Advanced One-Pot Synthesis of Tulobuterol: Enhancing Yield and Safety for Commercial Production
The pharmaceutical landscape for respiratory therapeutics continues to evolve, demanding more efficient and sustainable manufacturing processes for key active ingredients like tulobuterol. Patent CN110590569B introduces a groundbreaking synthetic methodology that addresses critical inefficiencies in the production of this selective beta-2 receptor agonist. Historically, the synthesis of tulobuterol has been plagued by low atom economy and the generation of hazardous waste, particularly during the halogenation steps. This new disclosure presents a robust solution by transforming a previously discarded byproduct into a valuable reactant, thereby optimizing the entire value chain. For R&D directors and procurement specialists, this represents a significant opportunity to enhance supply chain resilience while adhering to stricter environmental regulations. The core innovation lies in the simultaneous utilization of both mono-brominated and di-brominated intermediates, a strategy that fundamentally alters the economic model of producing high-purity tulobuterol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
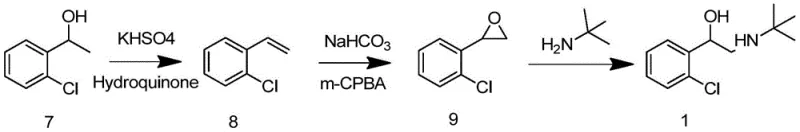
Traditional synthetic pathways for tulobuterol have long suffered from inherent structural inefficiencies and safety hazards that impede cost-effective manufacturing. As illustrated in the legacy routes, early methods relied heavily on the direct use of elemental bromine, a highly corrosive and toxic reagent that poses severe risks to personnel and requires extensive containment infrastructure. 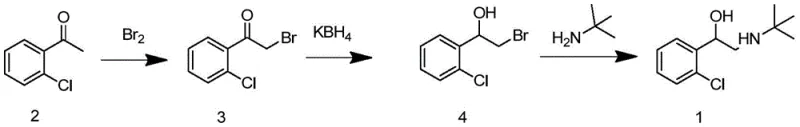 Furthermore, alternative approaches utilizing selenium dioxide for oxidation introduce heavy metal toxicity concerns, creating substantial burdens for waste treatment and environmental compliance. Another reported pathway involves high-temperature olefination reactions exceeding 200°C, which not only consume excessive energy but also generate complex impurity profiles that are difficult to purge.
Furthermore, alternative approaches utilizing selenium dioxide for oxidation introduce heavy metal toxicity concerns, creating substantial burdens for waste treatment and environmental compliance. Another reported pathway involves high-temperature olefination reactions exceeding 200°C, which not only consume excessive energy but also generate complex impurity profiles that are difficult to purge. 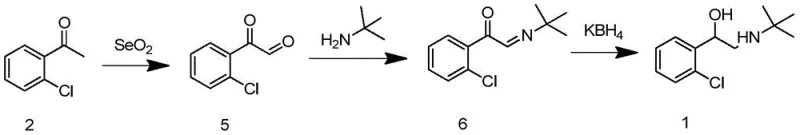
 These conventional methods often result in the formation of 1-(2-chlorophenyl)-2,2-dibromoethylketone (Compound 10) as an unavoidable byproduct during the initial bromination of the ketone precursor. In standard practice, this dibromo species constitutes roughly 10-15% of the reaction mass and is typically separated and discarded, representing a direct loss of raw materials and a reduction in overall process yield.
These conventional methods often result in the formation of 1-(2-chlorophenyl)-2,2-dibromoethylketone (Compound 10) as an unavoidable byproduct during the initial bromination of the ketone precursor. In standard practice, this dibromo species constitutes roughly 10-15% of the reaction mass and is typically separated and discarded, representing a direct loss of raw materials and a reduction in overall process yield.
