Advanced Synthesis of 4-Bromo-4'-Propylbiphenyl for High-Performance Liquid Crystal Applications
The pharmaceutical and electronic materials industries are constantly seeking robust synthetic pathways for critical intermediates that balance high purity with operational safety. Patent CN108129258B introduces a transformative synthesis process for 4-bromo-4'-propylbiphenyl, a pivotal building block in the manufacture of liquid crystal monomers and advanced pharmaceutical frameworks. This innovation addresses the longstanding challenges associated with traditional manufacturing methods, which often rely on hazardous reagents and extreme thermal conditions that complicate industrial scale-up. By leveraging a strategic sequence of Friedel-Crafts acylation followed by mild reduction and catalytic hydrogenation, this technology offers a viable pathway for producing high-purity 4-bromo-4'-propylbiphenyl with significantly improved efficiency. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable liquid crystal intermediate supplier capable of meeting stringent quality standards while optimizing production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biphenyl derivatives like 4-bromo-4'-propylbiphenyl has been plagued by inefficient routes that pose severe safety and economic burdens. One prevalent method involves a Friedel-Crafts acylation followed by a Huang Minlon reduction, as illustrated in the reaction scheme below. This legacy approach necessitates ultra-high temperature reaction conditions ranging from 150°C to 200°C, creating substantial energy demands and requiring specialized, high-pressure equipment that increases capital expenditure. Furthermore, the use of hydrazine hydrate in the reduction step introduces significant toxicity concerns, mandating rigorous containment protocols that slow down production throughput. Another common pathway utilizes halogen-lithium exchange reactions, which demand cryogenic conditions below -78°C. Maintaining such low temperatures on a commercial scale is energetically prohibitive and technically challenging, often leading to inconsistent batch quality and limited scalability for complex biphenyl derivatives.
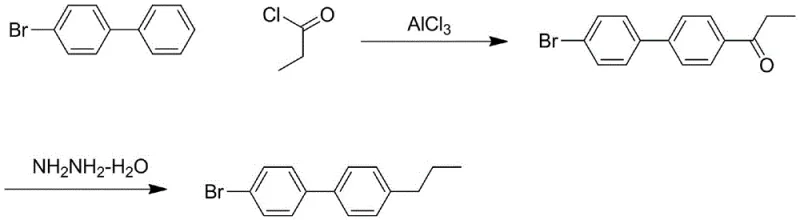
The Novel Approach
In stark contrast to these cumbersome legacy methods, the patented process outlined in CN108129258B employs a sophisticated five-step sequence that operates under remarkably mild conditions. The strategy initiates with the acylation of inexpensive biphenyl, followed by a controlled reduction using sodium borohydride, which avoids the need for hazardous hydrazine derivatives. A key innovation lies in the dehydration step catalyzed by p-toluenesulfonic acid, which proceeds efficiently at moderate temperatures of 80-90°C, eliminating the need for the extreme heat required by Huang Minlon reductions. The subsequent catalytic hydrogenation and final bromination steps are conducted at near-ambient temperatures, drastically reducing energy consumption and safety risks. This streamlined approach not only enhances operator safety by removing carcinogenic compounds like iodopropane but also simplifies post-treatment procedures, making it an ideal solution for cost reduction in electronic chemical manufacturing where margin optimization is critical.
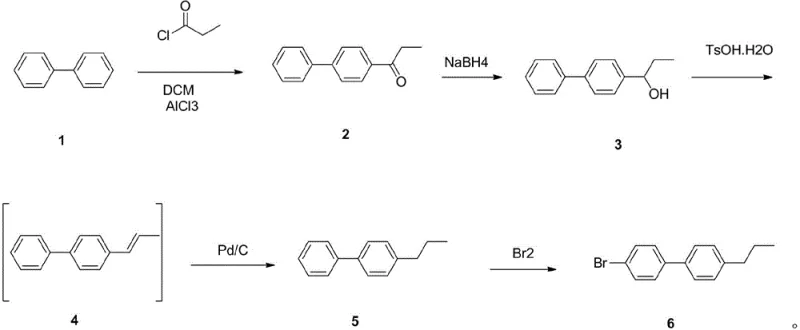
Mechanistic Insights into FeCl3-Catalyzed Bromination and Hydrogenation
The core of this synthetic breakthrough lies in its precise control over reaction kinetics and impurity profiles, particularly during the final functionalization stages. The process utilizes ferric chloride as a Lewis acid catalyst for the bromination of 4-propyl-biphenyl, ensuring high regioselectivity for the para-position. By maintaining the reaction temperature strictly between 20°C and 30°C, the system minimizes the formation of poly-brominated byproducts and ortho-substituted impurities that often plague less controlled electrophilic aromatic substitutions. This thermal moderation allows for a cleaner reaction profile, which directly translates to simplified purification workflows and higher overall recovery rates. For R&D teams focused on purity specifications, this mechanistic control is paramount, as it ensures that the final API intermediate or liquid crystal precursor meets the rigorous spectral purity required for downstream applications without extensive chromatographic separation.
