Optimizing Amiodarone Intermediate Production: A Novel Carbonate-Catalyzed Cyclization Strategy
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical antiarrhythmic agents, particularly amiodarone hydrochloride. Patent CN108675972B introduces a significant technological advancement in the preparation of 2-butyl benzofuran, a pivotal intermediate in the synthesis of this life-saving medication. Traditional methods often suffer from complex multi-step sequences, harsh reaction conditions, and difficult purification protocols that hinder efficient manufacturing. This new methodology leverages a streamlined carbonate-catalyzed cyclization strategy that not only enhances reaction efficiency but also drastically simplifies the downstream processing workflow. By shifting from strong alkaline conditions to a milder carbonate system in a polar aprotic solvent, the process achieves superior purity profiles while minimizing waste generation. For global supply chain leaders, this represents a vital opportunity to secure a more reliable source of high-quality intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-butyl benzofuran has been plagued by inefficient protocols that rely on aggressive reagents and cumbersome isolation techniques. Prior art, such as the methods disclosed in CN1858042A, typically employs strong bases like sodium hydroxide in acetone, necessitating prolonged reflux times often exceeding ten hours. Furthermore, these legacy processes require intricate post-reaction treatments, including acidification to strong acidity, phase separation with toluene, and subsequent azeotropic distillation to remove water, which can take an additional six hours. Such rigorous conditions not only increase energy consumption but also elevate the risk of forming undesirable by-products, leading to lower overall yields and necessitating further purification steps like distillation or column chromatography. These factors collectively contribute to higher production costs and extended lead times, creating bottlenecks for manufacturers aiming to meet the growing global demand for amiodarone.
The Novel Approach
In stark contrast, the innovative process detailed in CN108675972B utilizes a温和 yet effective catalytic system comprising potassium carbonate and potassium iodide in dimethylformamide (DMF). This approach facilitates a rapid cyclization of methyl 2-(2-formylphenoxy)hexanoate under reflux conditions, typically completing the reaction within a significantly shorter timeframe of approximately 1.5 to 5 hours. The use of a weak base mitigates the formation of complex impurities, allowing the crude product to be isolated simply by aqueous workup and organic extraction. This eliminates the need for the tedious acidification and azeotropic drying steps found in older methods. Consequently, the final product is obtained as a yellow transparent liquid with high purity, ready for subsequent synthetic steps without the need for further chromatographic purification. 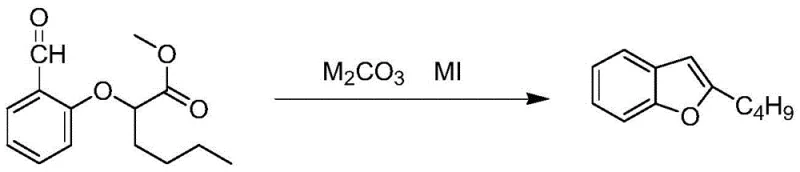
Mechanistic Insights into Carbonate-Catalyzed Cyclization
The core of this technological breakthrough lies in the precise selection of the catalytic system and solvent environment to drive the intramolecular cyclization. The reaction initiates with the deprotonation of the alpha-carbon adjacent to the ester group by the carbonate base, generating a nucleophilic enolate species. In the presence of potassium iodide, which likely acts as a phase transfer catalyst or nucleophilic promoter, the enolate attacks the ortho-formyl group on the aromatic ring. This intramolecular aldol-type condensation is followed by dehydration and subsequent decarboxylation to form the furan ring system. The choice of DMF as the solvent is critical, as its high polarity stabilizes the charged intermediates and allows the reaction to proceed at elevated temperatures (100-150°C) without degradation. This mechanistic pathway ensures a clean conversion with minimal side reactions, distinguishing it from the chaotic decomposition often observed under strongly alkaline conditions.
