Advanced Synthesis of Amino-Substituted Dibenzothiophene Sulfoxides for Next-Gen OLED Materials
Advanced Synthesis of Amino-Substituted Dibenzothiophene Sulfoxides for Next-Gen OLED Materials
The rapid evolution of organic photoelectric materials has placed a premium on compounds that offer superior charge transfer properties and tunable emission wavelengths. Patent CN114890981A introduces a groundbreaking preparation method for a class of amino-substituted dibenzothiophene sulfoxide derivatives, addressing critical bottlenecks in the synthesis of high-performance organic room temperature phosphorescent (RTP) materials. This technology leverages a sophisticated Donor-Acceptor-Donor molecular architecture, where the dibenzothiophene sulfoxide core acts as a potent electron acceptor flanked by strong electron-donating triphenylamine groups. For R&D directors and procurement specialists in the electronic chemicals sector, this patent represents a significant leap forward, offering a synthetic route that is not only chemically elegant but also commercially viable for the mass production of next-generation display and encryption materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex heterocyclic sulfoxides has been plagued by issues related to regioselectivity and over-oxidation. Conventional oxidative protocols often rely on harsh reagents or elevated temperatures that lack the finesse required to stop the reaction precisely at the sulfoxide stage, frequently leading to the formation of thermodynamically stable sulfone impurities. These impurities are notoriously difficult to separate due to their similar polarity and solubility profiles, necessitating multiple rounds of expensive and time-consuming chromatographic purification. Furthermore, older coupling strategies for attaching bulky amine donors to the dibenzothiophene core often suffered from low catalytic turnover, requiring excessive loading of precious metal catalysts and resulting in residual palladium contamination that is unacceptable for high-purity electronic applications. The cumulative effect of these inefficiencies is a dramatic increase in manufacturing costs and extended lead times, creating a fragile supply chain for critical optoelectronic intermediates.
The Novel Approach
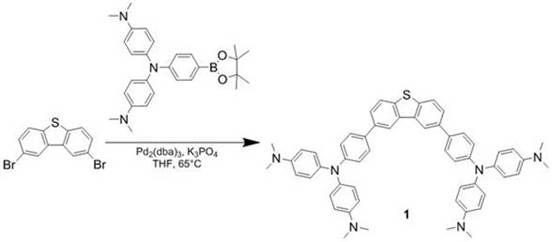
The methodology outlined in the patent data circumvents these historical challenges through a meticulously optimized two-step sequence involving Suzuki-Miyaura coupling followed by mild, selective oxidation. By utilizing tris(dibenzylideneacetone)dipalladium as the catalyst system in conjunction with potassium phosphate in tetrahydrofuran, the initial C-N bond formation proceeds efficiently at a moderate temperature of 65°C. This温和 condition preserves the integrity of the sensitive amine functionalities while ensuring high conversion rates. Subsequently, the oxidation step employs a mixture of 30% hydrogen peroxide and trifluoroacetic acid in dichloromethane at room temperature. This specific reagent combination provides the exact oxidative potential needed to convert the sulfide to the sulfoxide without pushing the reaction toward the sulfone. The result is a streamlined process that yields high-purity products with simplified workup procedures, such as straightforward extraction and recrystallization, rather than complex multi-stage purifications.
Mechanistic Insights into Pd-Catalyzed Coupling and Selective Oxidation
The success of this synthetic strategy lies in the precise control of the catalytic cycle and the oxidative environment. In the first stage, the palladium catalyst undergoes oxidative addition with the 2,8-dibromodibenzothiophene substrate, forming a reactive organopalladium intermediate. The presence of the bulky triphenylamine-based boronic ester requires a robust ligand environment, which the dba ligands provide, facilitating the subsequent transmetallation step with the boron species. The base, potassium phosphate, plays a dual role in activating the boronic ester and neutralizing the acidic byproducts, driving the equilibrium toward the coupled product. This mechanism ensures that even with sterically hindered substrates, the coupling efficiency remains high, as evidenced by the reported yields ranging from 64% to 75% for the intermediate sulfides. The rigorous exclusion of oxygen and moisture during this phase is critical to preventing catalyst deactivation and homocoupling side reactions.
Following the coupling, the transition to the sulfoxide is governed by the electrophilic nature of the activated peroxide species generated in situ by the interaction of hydrogen peroxide and trifluoroacetic acid. The sulfur atom in the dibenzothiophene ring is nucleophilic enough to attack this activated oxygen source, forming the S-O bond. Crucially, the reaction conditions are tuned such that the electron-withdrawing effect of the newly formed sulfoxide group deactivates the sulfur center towards further oxidation. This electronic deactivation acts as a self-limiting mechanism, effectively preventing the formation of the sulfone byproduct. For quality control teams, this means the impurity profile is significantly cleaner, with the primary contaminants being unreacted starting materials rather than structurally similar over-oxidized analogs, thereby simplifying the analytical validation and release testing processes for the final high-purity OLED material.
