Advanced Synthesis of Ethyl 4-(2-Chloroethyl)thiazole-2-Carboxylate for Cardiovascular Drug Intermediates
Introduction to the Novel Synthetic Route
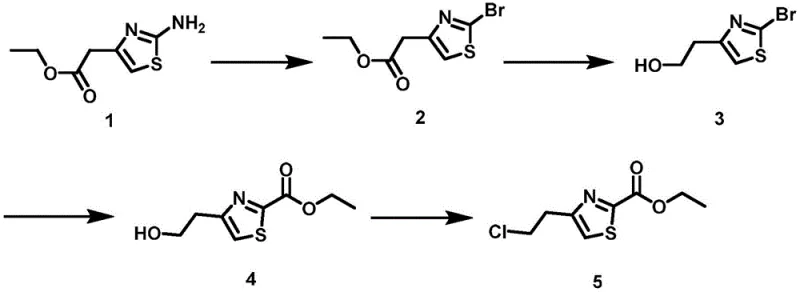
The pharmaceutical industry is constantly seeking robust and scalable pathways for complex heterocyclic intermediates, particularly those serving as building blocks for cardiovascular therapeutics. Patent CN113461635A discloses a groundbreaking preparation method for 4-(2-chloroethyl)thiazole-2-carboxylic acid ethyl ester, a critical intermediate in the synthesis of APJ receptor agonists. This technology addresses the long-standing lack of reported synthesis routes for this specific molecule, offering a stable, low-cost, and operationally simple four-step process. By starting from the commercially accessible 2-(2-aminothiazole-4-yl) ethyl acetate, the invention optimizes functional group conversions on the thiazole ring to ensure process stability. The route features a mild diazotization-halogenation step, a selective reduction, a palladium-catalyzed carbonyl insertion, and a final chlorination, collectively providing a reliable solution for high-purity pharmaceutical intermediates.
The strategic significance of this patent lies in its ability to bypass the harsh conditions typically associated with thiazole functionalization. Traditional approaches often suffer from low yields and difficult temperature controls, creating bottlenecks in the supply chain for heart failure medications. This new methodology not only improves the yield of the initial bromination step but also streamlines the entire workflow, making it an attractive option for commercial scale-up of complex pharmaceutical intermediates. For R&D teams focused on APJ receptor modulators, this route offers a viable path to secure raw material supply while maintaining stringent quality standards required for clinical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art references, such as US2012/108565 and WO 2018/106636, describe the synthesis of related thiazole derivatives but often rely on cumbersome reaction conditions that hinder industrial adoption. Specifically, the synthesis of the brominated intermediate typically requires strict temperature control below -20°C to manage the exothermic nature of the diazotization and subsequent halogenation. Such cryogenic requirements impose a heavy burden on manufacturing infrastructure, necessitating specialized cooling equipment and increasing energy consumption significantly. Furthermore, the operational complexity of maintaining sub-zero temperatures over extended reaction times increases the risk of human error and batch-to-batch variability. These factors contribute to higher production costs and longer lead times, creating friction in the cost reduction in pharmaceutical intermediate manufacturing initiatives that procurement managers strive to achieve.
The Novel Approach
In stark contrast, the novel approach detailed in CN113461635A introduces a optimized bromination protocol that operates effectively at room temperature. By utilizing benzyltriethylammonium bromide as a dual-function reagent acting as both a phase transfer catalyst and a halogen atom donor, the reaction activity is sufficiently enhanced to proceed without extreme cooling. This modification drastically simplifies the operational procedure, removing the need for energy-intensive cryogenic baths and allowing for more flexible reactor scheduling. The subsequent steps, including reduction and carbonylation, are also designed with mild conditions in mind, ensuring that the overall process remains stable and reproducible. This shift towards ambient temperature reactions represents a significant technological leap, facilitating easier commercial scale-up and reducing the barrier to entry for manufacturers aiming to supply this critical cardiovascular intermediate.
Mechanistic Insights into Pd-Catalyzed Carbonylation
The core of this synthetic strategy revolves around the efficient construction of the ester functionality via a transition metal-catalyzed carbonyl insertion. In the third step of the sequence, the bromo-alcohol intermediate undergoes a sophisticated transformation in the presence of carbon monoxide and a palladium catalyst. The mechanism initiates with the oxidative addition of the aryl bromide to the palladium center, forming a transition metal complex. Carbon monoxide from the system then coordinates with the metal, followed by migratory insertion of the carbonyl group into the carbon-palladium bond to generate an acyl metal species. This acyl intermediate subsequently undergoes ligand exchange with the solvent or added alcohol, followed by reductive elimination to release the desired ethyl ester product. This mechanistic pathway allows for the direct installation of the carboxylate group with high regioselectivity, avoiding the need for harsher oxidation states or multi-step functional group manipulations.
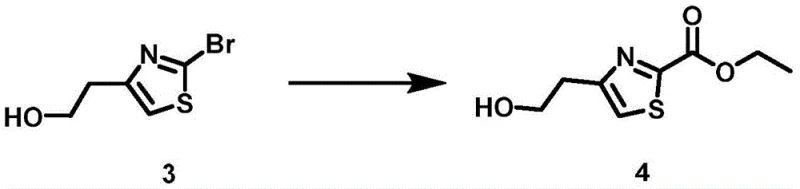
From an impurity control perspective, the use of a well-defined palladium catalyst system, specifically [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium, ensures a cleaner reaction profile compared to non-catalytic alternatives. The mild reaction temperature of 80°C prevents thermal degradation of the sensitive thiazole ring, which can often occur under more vigorous conditions. Furthermore, the post-treatment process is streamlined; after filtration to remove the catalyst, the solvent can be removed, and the product purified via neutral alumina column chromatography. This level of control over the reaction mechanism translates directly to a superior impurity profile, a critical metric for R&D Directors evaluating the feasibility of a synthesis route for GMP production. The ability to minimize side products reduces the burden on downstream purification, thereby enhancing overall process efficiency.
