Advanced Halomethylation Strategy for Commercial Scale Sevoflurane Production
The global demand for inhalational anesthetics continues to drive innovation in process chemistry, specifically for Sevoflurane, a critical agent in modern general anesthesia. Patent CN101381289A introduces a transformative halomethylation methodology that addresses long-standing inefficiencies in Sevoflurane manufacturing. Unlike traditional pathways that rely heavily on hazardous anhydrous hydrogen fluoride and complex purification sequences, this novel approach leverages hexafluoroisopropanol, formaldehyde equivalents, and aluminum chloride to achieve superior purity profiles. The technical breakthrough lies in the strategic use of Lewis acid catalysis to control the chloromethylation step, followed by a highly efficient fluorination in polyethylene glycol media. For pharmaceutical manufacturers, this represents a paradigm shift towards safer, more controllable, and economically viable production of high-purity API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sevoflurane has been plagued by severe operational challenges and safety hazards inherent to early patented methods. Prior art, such as U.S. Pat 3683092 and US3689571, describes reactions utilizing hexafluoroisopropanol and polyoxymethylene with anhydrous hydrogen fluoride at low temperatures (around 9°C). These processes suffer from low conversion rates, often yielding crystallized polyether intermediates with productivity as low as 52%, necessitating cumbersome recycling of unreacted hexafluoroisopropanol. Furthermore, methods disclosed in U.S. Pat 4250334 involve heating mixtures of sulfuric acid and hydrogen fluoride to 65°C, creating extremely corrosive environments that demand expensive, specialized pressure-resistant equipment. The reliance on liquid HF not only escalates capital expenditure for corrosion-proof reactors but also introduces significant safety liabilities regarding leak management and worker exposure. Additionally, these conventional routes frequently generate complex by-product profiles, including various polyethers and halogenated impurities, which complicate downstream purification and reduce overall process mass intensity.
The Novel Approach
In stark contrast, the methodology outlined in CN101381289A offers a streamlined, three-route strategy that bypasses the need for pressurized HF reactors in the initial stages. The core innovation involves the direct conversion of hexafluoroisopropanol and formaldehyde equivalents (such as trioxymethylene or paraformaldehyde) into a chloromethyl ether intermediate using aluminum chloride as a catalyst. This halomethylation step proceeds under mild conditions (0°C to 80°C) in the presence of strong acids like concentrated sulfuric acid or oleum, eliminating the immediate need for gaseous or anhydrous HF handling during the carbon-oxygen bond formation. The subsequent fluorination step utilizes solid metal fluorides (KF or NaF) in phase-transfer solvents like PEG-400, which facilitates the nucleophilic substitution of chlorine with fluorine under atmospheric pressure. This decoupling of the chloromethylation and fluorination steps allows for precise control over reaction kinetics, resulting in crude product purities exceeding 97% and final distilled purities greater than 99.995%.
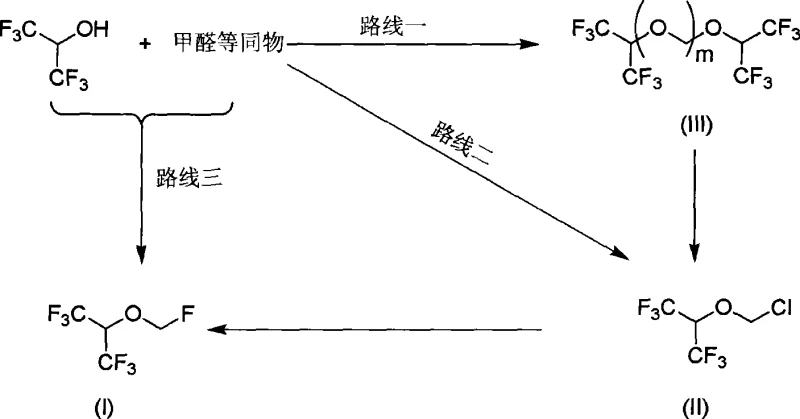
Mechanistic Insights into AlCl3-Catalyzed Halomethylation and Fluorination
The mechanistic elegance of this process centers on the activation of formaldehyde equivalents by aluminum chloride (AlCl3), a potent Lewis acid. In the chloromethylation phase, AlCl3 coordinates with the oxygen atoms of the formaldehyde source (e.g., trioxymethylene), increasing the electrophilicity of the methylene carbon. This activated species is then attacked by the hydroxyl group of hexafluoroisopropanol, leading to the formation of the critical chloromethyl 2,2,2-trifluoro-1-(trifluoromethyl) ethyl ether intermediate. The presence of strong acid further protonates the intermediate, facilitating the departure of leaving groups and stabilizing the transition state. Crucially, the reaction conditions are tuned to prevent over-polymerization, a common side reaction in prior art, by controlling the molar ratios and temperature gradients (preferably 20°C to 60°C). This precision ensures that the intermediate formed is predominantly the mono-chloromethyl ether rather than unwanted polyether chains, which are difficult to separate and reduce yield.
