Scalable Visible-Light Synthesis of Aryl Acetylene Alkyl Sulfones for Pharmaceutical Intermediates
Scalable Visible-Light Synthesis of Aryl Acetylene Alkyl Sulfones for Pharmaceutical Intermediates
The landscape of organic synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, particularly in the construction of complex sulfone motifs essential for modern drug discovery. A groundbreaking approach detailed in patent CN110683971B introduces a novel visible-light-mediated strategy for synthesizing aryl acetylene alkyl sulfone compounds. This technology leverages the unique photophysical properties of the organic photosensitizer 2,4,5,6-tetrakis(9-carbazolyl)-isophthalonitrile, commonly known as 4CzIPN, to drive the reaction under exceptionally mild conditions. By utilizing Hantzsch esters as versatile alkyl radical precursors and solid sulfur dioxide substitutes, this method circumvents the need for harsh thermal conditions or toxic gaseous reagents. For R&D directors and process chemists seeking reliable pharmaceutical intermediate suppliers, this protocol represents a significant advancement in constructing sulfonyl fragments with high atom economy and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the introduction of sulfonyl groups into organic frameworks has been fraught with significant synthetic challenges that hinder efficient large-scale production. Conventional routes often rely on the use of gaseous sulfur dioxide, which poses severe safety hazards due to its toxicity and corrosive nature, requiring specialized equipment and rigorous safety protocols that inflate operational costs. Furthermore, many established methods depend heavily on transition metal catalysts, such as palladium or copper, which not only increase the raw material expense but also introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). The removal of these trace metals to meet stringent regulatory standards often necessitates additional purification steps, such as scavenging or recrystallization, which drastically reduce overall yield and extend production timelines. Additionally, traditional radical sulfonylation frequently requires high temperatures or strong oxidants, leading to poor functional group tolerance and the formation of complex impurity profiles that are difficult to separate.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a visible-light-driven organocatalytic system that operates at room temperature, effectively eliminating the thermal energy barriers associated with classical synthesis. The core of this innovation lies in the synergistic interaction between the excited state of the 4CzIPN photosensitizer and Hantzsch esters, which serve as a stable and easy-to-handle source of alkyl radicals. This approach allows for the in situ generation of alkylsulfonyl radicals from solid sulfur dioxide substitutes like sodium metabisulfite, thereby avoiding the handling of hazardous gases. The reaction proceeds through a radical addition mechanism to aryl acetylene bromides, offering a direct and modular route to diverse sulfone structures. As illustrated in the general reaction scheme below, this method accommodates a wide range of substrates, providing a robust platform for the rapid assembly of chemical libraries.
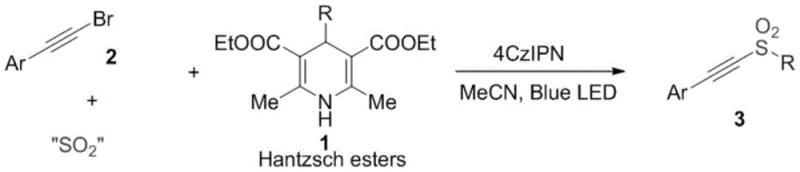
The versatility of this system is further evidenced by its compatibility with various aryl acetylene bromides bearing both electron-donating and electron-withdrawing substituents. For instance, the patent data highlights successful conversions with substrates containing ethyl, chloro, trifluoromethyl, and methyl groups, demonstrating broad substrate scope. Moreover, the use of different Hantzsch ester derivatives allows for the facile incorporation of cyclic alkyl groups, such as cyclopentyl and cyclohexyl moieties, into the final sulfone product. This modularity is crucial for medicinal chemists aiming to optimize the physicochemical properties of lead compounds without being constrained by synthetic limitations. The mildness of the reaction conditions ensures that sensitive functional groups remain intact, preserving the structural integrity of complex molecules throughout the synthesis.
Mechanistic Insights into 4CzIPN-Catalyzed Radical Sulfonylation
To fully appreciate the technical superiority of this process, one must delve into the intricate mechanistic pathway that governs the transformation. Upon irradiation with blue LED light, the 4CzIPN photosensitizer absorbs photons and transitions to an excited singlet state, which rapidly undergoes intersystem crossing to a long-lived triplet state. This excited photocatalyst possesses sufficient oxidative potential to abstract an electron from the Hantzsch ester, generating a radical cation intermediate. Subsequent fragmentation of this radical cation releases a proton and an alkyl radical, which is the key reactive species for the subsequent sulfonylation step. The alkyl radical then reacts with the sulfur dioxide surrogate to form an alkylsulfonyl radical. This transient species adds across the triple bond of the aryl acetylene bromide, followed by the elimination of a bromine radical to restore aromaticity and yield the final aryl acetylene alkyl sulfone product. The catalytic cycle is closed when the reduced form of the photocatalyst transfers an electron to the bromine radical or another intermediate, regenerating the ground state 4CzIPN for further turnover.
