Advanced Liquid Phase Oxidation Technology for Commercial Scale Production of 4-Bromophthalic Acid
Introduction to High-Purity 4-Bromophthalic Acid Manufacturing
The global demand for high-performance aromatic polyimides has surged in recent years, driven by critical applications in aerospace engineering, flexible electronics, and advanced automotive manufacturing. At the heart of this material revolution lies 4-bromophthalic acid, a specialized intermediate whose purity and structural integrity directly dictate the thermal stability and dielectric properties of the final polymer. Patent CN115010592A introduces a groundbreaking preparation method that addresses the longstanding bottlenecks of yield, safety, and environmental impact associated with traditional synthesis routes. By leveraging a sophisticated liquid phase catalytic oxidation system, this technology enables the production of high-purity 4-bromophthalic acid with yields exceeding 80%, setting a new benchmark for industrial efficiency.
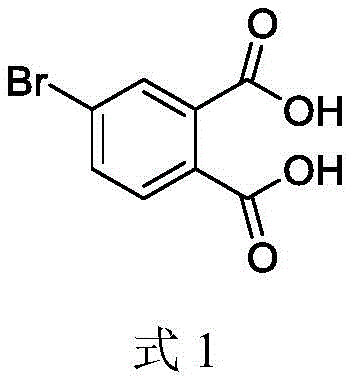
This proprietary methodology not only optimizes the reaction kinetics but also integrates a robust solvent recovery system, ensuring that the process is economically viable for large-scale operations. For R&D directors and procurement specialists seeking a reliable fine chemical intermediates supplier, understanding the nuances of this patented route is essential for securing a stable supply chain of this critical electronic chemical. The following analysis dissects the technical innovations that make this process superior to conventional methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-bromophthalic acid has been plagued by inefficient oxidation strategies that compromise both economic viability and environmental safety. Traditional approaches often rely on stoichiometric oxidants such as potassium permanganate, which, while effective in laboratory settings, result in abysmal yields of approximately 48% and generate vast quantities of hazardous manganese dioxide waste. Furthermore, alternative catalytic methods utilizing pure oxygen from the onset of the reaction pose significant safety risks due to uncontrolled exothermic temperature spikes, which can reach dangerous levels of 15-20°C above baseline, leading to potential runaway reactions and reduced selectivity. These legacy processes also suffer from poor atom economy and complex separation steps, making them unsuitable for the rigorous demands of modern commercial scale-up of complex polymer additives.
The Novel Approach
In stark contrast, the novel approach detailed in the patent employs a variable composition gas oxidizing strategy combined with a highly active cobalt-manganese catalytic system. By initially introducing air to gently initiate the oxidation and subsequently switching to pure oxygen to drive the reaction to completion, the process maintains a minimal temperature rise of only 5-8°C, drastically improving operational safety. This method achieves a remarkable yield improvement, consistently delivering over 80% conversion with purity levels exceeding 98.5%. The integration of tetrabromoethane as a promoter further enhances the reaction efficiency, allowing for reduced catalyst loading while preventing the formation of undesirable by-products. This represents a paradigm shift in cost reduction in electronic chemical manufacturing, transforming a hazardous, low-yield process into a streamlined, high-throughput operation.
Mechanistic Insights into Co-Mn Catalyzed Liquid Phase Oxidation
The core of this technological breakthrough lies in the synergistic interaction between cobalt acetate and manganese acetate within an acetic acid solvent matrix. The mechanism operates via a free-radical chain reaction where the metal catalysts facilitate the decomposition of hydroperoxide intermediates, generating the active radicals necessary for the oxidation of the methyl groups on the 4-bromo-o-xylene substrate. The addition of tetrabromoethane serves as a critical initiator, providing a source of bromine radicals that accelerate the propagation step of the chain reaction. This allows the system to operate effectively even with lower concentrations of the transition metal catalysts, which is a significant departure from prior art that required excessive catalyst loading to achieve acceptable conversion rates. The precise control of the oxidation state ensures that the aromatic ring remains intact while the alkyl side chains are fully oxidized to carboxylic acids.
Furthermore, the impurity control mechanism is inherently built into the reaction conditions. The use of a variable gas feed (air followed by oxygen) prevents the localized overheating that typically leads to decarboxylation or ring-opening side reactions. The specific pressure maintenance at 1.2-1.5 MPa ensures that the oxygen concentration in the liquid phase remains optimal throughout the reaction timeline. Post-reaction, the implementation of azeotropic distillation with n-heptane allows for the efficient removal of the acetic acid solvent and water by-products, facilitating the crystallization of the target molecule. This rigorous control over the reaction environment results in a product with a significantly cleaner impurity profile, reducing the burden on downstream purification units and ensuring consistent quality for high-purity OLED material or polyimide precursor applications.
