Advanced Synthesis of Achiral Carbazole Nanogrids for Next-Generation Organic Semiconductors
Advanced Synthesis of Achiral Carbazole Nanogrids for Next-Generation Organic Semiconductors
The rapid evolution of the organic electronics sector demands materials with precise geometric control and exceptional thermal stability. Patent CN111825680A introduces a groundbreaking methodology for synthesizing carbazole 1,3-position Friedel-Crafts ring-closed cyclic compounds, representing a significant leap forward in the design of organic nanogrids. Unlike traditional macrocycles, these nanogrid structures possess distinct vertices and side lengths on the nanometer scale, providing a rigid, three-dimensional closed-loop architecture that is critical for high-performance optoelectronic applications. This technology specifically addresses the longstanding challenge of chirality in fluorene-based systems by creating achiral Type I di-tertiary alcohol synthons that link the 9-position of fluorene directly to the 1,3-positions of carbazole. 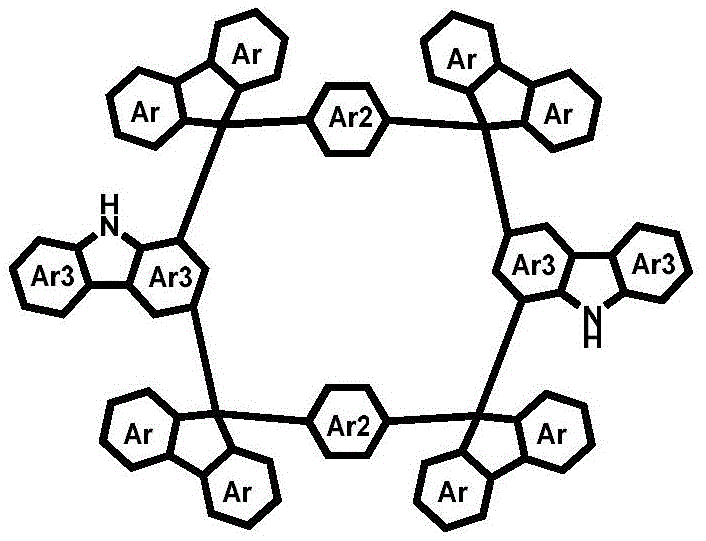 The resulting molecules exhibit superior molecular rigidity and adjustable cavity sizes, making them ideal candidates as a reliable OLED material supplier would seek for next-generation display technologies. By breaking away from the conventional 3,6-position ring-closing modes, this invention offers a pathway to materials with enhanced solubility and processability, which are essential parameters for the commercial scale-up of complex organic semiconductors.
The resulting molecules exhibit superior molecular rigidity and adjustable cavity sizes, making them ideal candidates as a reliable OLED material supplier would seek for next-generation display technologies. By breaking away from the conventional 3,6-position ring-closing modes, this invention offers a pathway to materials with enhanced solubility and processability, which are essential parameters for the commercial scale-up of complex organic semiconductors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of fluorene-carbazole hybrid structures has been plagued by stereochemical complications and synthetic inefficiencies. Traditional Friedel-Crafts reactions often target the 3,6-positions of the carbazole moiety, which, when combined with the inherent potential chirality at the 9-position of the fluorenyl group, leads to the formation of complex mixtures of chiral isomers. This stereochemical ambiguity necessitates rigorous and often low-yielding separation processes, drastically increasing the cost reduction in electronic chemical manufacturing efforts. Furthermore, the resulting 3,6-closed products frequently suffer from poor solubility in common organic solvents, hindering their application in solution-processable devices such as organic light-emitting diodes (OLEDs) and organic field-effect transistors (OFETs). The reliance on harsh conditions and the generation of difficult-to-remove impurities have limited the widespread adoption of these materials in high-volume production environments where consistency and purity are paramount.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by shifting the cyclization site to the 1,3-positions of the carbazole ring. This strategic modification results in a structurally rigid, achiral nanogrid that eliminates the formation of chiral isomers, thereby simplifying the purification workflow and ensuring batch-to-batch consistency. The new approach utilizes a simple, metal-free catalytic system that operates under mild conditions, significantly enhancing the atom economy and environmental profile of the synthesis. As demonstrated in the specific embodiments, this route successfully produces various derivatives, including DBCZ and LBCZ analogues, with manageable reaction times and straightforward workup procedures. 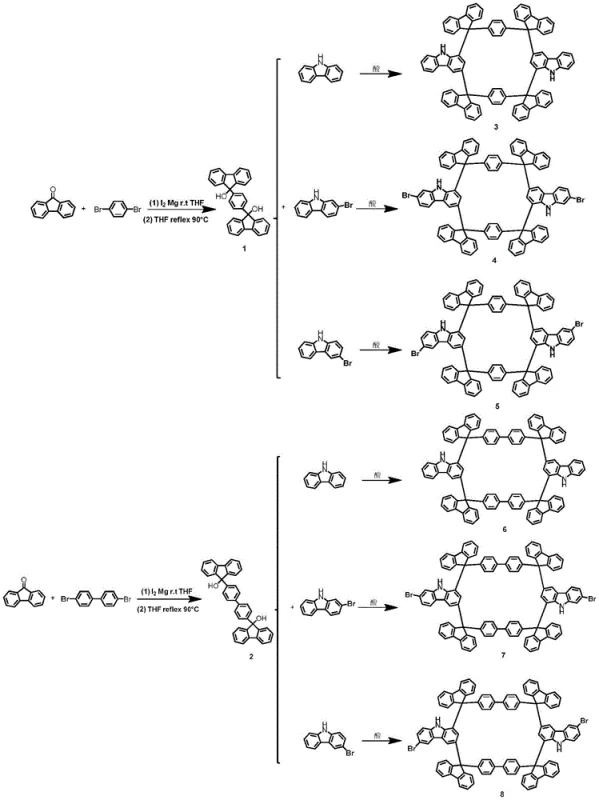 The versatility of this method allows for the incorporation of various substituents, such as alkyl chains or halogens, enabling fine-tuning of the electronic energy levels and solubility profiles. This flexibility is crucial for developing high-purity organic semiconductor materials tailored for specific device architectures, offering a robust platform for innovation in the field of organic molecular materials.
