Advanced One-Pot Synthesis Strategy for 3-Methylsulfonyl Nitrogen Heterocyclic Compounds
Introduction to Next-Generation Sulfonyl Heterocycle Synthesis
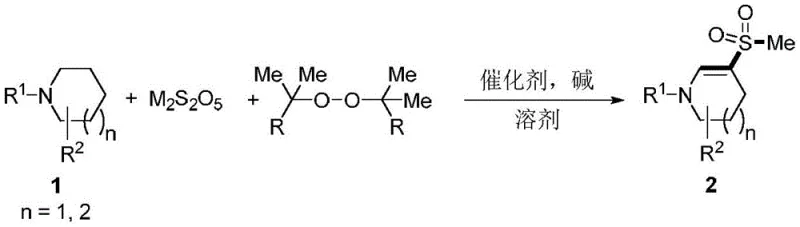
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical development, particularly in the construction of complex nitrogen-containing scaffolds. A significant breakthrough in this domain is detailed in patent CN112279801B, which discloses a highly efficient method for synthesizing 3-methylsulfonyl substituted nitrogen heterocyclic compounds. This technology represents a paradigm shift from traditional multi-step procedures to a streamlined one-pot multi-component series reaction. By leveraging readily available saturated cyclic amines, pyrosulfites, and methyl-containing peroxides, this novel approach achieves the direct installation of the mesyl group onto the heterocyclic core under mild oxidative conditions. For R&D directors and process chemists, this innovation offers a robust pathway to access valuable intermediates that are critical for the development of bioactive molecules, addressing long-standing challenges regarding operational complexity and raw material accessibility in the fine chemical sector.
The strategic importance of mesyl-substituted nitrogen heterocycles cannot be overstated, as these structural motifs serve as pivotal building blocks in the design of numerous therapeutic agents. The ability to introduce a sulfonyl group efficiently opens up avenues for further functionalization and optimization of biological activity. The methodology described in the patent utilizes an air atmosphere and moderate temperatures ranging from 100-140°C, which significantly lowers the energy barrier compared to harsh conventional methods. This technical advancement not only simplifies the synthetic route but also enhances the overall safety profile of the manufacturing process by avoiding the use of hazardous thiol-based reagents often required in older protocols. As a reliable pharmaceutical intermediate supplier, understanding and adopting such cutting-edge synthetic methodologies is essential for maintaining a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of mesyl-substituted nitrogen heterocyclic compounds has been fraught with significant technical and economic hurdles that impede efficient large-scale production. Traditional routes typically rely on the nucleophilic substitution reaction between substituted nitrogen heterocycles and sodium methyl mercaptide, a reagent that is not only difficult to obtain but also poses severe handling risks due to its toxicity and unpleasant odor. Furthermore, these legacy methods often necessitate multiple synthetic steps, requiring the isolation and purification of unstable intermediates, which drastically reduces the overall yield and increases the consumption of solvents and energy. The reliance on specialized sulfur sources also introduces variability in the supply chain, creating bottlenecks for procurement managers who struggle to secure consistent quality raw materials at reasonable costs. Additionally, the generation of sulfur-containing waste streams in these conventional processes complicates environmental compliance and waste treatment, adding hidden costs to the final product.
The Novel Approach
In stark contrast, the innovative technique outlined in the patent data revolutionizes this chemical transformation by employing a direct, one-pot strategy that bypasses the need for pre-functionalized sulfur reagents. By utilizing sodium metabisulfite—a cheap, stable, and widely available industrial chemical—alongside common organic peroxides like dicumyl peroxide, the process achieves the desired sulfonation in a single operational step. This approach eliminates the necessity for isolating intermediates, thereby reducing the total processing time and minimizing the exposure of personnel to hazardous substances. The reaction proceeds smoothly in the presence of a simple iron catalyst and a base, demonstrating remarkable tolerance to a wide array of functional groups on the amine substrate. This versatility allows for the rapid generation of diverse compound libraries, accelerating the drug discovery timeline. For manufacturers seeking cost reduction in pharmaceutical intermediate manufacturing, this transition from multi-step thiol chemistry to a direct oxidative sulfonation represents a substantial opportunity to optimize margins and improve process sustainability.