Following the formation of the chloromethyl ether, the fluorination mechanism relies on the unique solvation properties of polyethylene glycol (PEG). In this phase, the potassium fluoride (KF) is partially solubilized by the PEG chain, effectively acting as a phase-transfer catalyst that exposes the fluoride anion for nucleophilic attack. The fluoride ion displaces the chlorine atom on the chloromethyl group via an SN2 mechanism. The use of PEG-400 or triglycol is pivotal here, as it stabilizes the cation (K+) while leaving the anion (F-) relatively "naked" and highly reactive. This environment allows the fluorination to proceed efficiently at temperatures between 55°C and 120°C without the need for exotic fluorinating agents like DAST or Deoxo-Fluor, which are cost-prohibitive on a tonnage scale. The result is a clean conversion where the primary impurities are minimized, and the final distillation yields a product meeting stringent USP pharmacopeia standards with Impurity B completely absent.
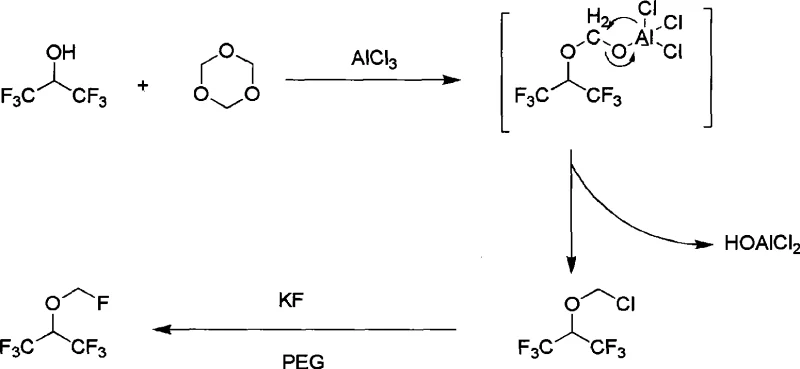
How to Synthesize Sevoflurane Efficiently
The synthesis of Sevoflurane via this halomethylation pathway is designed for operational simplicity and robustness, making it ideal for technology transfer from R&D to pilot and commercial scales. The process typically begins with the preparation of the polyether or direct chloromethyl ether intermediate by mixing hexafluoroisopropanol with trioxymethylene and a catalytic amount of concentrated sulfuric acid. Once the intermediate is formed, it is treated with anhydrous aluminum chloride, often without the need for isolation, to generate the chloromethyl ether species. The final step involves the addition of metal fluoride in a glycol solvent, followed by reflux and atmospheric distillation. This telescoped approach minimizes unit operations and solvent swaps, directly addressing the efficiency gaps found in legacy processes. For detailed standard operating procedures and specific stoichiometric ratios validated in our labs, please refer to the technical guide below.
- React hexafluoroisopropanol with formaldehyde equivalents (like trioxymethylene) under strong acid catalysis to form polyether intermediates.