The versatility of this method allows for the incorporation of various substituents, such as alkyl chains or halogens, enabling fine-tuning of the electronic energy levels and solubility profiles. This flexibility is crucial for developing high-purity organic semiconductor materials tailored for specific device architectures, offering a robust platform for innovation in the field of organic molecular materials.
Mechanistic Insights into Metal-Free Friedel-Crafts Cyclization
The core of this synthetic breakthrough lies in a meticulously orchestrated two-step sequence that prioritizes both efficiency and structural integrity. The process initiates with the formation of a Type I di-tertiary alcohol synthon, achieved through a Grignard reaction between p-bromobenzene derivatives and fluorenone. This step requires precise control, utilizing a molar ratio of magnesium particles to p-bromobenzene to fluorenone of approximately 3:1:2.5, with the Grignard reagent formation occurring at 60°C for 7 hours followed by reaction with fluorenone at 90°C for 24 hours. The resulting synthon serves as a pivotal intermediate, possessing the necessary steric and electronic properties to facilitate the subsequent ring-closing event. The second step involves an acid-catalyzed Friedel-Crafts cyclization where the synthon reacts with carbazole or its derivatives in a dry organic solvent. 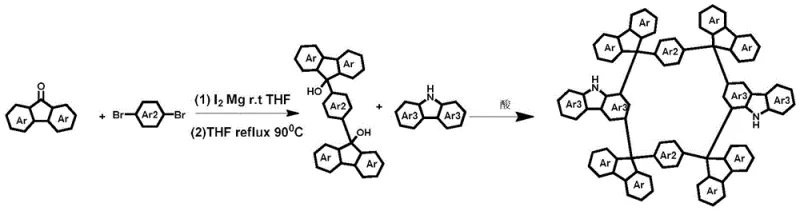 The use of strong Lewis acids such as trifluoromethanesulfonic acid or boron trifluoride etherate at room temperature drives the electrophilic aromatic substitution, effectively linking the fluorene 9-position to the carbazole 1,3-positions. This mechanism avoids the use of transition metals, which is a critical factor for reducing lead time for high-purity organic semiconductors, as it eliminates the need for extensive heavy metal scavenging steps that often complicate downstream processing.
The use of strong Lewis acids such as trifluoromethanesulfonic acid or boron trifluoride etherate at room temperature drives the electrophilic aromatic substitution, effectively linking the fluorene 9-position to the carbazole 1,3-positions. This mechanism avoids the use of transition metals, which is a critical factor for reducing lead time for high-purity organic semiconductors, as it eliminates the need for extensive heavy metal scavenging steps that often complicate downstream processing.
Impurity control is inherently built into this mechanistic pathway due to the high regioselectivity of the 1,3-substitution pattern on the carbazole ring. By ensuring that the carbazole NH position remains unsubstituted, the reaction is directed exclusively towards the desired 1,3-cyclization, preventing the formation of the undesired 3,6-isomers that plague conventional methods. The rigid geometry of the transition state further disfavors alternative cyclization pathways, resulting in a cleaner crude product profile. This high level of selectivity minimizes the burden on purification teams, allowing for more efficient isolation of the target nanogrid compounds via standard silica gel chromatography. The structural confirmation via NMR, MALDI-TOF-MS, and XRD validates the formation of the intended closed-lattice structure, confirming the success of this mechanistic strategy in producing well-defined molecular architectures.