Mechanistic Insights into Iron-Catalyzed Oxidative Sulfonation
The core of this synthetic breakthrough lies in the intricate interplay between the iron catalyst, the peroxide oxidant, and the sulfur source, which facilitates a radical-mediated transformation. Mechanistically, the ferric chloride catalyst likely promotes the homolytic cleavage of the O-O bond in the methyl-containing peroxide, generating reactive methyl radicals or alkoxy radicals that initiate the cascade. Simultaneously, the pyrosulfite species undergoes oxidation and fragmentation to provide the necessary sulfur equivalents for the formation of the sulfonyl moiety. The reaction environment, maintained under an air atmosphere at elevated temperatures (100-140°C), ensures sufficient thermal energy to drive the radical propagation steps while allowing molecular oxygen to potentially participate in the re-oxidation of the catalyst or the sulfur intermediates. This catalytic cycle is highly efficient, requiring only low loadings of the iron salt (0.2-1 equivalent relative to substrate in optimized examples), which minimizes metal contamination in the final product. Understanding this mechanism is crucial for process chemists aiming to fine-tune reaction parameters for specific substrates, ensuring high conversion rates and minimizing side reactions.
Furthermore, the compatibility of this system with various bases such as DABCO, quinoline, or 1,10-phenanthroline suggests a nuanced role for the base in stabilizing intermediates or facilitating proton transfer steps during the cyclization or functionalization process. The broad substrate scope demonstrated in the patent examples indicates that the electronic nature of the substituents on the nitrogen atom—whether electron-withdrawing groups like cyano and nitro or electron-donating groups like methoxy and methyl—does not significantly inhibit the reaction efficiency. This robustness implies that the rate-determining step is likely independent of the electronic density on the aromatic ring attached to the nitrogen, pointing towards a mechanism dominated by the activation of the sulfur and peroxide species. Such mechanistic resilience is a key attribute for commercial scale-up of complex pharmaceutical intermediates, as it ensures consistent performance across different batches and substrate variations, reducing the risk of failed runs in a production setting.
How to Synthesize 3-Methylsulfonyl Nitrogen Heterocycles Efficiently
Implementing this advanced synthetic route requires careful attention to reagent stoichiometry and reaction conditions to maximize yield and purity. The standard protocol involves dissolving the saturated cyclic amine, sodium metabisulfite, and the peroxide oxidant in acetonitrile, followed by the addition of the catalyst and base. The mixture is then heated under air, allowing the multi-component reaction to proceed to completion. Detailed operational guidelines, including specific molar ratios and workup procedures derived from the patent examples, are essential for reproducing the high yields reported in the literature. For instance, optimizing the ratio of metabisulfite to amine between 2:1 and 5:1 can significantly influence the conversion efficiency. To assist technical teams in adopting this methodology, we have compiled a standardized operational guide below.
- Dissolve saturated cyclic amine compound, sodium metabisulfite, and dicumyl peroxide (DCP) in acetonitrile solvent within a reaction vessel.
- Add ferric chloride catalyst and a base such as DABCO to the mixture, then heat the system to 100-140°C under an air atmosphere.