The Novel Approach
The patented methodology revolutionizes this landscape by adopting a "waste-to-value" philosophy, integrating the previously discarded dibromo byproduct directly into the final synthesis step. Instead of attempting the energetically costly and chemically difficult separation of the monobromo compound (Compound 3) from the dibromo impurity (Compound 10), the new process utilizes the crude mixture directly. 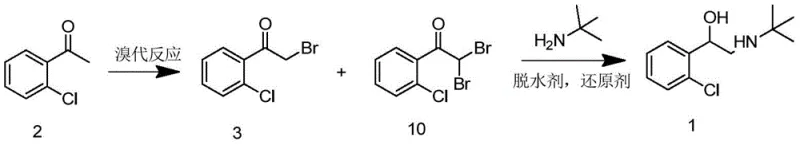 This one-pot strategy involves reacting the mixture with tert-butylamine in the presence of a dehydrating agent and a reducing agent, effectively converting both species into the desired tulobuterol backbone. By eliminating the purification of the intermediate haloketones, the process drastically reduces solvent consumption and processing time. This approach not only simplifies the operational workflow but also significantly boosts the total molar yield, reportedly improving efficiency by approximately 10% compared to the original routes. For a reliable tulobuterol intermediate supplier, this translates to a more competitive cost structure and a reduced environmental footprint, aligning perfectly with modern green chemistry principles.
This one-pot strategy involves reacting the mixture with tert-butylamine in the presence of a dehydrating agent and a reducing agent, effectively converting both species into the desired tulobuterol backbone. By eliminating the purification of the intermediate haloketones, the process drastically reduces solvent consumption and processing time. This approach not only simplifies the operational workflow but also significantly boosts the total molar yield, reportedly improving efficiency by approximately 10% compared to the original routes. For a reliable tulobuterol intermediate supplier, this translates to a more competitive cost structure and a reduced environmental footprint, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into One-Pot Reductive Amination
The chemical elegance of this process lies in its ability to manage the reactivity of two distinct electrophilic species within a single reaction vessel. The mechanism initiates with the nucleophilic attack of tert-butylamine on the carbonyl carbon of both the monobromo and dibromo ketones, forming imine or iminium intermediates. The presence of a dehydrating agent, such as anhydrous sodium sulfate or magnesium sulfate, is critical here to drive the equilibrium towards imine formation by sequestering the water produced during condensation. Subsequently, the addition of a hydride reducing agent, typically sodium borohydride or potassium borohydride, facilitates the reduction of these nitrogen-containing intermediates to the corresponding amines. Crucially, the reaction conditions are meticulously controlled, starting at low temperatures (around -10°C to 10°C) during the reduction phase to prevent side reactions, followed by a reflux period to ensure complete conversion. This careful thermal management ensures that the more reactive dibromo species does not undergo unwanted elimination or degradation, but rather follows the desired reductive amination pathway to yield the target beta-amino alcohol structure.
From an impurity control perspective, this unified approach offers superior selectivity compared to stepwise syntheses. In traditional multi-step routes, isolating the alpha-halo ketone often leads to decomposition or polymerization, introducing hard-to-remove impurities that persist through to the final API. By keeping the reactive haloketones in solution and immediately converting them to the more stable amine product, the formation of degradation byproducts is minimized. The use of alcoholic solvents like ethanol or methanol further aids in solubilizing the transition states and stabilizing the borohydride species, ensuring a clean reaction profile. This mechanistic robustness is vital for meeting the stringent purity specifications required for respiratory drug intermediates, where trace impurities can impact the safety profile of the final inhalation or patch therapy products. The result is a high-purity tulobuterol output that requires less intensive downstream purification, streamlining the path from kilogram to metric ton scale.
How to Synthesize Tulobuterol Efficiently
The execution of this synthesis requires precise control over stoichiometry and thermal conditions to maximize the conversion of the brominated mixture. The process begins with the preparation of the haloketone mixture, followed by a carefully orchestrated one-pot reductive amination sequence. Operators must adhere to strict temperature protocols during the addition of the reducing agent to maintain safety and selectivity. The detailed standardized synthesis steps, including specific reagent ratios and workup procedures, are outlined in the guide below.
- Prepare a mixture of 1-(2-chlorophenyl)-2-bromoethanone (Compound 3) and 1-(2-chlorophenyl)-2,2-dibromoethylketone (Compound 10) via bromination of 1-(2-chlorophenyl)-ethanone.
