Advanced Rhodium-Catalyzed Pauson-Khand Route for Commercial Methyl Dihydrojasmonate Production
The global demand for high-quality fragrance ingredients continues to drive innovation in synthetic organic chemistry, particularly for staple compounds like methyl dihydrojasmonate. A significant technological breakthrough in this sector is detailed in patent CN107805201B, which discloses a highly efficient synthesis method leveraging homogeneous rhodium catalysis. This novel approach fundamentally shifts the paradigm from traditional aldol-based condensations to a streamlined Pauson-Khand reaction strategy. By utilizing inexpensive and readily available starting materials such as acetylene, ethylene, and 1-bromo-n-pentane, the process addresses critical bottlenecks associated with raw material sourcing and cost volatility. The integration of an organic nitrogen oxide promoter not only enhances the reaction yield of the key cyclization step but also drastically reduces the requisite loading of the precious rhodium catalyst. For industry stakeholders, this represents a pivotal advancement in cost reduction in flavors & fragrances manufacturing, offering a pathway to high-purity products with superior atom economy and minimized environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of methyl dihydrojasmonate has relied heavily on routes originating from cyclopentanone and n-valeraldehyde. While these pathways are chemically established, they suffer from inherent economic and logistical vulnerabilities that impact the reliable synthetic flavors & fragrances supplier landscape. The primary precursors, cyclopentanone and n-valeraldehyde, are subject to significant price fluctuations and supply chain constraints due to their dependence on specific petrochemical streams. Furthermore, the traditional synthesis involves multiple steps including aldol condensation and strong acid isomerization, which often require harsh reaction conditions and generate substantial amounts of acidic waste. The reliance on these specific aldehydes limits the scalability of production, as any disruption in the supply of n-valeraldehyde can halt the entire manufacturing line. Additionally, the atom economy of these older routes is suboptimal, leading to higher waste disposal costs and a larger environmental burden, which is increasingly scrutinized under modern regulatory frameworks.
The Novel Approach
In stark contrast, the methodology outlined in patent CN107805201B introduces a robust alternative that bypasses the need for expensive aldehydes entirely. By employing a Pauson-Khand reaction between 1-heptyne and ethylene, the process constructs the critical 2-pentyl-2-cyclopentenone skeleton in a single, highly efficient step. This transformation is catalyzed by a homogeneous rhodium system in the presence of an organic nitrogen oxide, operating under supercritical ethylene conditions. This innovative strategy not only simplifies the synthetic sequence but also leverages ethylene as both a reactant and a solvent, thereby eliminating the need for additional volatile organic compounds. The result is a process with exceptional atom economy and a significantly reduced operational complexity. For procurement teams, this translates to a decoupling from the volatile aldehyde market, securing a more stable and predictable cost structure for commercial scale-up of complex fragrance intermediates.
Mechanistic Insights into Rhodium-Catalyzed Pauson-Khand Cyclization
The core of this technological advancement lies in the sophisticated catalytic cycle driven by the dirhodium tetracarbonyl dichloride complex, [Rh(CO)2Cl]2. In a standard Pauson-Khand reaction, the coordination of the alkyne and alkene to the metal center is followed by CO insertion and reductive elimination to form the cyclopentenone ring. However, the efficiency of this cycle is often limited by the stability of the low-valent rhodium species. The patent highlights the critical role of the organic nitrogen oxide promoter, specifically N-methylmorpholine N-oxide (NMO), in overcoming this limitation. NMO functions as a mild oxidant that effectively regenerates the active high-valent rhodium species from the reduced state formed during the catalytic turnover. This oxidative promotion ensures that the catalyst remains active for a prolonged period, preventing premature deactivation and allowing for extremely high substrate-to-catalyst ratios.
Furthermore, the reaction environment plays a pivotal role in the mechanism's success. The use of supercritical ethylene creates a unique phase where the alkene substrate acts as the reaction medium. This eliminates mass transfer limitations typically associated with gas-liquid reactions, ensuring that the concentration of ethylene at the catalytic center remains saturating throughout the process. The presence of carbon monoxide at controlled partial pressures facilitates the necessary carbonylation step without inhibiting the initial coordination of the alkene. This precise balance of pressure and temperature, typically around 5.0 to 6.0 MPa and 80 to 90°C, optimizes the kinetic profile of the reaction. The outcome is a highly selective formation of the 2-pentyl-2-cyclopentenone intermediate with minimal byproduct formation, ensuring that downstream purification is straightforward and efficient.