- After stirring for approximately 2 hours, quench the reaction with saturated sodium chloride solution, extract with dichloromethane, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this one-pot synthesis method offers transformative benefits for procurement and supply chain management within the fine chemical industry. The primary advantage stems from the drastic simplification of the raw material portfolio; replacing expensive and hard-to-source sodium methyl mercaptide with commodity chemicals like sodium metabisulfite and dicumyl peroxide leads to immediate and significant cost savings. This shift not only lowers the direct material costs but also mitigates supply chain risks associated with sourcing specialized reagents from limited vendors. Moreover, the elimination of intermediate isolation steps reduces the consumption of solvents and the labor hours required for purification, contributing to a leaner and more agile manufacturing process. These efficiencies translate into a more competitive pricing structure for the final intermediates, allowing downstream partners to optimize their own production budgets without compromising on quality.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, driven by the replacement of high-cost thiol reagents with inexpensive inorganic sulfites and the reduction of unit operations. By consolidating multiple reaction steps into a single pot, manufacturers can significantly lower energy consumption and solvent usage, which are major cost drivers in chemical production. The use of earth-abundant iron catalysts further avoids the expense associated with precious metal catalysts like palladium or platinum, removing the need for costly metal scavenging steps to meet strict residual metal specifications. This holistic reduction in operational expenditure ensures that the production of high-purity pharmaceutical intermediates becomes more economically viable, supporting margin expansion for suppliers and cost stability for buyers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on globally available bulk chemicals that are not subject to the same geopolitical or logistical constraints as specialty fine chemicals. Sodium metabisulfite and common organic peroxides are produced at massive scales for various industries, ensuring a steady and reliable flow of materials even during market fluctuations. This availability reduces the lead time for raw material procurement and minimizes the risk of production stoppages due to supply shortages. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, further enhancing the reliability of the supply chain. For supply chain heads, this translates to greater predictability in delivery schedules and the ability to maintain safety stock levels more effectively.
- Scalability and Environmental Compliance: The scalability of this process is inherently high due to its simplicity and the absence of hazardous gas evolution or highly exothermic events that are difficult to manage on a large scale. The reaction operates at moderate temperatures and atmospheric pressure, making it compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. From an environmental standpoint, the avoidance of malodorous thiols and the generation of less toxic byproducts simplify waste treatment protocols, aiding in compliance with increasingly stringent environmental regulations. This green chemistry profile not only reduces disposal costs but also aligns with the sustainability goals of modern pharmaceutical companies, making the supplier a more attractive partner for long-term collaborations focused on responsible manufacturing.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation and optimization of this synthesis method, we have compiled a set of frequently asked questions based on the patent data and practical experience. These insights cover critical aspects such as catalyst selection, substrate compatibility, and safety considerations, providing a quick reference for technical teams evaluating this route. Understanding these nuances is vital for ensuring a smooth technology transfer from the laboratory to the pilot plant and eventually to full commercial production. We encourage potential partners to review these details to assess the feasibility of this method for their specific project requirements.
Q: What are the critical reaction conditions for this synthesis?
A: The reaction requires heating between 100-140°C in an air atmosphere using acetonitrile as the solvent. Key reagents include sodium metabisulfite and a methyl-containing peroxide like dicumyl peroxide, with ferric chloride acting as the catalyst.
Q: Does this method support diverse substrate scopes?
A: Yes, the protocol demonstrates excellent tolerance for various substituents on the nitrogen atom, including phenyl, substituted phenyl (fluoro, chloro, cyano, ester, trifluoromethyl), naphthyl, and benzyl groups, making it highly versatile for drug discovery.
Q: Is this process suitable for industrial scale-up?
A: Absolutely. The use of inexpensive, commercially available raw materials like sodium metabisulfite and the elimination of complex multi-step sequences make this one-pot strategy ideal for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylsulfonyl Nitrogen Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in patent CN112279801B and are fully equipped to leverage this technology for our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are designed to handle oxidative reactions and multi-component syntheses with the highest standards of safety and quality control. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-methylsulfonyl nitrogen heterocycles meets the exacting requirements of the pharmaceutical industry, delivering consistency and reliability that you can trust for your critical drug development programs.
We invite you to collaborate with us to unlock the full commercial potential of this innovative synthesis route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this one-pot strategy can optimize your budget. Please contact our technical procurement team today to request specific COA data for relevant analogues and comprehensive route feasibility assessments. By partnering with us, you gain access to a supply chain that is not only cost-effective and reliable but also deeply committed to advancing the frontiers of organic synthesis for the benefit of global healthcare.