- Conduct a one-pot reaction with tert-butylamine, a dehydrating agent, and a reducing agent (e.g., sodium borohydride) in an alcoholic solvent.
- Perform extraction and acid-base workup to isolate high-purity tulobuterol with improved molar yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling strategic advantages that extend beyond simple yield improvements. The primary value driver is the significant reduction in raw material costs achieved by valorizing what was previously considered waste. By utilizing the dibromo byproduct rather than discarding it, the effective cost per kilogram of the starting ketone is lowered, directly impacting the bottom line. Additionally, the elimination of intermediate isolation steps reduces the consumption of solvents and energy, contributing to substantial cost reduction in pharmaceutical intermediates manufacturing. The simplified workflow also means fewer unit operations, which lowers the risk of batch failures and equipment downtime, thereby enhancing overall supply chain reliability. This efficiency allows suppliers to offer more competitive pricing while maintaining healthy margins, a critical factor in the highly price-sensitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the fundamental improvement in atom economy. By avoiding the separation of Compound 3 and Compound 10, manufacturers save on the significant costs associated with distillation or crystallization of unstable intermediates. Furthermore, the ability to use safer brominating agents like N-bromosuccinimide (NBS) instead of elemental bromine reduces the need for specialized corrosion-resistant equipment and expensive safety mitigation systems. The qualitative reduction in waste disposal costs, particularly regarding hazardous halogenated organic waste and heavy metals like selenium, further enhances the financial viability of the process. These cumulative savings allow for a more robust pricing strategy in the global market for respiratory drug precursors.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on hazardous or regulated reagents. This new method mitigates those risks by enabling the use of solid, easier-to-handle brominating agents and common reducing agents like sodium borohydride. The robustness of the one-pot reaction reduces the sensitivity to minor variations in operating conditions, leading to more consistent batch-to-batch quality. For a reliable tulobuterol intermediate supplier, this consistency is paramount to securing long-term contracts with major pharmaceutical companies. The reduced complexity of the process also shortens the production cycle time, allowing for faster response to fluctuations in market demand and reducing lead time for high-purity respiratory drug intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this methodology is inherently designed for industrial expansion. The reaction operates at near-ambient temperatures for the critical amination step, reducing the thermal load on reactors and cooling systems. The avoidance of toxic selenium compounds and elemental bromine aligns the process with increasingly strict global environmental regulations, such as REACH in Europe. This compliance future-proofs the supply chain against regulatory bans or restrictions on specific reagents. Moreover, the simplified effluent profile makes wastewater treatment more manageable, facilitating the commercial scale-up of complex beta-agonists without incurring prohibitive environmental remediation costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel tulobuterol synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: How does this new method improve upon traditional tulobuterol synthesis routes?
A: Traditional routes often discard the dibromo byproduct (Compound 10) generated during bromination, leading to material loss. This patented method utilizes both the monobromo and dibromo species in a single pot, increasing total molar yield by approximately 10% and eliminating the need for difficult intermediate separations.
Q: What are the safety advantages of this process compared to using elemental bromine?
A: While early routes utilized hazardous elemental bromine, this optimized process can employ safer brominating agents like N-bromosuccinimide (NBS). Furthermore, by avoiding the isolation of unstable alpha-halo ketones and proceeding directly to the stable amine product, the overall process safety profile is significantly enhanced for industrial operators.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the method is explicitly designed for industrial scalability. It operates under mild temperatures (20-30°C initially) and uses common solvents like ethanol or methanol. The one-pot nature reduces unit operations, lowering capital expenditure and operational complexity, making it ideal for reliable tulobuterol intermediate suppliers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tulobuterol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised by patent CN110590569B are fully realized in a GMP-compliant environment. We are committed to delivering high-purity tulobuterol that meets stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Our capability to manage complex halogenation and reductive amination chemistries safely positions us as a strategic partner for your respiratory drug supply chain.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your product's competitiveness in the global marketplace.