How to Synthesize 4-Bromophthalic Acid Efficiently
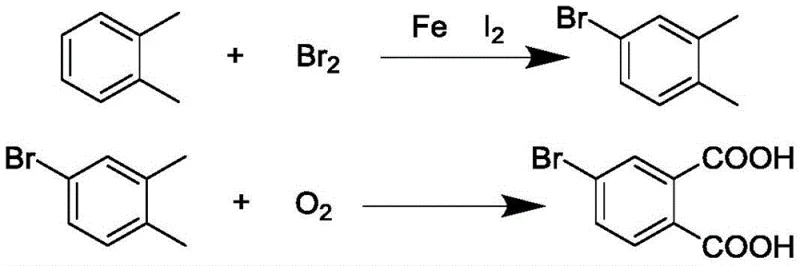
The synthesis protocol outlined in the patent provides a clear roadmap for replicating these high-yield results in a pilot or production setting. The process begins with the careful bromination of o-xylene under controlled低温 conditions to ensure regioselectivity, followed by the critical oxidation step in a high-pressure autoclave. Operators must strictly adhere to the gas switching timeline—introducing air for the initial hour to stabilize the radical generation before transitioning to oxygen for the bulk of the conversion. The detailed standardized synthesis steps below outline the precise reagent ratios, temperature profiles, and workup procedures required to achieve the reported 80.6% yield and 99% purity.
- Prepare 4-bromo-o-xylene by reacting o-xylene with liquid bromine using iron powder and iodine as catalysts at low temperature.
- Perform liquid phase oxidation of 4-bromo-o-xylene in acetic acid using cobalt acetate, manganese acetate, and tetrabromoethane, introducing air first then oxygen at 150-220°C.
- Recover solvents via azeotropic distillation with n-heptane and isolate the final product through alkaline dissolution and acid precipitation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple yield improvements. The shift from stoichiometric oxidants to a catalytic aerobic oxidation system fundamentally alters the cost structure of production by eliminating the need for expensive and waste-generating reagents like potassium permanganate. Additionally, the ability to recover and recycle the acetic acid solvent and the n-heptane entrainer creates a closed-loop system that minimizes raw material consumption and waste disposal costs. This efficiency translates directly into a more competitive pricing structure for the final product, providing a distinct advantage in cost reduction in polymer additive manufacturing without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of heavy metal waste streams and the reduction in catalyst loading significantly lower the operational expenditure associated with waste treatment and raw material procurement. By utilizing air as a partial oxidant, the process reduces the consumption of pure oxygen, further driving down utility costs. The high recovery rate of the solvent system means that fresh solvent purchases are minimized, leading to substantial long-term savings that enhance the overall margin profile of the manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as o-xylene, acetic acid, and air/oxygen ensures that the supply chain is resilient against the volatility often seen with specialized reagents. The robustness of the catalytic system allows for consistent batch-to-batch reproducibility, which is critical for maintaining uninterrupted supply to downstream polymer manufacturers. This stability reduces the risk of production delays and ensures that customers receive their orders of high-purity 4-bromophthalic acid on schedule, reinforcing trust in the supplier relationship.
- Scalability and Environmental Compliance: The inherent safety features of the variable gas oxidation method, specifically the controlled temperature profile, make the process highly scalable from pilot plants to multi-ton commercial reactors without the need for exotic cooling infrastructure. Furthermore, the drastic reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations, future-proofing the production facility against regulatory changes. This commitment to green chemistry principles not only mitigates legal risks but also enhances the brand reputation of the manufacturer as a sustainable partner in the value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the practical aspects of the process. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific application needs.
Q: What is the primary advantage of the variable gas oxidation method described in CN115010592A?
A: The method utilizes a sequential introduction of air followed by pure oxygen. This strategy mitigates the severe temperature spikes associated with pure oxygen initiation, enhancing safety while maintaining high reaction rates and selectivity in the later stages.
Q: How does this process improve environmental compliance compared to potassium permanganate oxidation?
A: Unlike stoichiometric oxidations using potassium permanganate which generate massive amounts of manganese dioxide sludge, this catalytic liquid phase oxidation uses molecular oxygen from air, significantly reducing solid waste discharge and simplifying downstream purification.
Q: What role does tetrabromoethane play in the catalytic system?
A: Tetrabromoethane acts as a highly effective initiator (promoter) for the radical chain reaction. Its presence allows for a substantial reduction in the loading of expensive cobalt and manganese catalysts while maintaining high conversion rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Bromophthalic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic oxidation techniques described in patent CN115010592A can be seamlessly integrated into your supply chain. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4-bromophthalic acid meets the exacting standards required for high-performance polyimide and electronic material applications.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits tailored to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of scientific excellence and commercial reliability.
