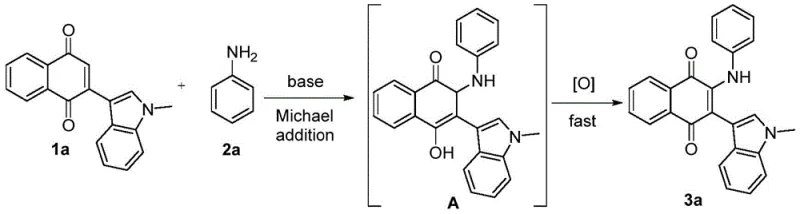
This oxidative step is crucial as it drives the equilibrium towards the final stable product, preventing the accumulation of reduced byproducts that often plague similar conjugate additions. The tolerance of the reaction to air atmosphere is a significant advantage for industrial scalability, as it removes the logistical burden and cost associated with maintaining strict inert gas environments. Furthermore, the mechanism allows for broad substrate scope, accommodating various substituents on both the indole and the aniline rings without significant loss in efficiency. Understanding this dual-step cascade is vital for process chemists aiming to optimize high-purity pharmaceutical intermediates, as controlling the base strength and oxygen exposure can fine-tune the reaction kinetics to minimize impurity profiles.
How to Synthesize Aromatic Amine Indole Naphthoquinone Derivatives Efficiently
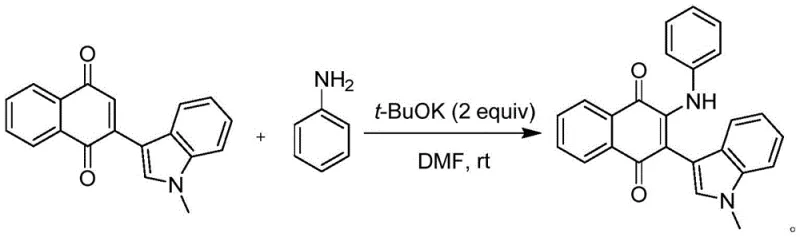
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to stoichiometry and mixing, although the overall procedure remains remarkably straightforward. The patent details specific embodiments where the molar ratio of the indole naphthoquinone to the aromatic amine is optimized, typically ranging from 1:1 to 1:5, with a preference for a slight excess of the amine to drive the reaction to completion. The choice of base is also critical, with potassium tert-butoxide (t-BuOK) demonstrating superior performance in terms of yield and reaction rate compared to weaker bases. Below is a representative example of the standardized protocol derived from the patent data, specifically illustrating the synthesis of the unsubstituted phenyl derivative which serves as a benchmark for the series.
- Combine the indole naphthoquinone substrate and the selected aromatic amine in a reaction vessel containing a polar aprotic solvent such as DMF.
- Add a strong base, preferably potassium tert-butoxide (t-BuOK), to the mixture to initiate the Michael addition reaction.
- Stir the reaction at room temperature under an air atmosphere for 1 to 24 hours to allow for spontaneous oxidation and product formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of expensive transition metal catalysts and the reliance on commodity chemicals like t-BuOK and DMF significantly lowers the raw material cost baseline. Moreover, the ability to run reactions at ambient temperature and pressure drastically reduces energy consumption, contributing to a lower carbon footprint and aligning with modern sustainability goals. The simplicity of the workup procedure, which typically involves standard aqueous quenching and extraction, minimizes solvent usage and waste generation, thereby reducing disposal costs and enhancing the overall green chemistry profile of the manufacturing process.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts and the use of readily available starting materials lead to substantial cost savings. By avoiding the need for specialized halogenated precursors, the supply chain becomes less vulnerable to price volatility associated with niche reagents. The one-pot nature of the reaction reduces labor hours and equipment occupancy time, allowing for higher throughput in existing facilities without the need for capital-intensive upgrades. This efficiency translates directly into a more competitive pricing structure for the final pharmaceutical intermediate, providing a clear margin advantage in contract manufacturing negotiations.
- Enhanced Supply Chain Reliability: The robustness of this method against atmospheric moisture and oxygen means that production is less susceptible to interruptions caused by utility failures or equipment leaks. Since the raw materials are common industrial chemicals with established global supply networks, the risk of sourcing bottlenecks is minimized. This reliability ensures consistent delivery schedules for downstream clients, a critical factor for pharmaceutical companies managing tight development timelines. The scalability of the process from gram to kilogram scales has been demonstrated, ensuring that supply can be ramped up quickly to meet commercial demand without extensive re-validation efforts.
- Scalability and Environmental Compliance: The reaction generates minimal hazardous waste compared to traditional halogenation-substitution routes, simplifying compliance with increasingly stringent environmental regulations. The absence of heavy metal residues in the crude product reduces the burden on downstream purification steps, such as chromatography or recrystallization, which are often resource-intensive. This streamlined purification process not only saves time but also reduces the volume of organic solvents required, supporting corporate sustainability initiatives. The inherent safety of operating at room temperature further lowers insurance and safety management costs, making this a highly attractive option for long-term commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and technical disclosures within CN112979529A, providing clarity on reaction scope, conditions, and potential applications. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the primary advantages of this synthesis method over traditional nucleophilic substitution?
A: Unlike traditional methods requiring pre-functionalized halogenated substrates and harsh conditions, this patent describes a direct Michael addition-oxidation cascade that operates at room temperature under air, significantly simplifying the process and reducing energy consumption.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is applicable to industrial production due to the use of easily obtainable raw materials, simple operation procedures, and the elimination of expensive transition metal catalysts.
Q: What is the role of the base in this reaction mechanism?
A: The base, typically t-BuOK, deprotonates the aromatic amine or activates the nucleophile to facilitate the initial Michael addition to the electron-deficient naphthoquinone system, forming a hydroquinone intermediate that is subsequently oxidized.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Amine Indole Naphthoquinone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in CN112979529A for the development of next-generation therapeutic agents. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of indole naphthoquinone derivatives meets the exacting standards required by global regulatory bodies. Our state-of-the-art facilities are equipped to handle the specific solvent and base requirements of this chemistry safely and effectively.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this streamlined protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will accelerate your drug development pipeline and optimize your supply chain resilience.