Impurity control is inherently built into this mechanism due to the mildness of the potassium carbonate catalyst. Unlike strong bases that can induce polymerization or hydrolysis of sensitive functional groups, the carbonate system maintains a controlled pH environment that favors the desired cyclization pathway. The absence of harsh acidic workups further preserves the integrity of the benzofuran scaffold. Analytical data from the patent indicates that the resulting reaction mixture contains significantly fewer impurities compared to prior art, as evidenced by TLC monitoring which shows the disappearance of raw materials with minimal spot formation for by-products. This high selectivity reduces the burden on quality control laboratories and ensures that the intermediate meets stringent specifications required for pharmaceutical grade synthesis.
How to Synthesize 2-Butyl Benzofuran Efficiently
The operational simplicity of this new route makes it highly attractive for process chemists looking to implement robust manufacturing protocols. The procedure involves charging the reaction vessel with the substrate, potassium carbonate, and potassium iodide in DMF, followed by heating to reflux until conversion is complete. Upon cooling, the mixture is treated with water and an extraction solvent like n-hexane to isolate the product. This straightforward workflow minimizes operator intervention and reduces the potential for human error during complex manipulations. For a detailed breakdown of the specific molar ratios, temperature controls, and safety considerations, please refer to the standardized synthesis guide below.
- React methyl 2-(2-formylphenoxy)hexanoate with potassium carbonate and potassium iodide in DMF under reflux until raw materials are consumed.
- Cool the reaction mixture and perform liquid-liquid extraction using water and an organic solvent such as n-hexane.
- Separate the organic phase, dry, and concentrate under reduced pressure to obtain high-purity 2-butyl benzofuran.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers substantial strategic benefits for procurement managers and supply chain directors. The elimination of complex purification steps such as column chromatography and azeotropic distillation translates directly into reduced operational expenditures and faster batch turnover. By simplifying the process flow, manufacturers can achieve higher throughput with existing infrastructure, thereby enhancing supply security for downstream API producers. Furthermore, the use of readily available and cost-effective reagents like potassium carbonate and DMF ensures stable raw material sourcing, mitigating the risks associated with specialty chemical shortages.
- Cost Reduction in Manufacturing: The new method significantly lowers production costs by removing the need for expensive silica gel columns and the large volumes of solvents required for chromatography. Additionally, the shorter reaction times and reduced energy consumption from eliminating prolonged azeotropic distillation contribute to a leaner cost structure. The high yield and purity achieved mean less material is wasted, maximizing the return on investment for every kilogram of starting material purchased.
- Enhanced Supply Chain Reliability: Simplifying the synthesis reduces the number of potential failure points in the manufacturing process, leading to more consistent batch-to-batch quality and reliability. The robustness of the reaction conditions allows for greater flexibility in production scheduling, ensuring that delivery timelines can be met even during periods of high demand. This stability is crucial for maintaining uninterrupted supply chains for critical cardiovascular medications.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard unit operations that are easily transferred from laboratory to pilot and commercial scales. The reduction in solvent usage and the avoidance of hazardous acidic waste streams align with modern green chemistry principles, facilitating easier regulatory compliance and waste management. This environmental stewardship not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-butyl benzofuran using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their supply networks.
Q: What are the advantages of using potassium carbonate over sodium hydroxide in this synthesis?
A: Using potassium carbonate as a weak base avoids the harsh conditions associated with strong bases like sodium hydroxide, reducing side reactions and simplifying the workup process by eliminating the need for acidification and azeotropic distillation.
Q: Is column chromatography required for purifying the final 2-butyl benzofuran product?
A: No, the novel method described in patent CN108675972B yields a product of sufficiently high purity through simple extraction and concentration, completely avoiding the need for costly and time-consuming column chromatography.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the process utilizes common industrial solvents like DMF and n-hexane and involves straightforward unit operations like reflux and extraction, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Butyl Benzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major API manufacturers. We are committed to delivering 2-butyl benzofuran with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to implement the efficient carbonate-catalyzed synthesis described in CN108675972B allows us to offer a product that is both cost-effective and reliable.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our supply source. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your production of amiodarone hydrochloride remains competitive and uninterrupted.