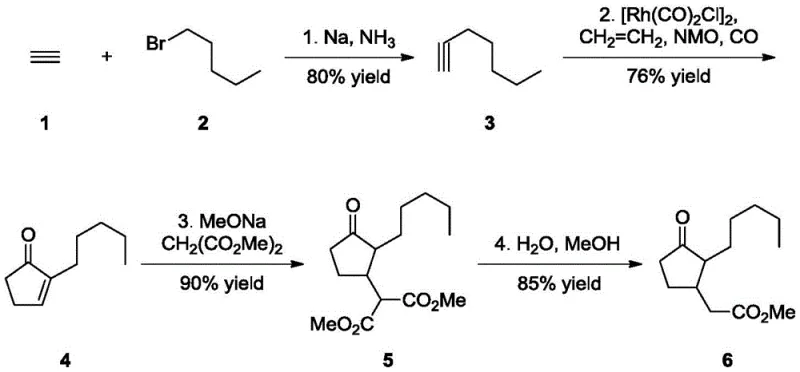
How to Synthesize Methyl Dihydrojasmonate Efficiently
The synthesis protocol described in the patent provides a clear roadmap for replicating this high-efficiency process in a pilot or production setting. The procedure begins with the generation of the alkyne precursor, followed by the key cyclization, Michael addition, and final decarboxylation. Each step is optimized for yield and purity, utilizing standard chemical engineering unit operations that are easily adaptable to existing infrastructure. The detailed standardized synthesis steps below outline the specific conditions and reagent ratios required to achieve the reported performance metrics, serving as a foundational guide for process engineers aiming to implement this technology.
- Synthesize 1-heptyne by reacting acetylene with 1-bromo-n-pentane in liquid ammonia using sodium metal as a promoter at cryogenic temperatures.
- Perform the Pauson-Khand reaction on 1-heptyne with ethylene and carbon monoxide using a dirhodium tetracarbonyl dichloride catalyst and N-methylmorpholine N-oxide promoter in supercritical ethylene.
- Conduct a Michael addition of dimethyl malonate to the resulting 2-pentyl-2-cyclopentenone intermediate using sodium methoxide as a base.
- Complete the synthesis by heating the addition product in a mixture of water and methanol to induce decarboxylation, yielding high-purity methyl dihydrojasmonate.
Commercial Advantages for Procurement and Supply Chain Teams
For decision-makers focused on the bottom line and operational resilience, the shift to this rhodium-catalyzed route offers compelling strategic advantages. The most immediate impact is seen in the raw material cost structure. By replacing specialized aldehydes with commodity gases like ethylene and acetylene, manufacturers can insulate themselves from the price volatility inherent in the fine chemical aldehyde market. Ethylene is one of the most produced organic chemicals globally, ensuring a virtually unlimited supply at competitive prices. This fundamental change in the bill of materials drives a substantial reduction in the variable cost of goods sold, enhancing overall margin potential without compromising product quality.
- Cost Reduction in Manufacturing: The implementation of the NMO-promoted rhodium catalyst system allows for a dramatic decrease in catalyst loading. With substrate-to-catalyst ratios reaching up to 10,000:1, the consumption of expensive rhodium metal is minimized to trace levels. This efficiency eliminates the need for complex and costly metal scavenging processes that are typically required to meet heavy metal specifications in fragrance applications. Consequently, the overall processing cost is significantly lowered, as the expense associated with precious metal recovery and waste treatment is drastically curtailed, leading to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Diversifying the raw material base from niche aldehydes to bulk petrochemicals significantly de-risks the supply chain. Acetylene, ethylene, and 1-bromo-n-pentane are widely available from multiple global suppliers, reducing the dependency on single-source vendors. This abundance ensures continuity of supply even during regional disruptions or maintenance shutdowns at specific chemical plants. Furthermore, the simplified synthetic route reduces the number of intermediate storage requirements and handling steps, streamlining logistics and reducing the lead time for high-purity fragrance intermediates from order to delivery.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing supercritical fluid technology which is well-suited for large-scale continuous or batch processing. The elimination of organic solvents in the key cyclization step reduces the facility's VOC (Volatile Organic Compound) emissions, simplifying compliance with stringent environmental regulations. The high atom economy means less waste is generated per kilogram of product, lowering disposal costs and aligning with corporate sustainability goals. This green chemistry profile not only reduces operational overhead but also enhances the brand value of the final fragrance product in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and process descriptions found in the source documentation, providing clarity on the feasibility and benefits of adopting this technology for industrial production.
Q: How does the new rhodium-catalyzed route improve upon traditional aldol condensation methods?
A: Traditional methods rely on cyclopentanone and n-valeraldehyde, which are expensive and supply-constrained. The new route utilizes commodity chemicals like acetylene and ethylene, significantly lowering raw material costs and improving atom economy through a direct Pauson-Khand cyclization.
Q: What is the role of N-methylmorpholine N-oxide (NMO) in this catalytic system?
A: NMO acts as a crucial co-catalyst or promoter that effectively oxidizes the rhodium species to a higher valence state. This regeneration of the active catalytic species allows for a drastic reduction in rhodium loading, achieving substrate-to-catalyst ratios as high as 10,000:1, which is vital for cost-effective industrial production.
Q: Is this process suitable for large-scale manufacturing regarding safety and waste?
A: Yes, the process is designed for scalability. It utilizes supercritical ethylene as both reactant and solvent, eliminating the need for volatile organic solvents in the key cyclization step. Furthermore, the high atom economy and simplified workup procedures minimize three-waste generation, aligning with modern green chemistry standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl Dihydrojasmonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the rhodium-mediated Pauson-Khand reaction for the fragrance industry. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate such complex laboratory methodologies into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot to plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of methyl dihydrojasmonate meets the exacting standards required by top-tier flavor and fragrance houses.
We invite you to explore how this innovative synthesis route can optimize your supply chain and reduce costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals. Let us partner with you to secure a sustainable and cost-effective supply of high-quality fragrance ingredients.