How to Synthesize Ethyl 4-(2-chloroethyl)thiazole-2-carboxylate Efficiently
The execution of this four-step synthesis requires precise adherence to the molar ratios and reaction conditions outlined in the patent to maximize yield and purity. The process begins with the room temperature bromination, followed by a controlled reduction using sodium borohydride. The pivotal carbonylation step demands a carbon monoxide atmosphere and specific catalyst loading, while the final chlorination utilizes thionyl chloride under cooled conditions to prevent over-reaction. Each stage has been optimized to balance reaction kinetics with product stability, ensuring that the final chloroethyl derivative is obtained with high chemical integrity. For laboratory and pilot plant teams, following these standardized parameters is essential to replicate the success of the patented method.
- Dissolve 2-(2-aminothiazole-4-yl) ethyl acetate in dichloromethane, add benzyltriethylammonium bromide, and drip tert-butyl nitrite at room temperature to obtain the bromo-intermediate.
- Reduce the bromo-ester intermediate using sodium borohydride in ethanol at 0°C to room temperature to form the corresponding alcohol.
- Perform carbonyl insertion on the alcohol using a palladium catalyst and triethylamine under carbon monoxide atmosphere at 80°C to form the ethyl ester.
- React the hydroxyethyl thiazole ester with thionyl chloride in dichloromethane at 0°C to room temperature to finalize the chloroethyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain leaders, the adoption of this novel synthesis route offers tangible benefits that extend beyond mere chemical feasibility. The elimination of cryogenic cooling in the first step translates directly into reduced utility costs and lower capital expenditure on specialized refrigeration equipment. By shifting to room temperature operations, manufacturers can utilize standard reactor vessels without modification, increasing facility flexibility and throughput. Additionally, the use of readily available starting materials like 2-(2-aminothiazole-4-yl) ethyl acetate ensures a stable upstream supply chain, mitigating the risk of raw material shortages that often plague niche intermediate markets. These factors combine to create a more resilient supply network capable of meeting the growing demand for cardiovascular therapeutics.
- Cost Reduction in Manufacturing: The optimization of the bromination reagent serves as a primary driver for cost efficiency. By replacing the need for strict low-temperature control with a room temperature phase-transfer catalyzed reaction, the process significantly lowers energy consumption. Furthermore, the simplified workup procedures reduce the man-hours required for batch processing. The avoidance of expensive or hazardous reagents in favor of common chemicals like benzyltriethylammonium bromide and thionyl chloride further drives down the bill of materials. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, making the final API more affordable.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply continuity by minimizing the risk of batch failures due to temperature excursions. Conventional methods requiring -20°C are prone to deviations if cooling systems falter, whereas this ambient temperature process is far more forgiving. The use of standard solvents like dichloromethane and ethanol ensures that solvent availability is not a bottleneck. For reliable pharmaceutical intermediate suppliers, this reliability means they can commit to tighter delivery schedules and larger volume contracts with greater confidence, securing the production timelines for downstream drug manufacturers.
- Scalability and Environmental Compliance: The transition to milder reaction conditions inherently supports greener chemistry principles by reducing the energy footprint of the manufacturing process. The catalytic nature of the carbonylation step minimizes waste generation compared to stoichiometric oxidation methods. Moreover, the straightforward isolation techniques, such as extraction and filtration, simplify waste stream management. As regulatory bodies increasingly scrutinize the environmental impact of pharmaceutical manufacturing, adopting a process that is both energy-efficient and waste-minimized positions companies favorably for future compliance audits and sustainability certifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this thiazole intermediate. Understanding these details is crucial for stakeholders evaluating the integration of this technology into their existing supply chains. The answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the primary advantage of the new bromination method described in CN113461635A?
A: The novel method utilizes benzyltriethylammonium bromide as both a phase transfer catalyst and halogen donor, allowing the reaction to proceed at room temperature. This eliminates the need for strict cryogenic control below -20°C required by conventional methods, significantly simplifying operation and reducing energy consumption.
Q: Which catalyst is used for the critical carbonylation step?
A: The process employs [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium (Pd(dppf)Cl2) as the transition metal catalyst. This facilitates the coordination of carbon monoxide and subsequent insertion into the carbon-halogen bond to form the acyl metal compound efficiently.
Q: What is the therapeutic application of this intermediate?
A: This compound serves as a key intermediate in the synthesis of APJ receptor agonists. These agonists are being developed for treating cardiovascular diseases, specifically for improving contractility and ejection fraction in patients with chronic heart failure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 4-(2-chloroethyl)thiazole-2-carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving cardiovascular medications. Our technical team has extensively analyzed the route disclosed in CN113461635A and possesses the expertise to implement this advanced synthesis at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ethyl 4-(2-chloroethyl)thiazole-2-carboxylate meets the highest industry standards for API synthesis.
We invite you to collaborate with us to leverage this innovative technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments. Let us help you secure a stable, cost-effective supply of this vital intermediate, accelerating your journey from benchtop discovery to commercial market success.