Furthermore, the hydrogenation step employing palladium carbon demonstrates exceptional chemoselectivity, reducing the alkene intermediate to the corresponding alkane without affecting the aromatic rings or the bromine substituent in later stages. The protocol specifies a temperature range of 35-45°C, which is sufficient to drive the reaction to completion while preventing catalyst deactivation or unwanted side reactions. This careful balancing of catalytic activity and thermal input results in a total yield reaching 57%, a substantial improvement over the 20-30% yields typical of prior art. The ability to telescope certain steps, such as using the organic phase from the dehydration reaction directly in hydrogenation, further reduces solvent waste and handling time. This level of process intensification is crucial for achieving commercial scale-up of complex biphenyl derivatives, ensuring that the manufacturing process remains both economically viable and environmentally compliant.
How to Synthesize 4-Bromo-4'-Propylbiphenyl Efficiently
The implementation of this synthesis route requires strict adherence to the specified stoichiometric ratios and thermal profiles to maximize the 57% total yield reported in the patent data. The process begins with the activation of biphenyl using propionyl chloride and aluminum chloride, where temperature control at 5-10°C is critical to prevent over-acylation. Subsequent reduction with sodium borohydride must be managed carefully to ensure complete conversion to the alcohol intermediate before proceeding to the acid-catalyzed elimination. The detailed standardized synthesis steps below outline the precise operational parameters required to replicate this high-efficiency pathway in a GMP-compliant environment.
- Perform Friedel-Crafts acylation on biphenyl using propionyl chloride and aluminum chloride at 5-10°C to form 4-propionyl-biphenyl.
- Reduce the ketone intermediate using sodium borohydride in tetrahydrofuran at 10-15°C to obtain the alcohol derivative.
- Execute acid-catalyzed dehydration using p-toluenesulfonic acid at 80-90°C to generate the alkene intermediate without isolation.
- Conduct catalytic hydrogenation using palladium carbon at 35-45°C to saturate the double bond and form 4-propyl-biphenyl.
- Finalize the synthesis via electrophilic aromatic substitution using liquid bromine and ferric chloride at 20-30°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers profound strategic advantages beyond mere technical feasibility. By eliminating the need for ultra-low temperature infrastructure and hazardous reagents like iodopropane, manufacturers can significantly reduce capital expenditure on specialized equipment and safety containment systems. The shift to milder reaction conditions also lowers operational energy costs, contributing to substantial cost savings in the long-term production lifecycle. Moreover, the use of cheap and easily available biphenyl as a starting raw material stabilizes the supply chain against volatility in precursor pricing, ensuring consistent availability of high-purity 4-bromo-4'-propylbiphenyl. This reliability is essential for maintaining continuous production schedules in the fast-paced electronics and pharmaceutical sectors.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as iodopropane and hydrazine hydrate directly lowers raw material costs and waste disposal fees. Additionally, the avoidance of energy-intensive cryogenic or ultra-high temperature processes reduces utility consumption, leading to a more lean and cost-effective manufacturing operation. The improved total yield of 57% means less raw material is wasted per unit of product, further enhancing the economic efficiency of the supply chain.
- Enhanced Supply Chain Reliability: Utilizing biphenyl, a commodity chemical with stable global availability, mitigates the risk of supply disruptions associated with specialized or regulated precursors. The simplified post-treatment and easier control of synthesis process conditions allow for faster batch turnover, effectively reducing lead time for high-purity liquid crystal monomers. This agility enables suppliers to respond more rapidly to fluctuating market demands without compromising on quality or delivery commitments.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of carcinogenic compounds simplify the regulatory approval process for new manufacturing plants. The process generates less hazardous waste, aligning with increasingly stringent environmental regulations and sustainability goals. This ease of scale-up ensures that production can be expanded from pilot batches to multi-ton commercial volumes without encountering the technical bottlenecks typical of more complex synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the comparative data and beneficial effects outlined in the patent documentation, providing clarity on how this method outperforms traditional approaches in terms of safety, yield, and operational simplicity.
Q: Why is the new synthesis route safer than the Huang Minlon reduction method?
A: The conventional Huang Minlon reduction requires ultra-high temperatures of 150-200°C and uses hydrazine hydrate, posing significant safety hazards. The new process operates under mild conditions below 90°C and eliminates carcinogenic reagents like iodopropane.
Q: How does this process improve atom economy compared to halogen-lithium exchange?
A: Traditional halogen-lithium exchange routes suffer from low atom economy due to the replacement of heavy bromine and iodine atoms, yielding only 20-30%. The patented method achieves a total yield of 57% by utilizing efficient acylation and hydrogenation steps.
Q: What are the critical temperature controls for the acylation step?
A: Precise temperature control is vital during the addition of aluminum chloride. The reaction must be cooled to 5-10°C before catalyst addition and maintained carefully to prevent side reactions, ensuring high purity of the 4-propionyl-biphenyl intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Bromo-4'-Propylbiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering 4-bromo-4'-propylbiphenyl with stringent purity specifications, supported by our rigorous QC labs that validate every batch against the highest industry standards. Our capability to implement advanced synthetic routes like the one described in CN108129258B demonstrates our dedication to technological leadership and customer-centric solutions in the fine chemical sector.
We invite you to collaborate with us to optimize your supply chain and achieve your cost targets without compromising on quality. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and manufacturing goals, ensuring a partnership built on transparency, reliability, and scientific excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