How to Synthesize Carbazole Nanogrids Efficiently
The synthesis of these advanced nanogrid materials follows a standardized protocol designed for reproducibility and scalability in a laboratory or pilot plant setting. The process begins with the rigorous preparation of the Grignard reagent under inert atmosphere, followed by the addition of fluorenone to generate the di-tertiary alcohol precursor. Once the synthon is isolated and purified, it is subjected to the cyclization conditions using a catalytic amount of strong acid in a dry solvent like dichloromethane. Detailed standard operating procedures regarding stoichiometry, temperature profiles, and quenching methods are essential for achieving the reported yields and purity levels. For a comprehensive guide on the specific experimental parameters and safety considerations, please refer to the standardized synthesis steps outlined below.
- Prepare Type I di-tertiary alcohol synthon by reacting p-bromobenzene derivatives with magnesium and fluorenone in THF at elevated temperatures.
- Conduct the Friedel-Crafts cyclization by reacting the synthon with carbazole derivatives using a Lewis acid catalyst like trifluoromethanesulfonic acid at room temperature.
- Purify the resulting cyclic compound via silica gel column chromatography to achieve high-purity organic semiconductor materials.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented technology offers substantial benefits that align with the strategic goals of cost efficiency and operational reliability. The elimination of expensive transition metal catalysts, such as palladium or platinum complexes, directly translates to significant raw material cost savings and simplifies the procurement process. Furthermore, the absence of heavy metals in the final reaction mixture removes the necessity for specialized and costly metal scavenging resins or complex extraction protocols, streamlining the manufacturing workflow. This simplification not only reduces the overall production cost but also shortens the cycle time from synthesis to final packaging, enhancing the responsiveness of the supply chain to market demands. The use of common, commercially available starting materials like fluorenone and carbazole ensures a stable supply base, mitigating the risks associated with sourcing exotic or proprietary reagents.
- Cost Reduction in Manufacturing: The metal-free nature of the catalytic system fundamentally alters the cost structure of producing these high-value intermediates. By relying on abundant Lewis acids instead of precious metals, manufacturers can avoid the volatility associated with noble metal pricing. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal costs, contributing to a leaner and more economical production process. The high atom economy of the reaction ensures that a greater proportion of the input materials are converted into the desired product, minimizing waste generation and maximizing resource utilization efficiency.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain resilience by reducing dependency on complex catalytic systems that may be subject to supply disruptions. The mild reaction conditions, particularly the room temperature cyclization step, lower the energy requirements and equipment stress, allowing for more flexible production scheduling. This operational flexibility enables manufacturers to respond more quickly to fluctuations in demand, ensuring a consistent flow of high-quality materials to downstream customers. The scalability of the process from gram to kilogram scales has been demonstrated, providing confidence in the ability to meet large-volume orders without compromising quality.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior to traditional methods, aligning with increasingly stringent global regulations on chemical manufacturing. The avoidance of toxic heavy metals simplifies wastewater treatment and hazardous waste management, reducing the environmental footprint of the production facility. The use of standard organic solvents that can be easily recovered and recycled further supports sustainability initiatives. This green chemistry approach not only ensures compliance with environmental standards but also enhances the brand reputation of the manufacturer as a responsible supplier of advanced electronic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carbazole nanogrid compounds. The answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on the capabilities and limitations of this technology. Understanding these details is crucial for R&D teams evaluating the material for specific device applications and for procurement officers assessing the feasibility of long-term supply agreements.
Q: What is the primary advantage of 1,3-position cyclization over traditional 3,6-position methods?
A: The 1,3-position cyclization creates an achiral, rigid nanogrid structure that avoids the potential chirality issues at the fluorene 9-position found in other methods, resulting in easier separation and higher structural regularity.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the process utilizes common Lewis acids such as trifluoromethanesulfonic acid or boron trifluoride etherate, eliminating the need for costly palladium or platinum catalysts and simplifying purification.
Q: Are these carbazole nanogrids suitable for solution processing in OLED fabrication?
A: Yes, the resulting compounds exhibit improved solubility compared to traditional 3,6-closed products, making them highly suitable for solution-processing techniques required in modern display manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazole Nanogrid Supplier
As the demand for high-performance organic semiconductors continues to surge, partnering with an experienced CDMO is essential for translating innovative patents into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from the laboratory to the marketplace. Our state-of-the-art facilities are equipped to handle the specific requirements of air- and moisture-sensitive reactions, such as the Grignard steps involved in this synthesis, while our rigorous QC labs enforce stringent purity specifications to guarantee material consistency. We understand the critical nature of supply continuity in the electronics industry and are committed to delivering high-purity organic semiconductor materials that meet the exacting standards of global manufacturers.
We invite you to collaborate with us to leverage this cutting-edge technology for your next-generation products. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and process constraints. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for the future of organic electronics.