How to Synthesize Amino-Substituted Dibenzothiophene Sulfoxide Efficiently
The practical implementation of this synthesis requires strict adherence to the optimized parameters regarding stoichiometry, temperature, and solvent quality to maximize yield and purity. The process begins with the assembly of the carbon-nitrogen framework under inert atmosphere, followed by a controlled oxidative workup that capitalizes on the differential solubility of the products. While the general workflow is robust, attention to detail in the purification stages—specifically the choice of eluent ratios for column chromatography and solvent pairs for recrystallization—is paramount for achieving the electronic-grade purity required for device fabrication. The detailed standardized synthesis steps, including specific reagent quantities and isolation protocols validated in the patent examples, are provided below to guide your process development team.
- Perform Suzuki-Miyaura coupling between 2,8-dibromodibenzothiophene and substituted phenylboronic acid pinacol esters using Pd2(dba)3 catalyst in THF at 65°C.
- Isolate the intermediate sulfide derivative via solvent extraction and silica gel column chromatography to ensure high purity before oxidation.
- Execute selective oxidation of the sulfur atom using 30% hydrogen peroxide and trifluoroacetic acid in dichloromethane at room temperature to yield the final sulfoxide.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented methodology offers substantial benefits that extend beyond mere chemical yield. The reliance on commodity chemicals such as tetrahydrofuran, dichloromethane, and potassium phosphate reduces the dependency on exotic or single-source reagents, thereby mitigating supply chain risks associated with raw material shortages. The moderate reaction temperatures eliminate the need for energy-intensive heating or cryogenic cooling systems, directly translating to lower utility costs per kilogram of produced material. Furthermore, the simplicity of the isolation protocol, which avoids complex distillation or specialized filtration equipment, allows for faster batch turnover times in multipurpose reactors. This operational efficiency enables manufacturers to respond more agilely to fluctuating market demands for organic photoelectric materials without compromising on product quality or delivery schedules.
- Cost Reduction in Manufacturing: The elimination of harsh oxidants and the reduction in catalyst loading significantly lower the direct material costs associated with production. By avoiding the formation of difficult-to-remove sulfone impurities, the process drastically reduces the consumption of silica gel and organic solvents typically required for extensive purification, leading to substantial cost savings in waste disposal and solvent recovery operations. Additionally, the high selectivity of the reaction minimizes the loss of valuable intermediates, improving the overall atom economy and reducing the effective cost per gram of the final high-purity OLED material.
- Enhanced Supply Chain Reliability: The synthetic route utilizes widely available starting materials, including various substituted boronic acid pinacol esters and dibromodibenzothiophene, which are stocked by multiple global suppliers. This diversification of the supply base ensures continuity of supply even in the face of regional disruptions or logistics bottlenecks. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the frequency of batch failures and ensuring a consistent flow of product to downstream customers who rely on just-in-time delivery models for their own manufacturing lines.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is facilitated by the use of standard unit operations such as liquid-liquid extraction and crystallization, which are easily scalable in stirred tank reactors. The avoidance of heavy metal oxidants like chromium or manganese salts aligns with increasingly stringent environmental regulations regarding hazardous waste generation. The aqueous waste streams generated are primarily saline and acidic, which can be neutralized and treated using conventional wastewater treatment infrastructure, simplifying the permitting process for new production facilities and enhancing the overall sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced sulfoxide derivatives. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring that the information reflects the actual capabilities and limitations of the described technology. Understanding these nuances is essential for integrating these materials into your specific product development pipelines.
Q: What is the primary advantage of the oxidation step in this patent?
A: The use of hydrogen peroxide with trifluoroacetic acid at room temperature allows for highly selective oxidation of the sulfur atom to the sulfoxide state, minimizing the formation of over-oxidized sulfone byproducts which are common in harsher oxidative conditions.
Q: Why is the Donor-Acceptor-Donor structure significant for these derivatives?
A: This architecture enhances intramolecular charge transfer properties, resulting in longer wavelength emission and improved efficiency in organic room temperature phosphorescence (RTP), which is critical for advanced bio-imaging and encryption applications.
Q: Is this synthesis route suitable for large-scale production?
A: Yes, the process utilizes commercially available raw materials, mild reaction temperatures (65°C for coupling, room temperature for oxidation), and standard purification techniques like recrystallization, making it highly amenable to kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amino-Substituted Dibenzothiophene Sulfoxide Supplier
As the demand for high-performance organic photoelectric materials continues to surge, having a manufacturing partner with deep technical expertise is crucial for maintaining a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pilot phase or full-scale manufacturing. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify the absence of critical impurities such as residual palladium and over-oxidized byproducts, guaranteeing material consistency for your OLED or encryption applications.
We invite you to leverage our technical capabilities to optimize your supply chain and reduce your overall cost of goods. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of this patented synthesis can accelerate your time-to-market and enhance the performance of your final electronic devices.