- Treat the intermediate with aluminum chloride (AlCl3) to generate chloromethyl 2,2,2-trifluoro-1-(trifluoromethyl) ethyl ether.
- Perform nucleophilic substitution using metal fluoride (KF/NaF) in a phase transfer solvent like PEG-400 to yield high-purity Sevoflurane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this halomethylation technology translates into tangible strategic advantages beyond mere chemical yield. The most significant impact is the drastic reduction in capital and operational expenditures associated with corrosion management. By shifting away from continuous anhydrous HF usage in the primary bond-forming steps, manufacturers can utilize standard glass-lined or high-grade stainless steel reactors instead of expensive Monel or Hastelloy vessels required for high-pressure HF processes. This material flexibility significantly lowers the barrier to entry for production and reduces maintenance downtime. Furthermore, the ability to recover and reuse solvents like PEG and the crude Sevoflurane itself as a reaction medium creates a closed-loop system that minimizes raw material consumption. The simplified post-treatment workflow, which avoids complex extraction and washing sequences for intermediate purification, shortens the batch cycle time considerably, thereby enhancing overall plant throughput and asset utilization rates.
- Cost Reduction in Manufacturing: The elimination of expensive fluorinating reagents and the reduction in corrosion-resistant equipment requirements lead to substantial cost savings. The process utilizes commodity chemicals like aluminum chloride and potassium fluoride, which are readily available and inexpensive compared to specialized fluorinating agents. Additionally, the high selectivity of the reaction minimizes the loss of valuable hexafluoroisopropanol, a costly starting material, ensuring that raw material costs are optimized through high atom economy and efficient recycling protocols.
- Enhanced Supply Chain Reliability: Reliance on hazardous gases like anhydrous HF often subjects supply chains to regulatory bottlenecks and transportation restrictions. This solid-state and liquid-phase methodology mitigates those risks by using stable, transportable solids and liquids. The robustness of the reaction conditions, which tolerate a wider range of temperatures and do not require cryogenic cooling (as seen in some prior art requiring 9°C), ensures consistent production output regardless of minor environmental fluctuations, securing a steady flow of high-purity API intermediates to downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently green, generating minimal "three wastes" (waste water, gas, and residue) due to the high conversion rates and solvent recyclability. The absence of heavy metal catalysts simplifies waste treatment and disposal, aligning with increasingly strict global environmental regulations. Scalability is proven by the patent's demonstration of successful scale-up from gram-scale experiments to 50kg batches without loss of efficiency, confirming that the chemistry holds up under the thermal and mixing dynamics of large industrial reactors, facilitating rapid commercial ramp-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Sevoflurane synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on purity profiles, safety protocols, and scalability potential. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary impurities in conventional Sevoflurane synthesis?
A: Conventional methods often struggle with Impurity A, Impurity B, and residual chloromethyl ether. The novel halomethylation route described in CN101381289A effectively eliminates Impurity B and reduces Impurity A to trace levels (1-5 ppm).
Q: How does the new method improve safety compared to traditional HF processes?
A: Traditional routes rely heavily on anhydrous hydrogen fluoride (HF) and concentrated sulfuric acid under pressure. The new method utilizes aluminum chloride and solid metal fluorides in PEG solvents, significantly reducing corrosion risks and the need for specialized pressure-resistant equipment.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the patent explicitly demonstrates scalability from laboratory flasks to 50kg+ batches (Embodiment 6). The process avoids complex separation steps, allows for solvent recovery, and maintains high yields (>85% after rectification) suitable for commercial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sevoflurane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the halomethylation process are fully realized in practice. Our facilities are equipped with state-of-the-art corrosion-resistant reactors and rigorous QC labs capable of detecting impurities at the ppm level, guaranteeing that every batch of Sevoflurane meets stringent purity specifications required by global pharmacopeias. We are committed to delivering not just a chemical product, but a reliable supply solution that enhances your operational efficiency.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this greener, more efficient route. Contact us today to obtain specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technological excellence can drive value for your pharmaceutical supply chain.