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic pathways. The mild reaction temperature minimizes thermal degradation and rearrangement side reactions that are common in high-heat processes. Furthermore, the selectivity of the radical addition to the alkyne bond is high, reducing the formation of regioisomers or over-sulfonylated byproducts. The use of solid sulfur dioxide sources ensures a controlled release of SO2 equivalents, preventing the accumulation of excess reagent that could lead to polysulfone formation. For quality assurance teams, this translates to a cleaner crude reaction profile, which significantly simplifies the downstream purification process. The ability to achieve high purity with minimal chromatographic effort is a critical factor in reducing the cost of goods sold (COGS) and accelerating the timeline from bench-scale discovery to commercial manufacturing.
How to Synthesize Aryl Acetylene Alkyl Sulfones Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory equipment, making it an attractive option for process development teams. The procedure typically involves charging a reaction vessel with the aryl acetylene bromide, a solid sulfur dioxide substitute such as sodium metabisulfite, the appropriate Hantzsch ester derivative, and a catalytic amount of 4CzIPN. The system is purged with an inert gas like nitrogen or argon to create an oxygen-free environment, which is essential for the success of radical reactions. Anhydrous acetonitrile is added as the solvent, and the mixture is stirred under blue LED irradiation at room temperature. Reaction monitoring via thin-layer chromatography (TLC) indicates completion typically within 48 hours, after which the solvent is removed under reduced pressure. The resulting residue is purified by flash column chromatography to afford the target sulfone compound in high yield.
- Combine aryl acetylene bromide, sulfur dioxide solid substitute (e.g., Na2S2O5), Hantzsch esters, and 4CzIPN photosensitizer in a reaction tube under inert atmosphere.
- Add anhydrous acetonitrile solvent and irradiate the mixture with blue LED light while stirring at room temperature for approximately 48 hours.
- Upon completion, concentrate the reaction mixture under reduced pressure and purify the crude product via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic methodology offers compelling economic and logistical benefits that directly impact the bottom line. The elimination of transition metal catalysts removes the need for expensive metal salts and the associated costs of metal scavenging resins or specialized filtration equipment. This reduction in material complexity leads to a streamlined supply chain where fewer specialized reagents need to be sourced and qualified. Furthermore, the use of solid sulfur dioxide substitutes enhances workplace safety and reduces the regulatory burden associated with storing and transporting hazardous compressed gases. These factors collectively contribute to a more resilient and cost-effective manufacturing process that is less susceptible to supply disruptions.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of inexpensive and commercially available starting materials. Hantzsch esters are readily synthesized from simple aldehydes and beta-keto esters, ensuring a stable and low-cost supply of the radical precursor. The photosensitizer 4CzIPN, while specialized, is used in very low catalytic loadings (typically 1-5 mol%), minimizing its impact on the overall material cost. Additionally, the ambient temperature operation eliminates the energy costs associated with heating or cooling large-scale reactors, leading to substantial utility savings. The simplified workup procedure, which often requires only concentration and chromatography, reduces labor hours and solvent consumption compared to multi-step traditional syntheses.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents rather than volatile gases or air-sensitive organometallics greatly improves the robustness of the supply chain. Solid sulfur dioxide surrogates like DABSO or sodium metabisulfite have long shelf lives and can be stored under standard conditions, reducing the risk of reagent degradation and batch-to-batch variability. This stability ensures consistent reaction performance and product quality, which is critical for maintaining continuous production schedules. Moreover, the broad substrate scope allows for the use of diverse aryl acetylene bromides, providing flexibility in sourcing raw materials and mitigating the risk of single-source dependency for specific building blocks.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but advancements in flow chemistry and LED reactor design have made this technology industrially viable. The mild conditions and lack of exothermic hazards make this process inherently safer to scale up from gram to kilogram quantities. From an environmental standpoint, the method aligns with green chemistry principles by avoiding toxic heavy metals and reducing waste generation. The high atom economy and selective nature of the reaction minimize the formation of hazardous byproducts, simplifying waste treatment and disposal. This environmental compatibility facilitates easier regulatory approval and supports corporate sustainability goals, making it an ideal choice for forward-thinking chemical manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this visible-light sulfonylation protocol. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for technical teams evaluating this methodology for their own pipelines. Understanding these nuances is essential for successful technology transfer and process optimization.
Q: What is the role of 4CzIPN in this synthesis?
A: 4CzIPN acts as a highly efficient organic photosensitizer that, upon excitation by visible light, oxidizes Hantzsch esters to generate the crucial alkyl radicals needed for the sulfonylation process.
Q: Can this method tolerate electron-withdrawing groups on the aryl ring?
A: Yes, the protocol demonstrates excellent tolerance for various substituents, including electron-withdrawing groups like fluorine, chlorine, and trifluoromethyl, as well as electron-donating alkyl groups.
Q: Is transition metal catalysis required for this transformation?
A: No, this method is distinctively metal-free, utilizing an organic photocatalyst (4CzIPN) instead of expensive and potentially toxic transition metals, which simplifies downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetylene Alkyl Sulfones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible-light photocatalysis in the synthesis of high-value pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic discoveries are seamlessly translated into robust industrial processes. We are committed to delivering high-purity aryl acetylene alkyl sulfones that meet the most stringent purity specifications required by global regulatory agencies. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee the quality and consistency of every batch we produce.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us help you accelerate your drug development pipeline with our reliable supply of complex sulfone intermediates.
