Revolutionizing Ribociclib Production: A Cost-Effective Industrial Synthesis Route for Global Supply Chains
Revolutionizing Ribociclib Production: A Cost-Effective Industrial Synthesis Route for Global Supply Chains
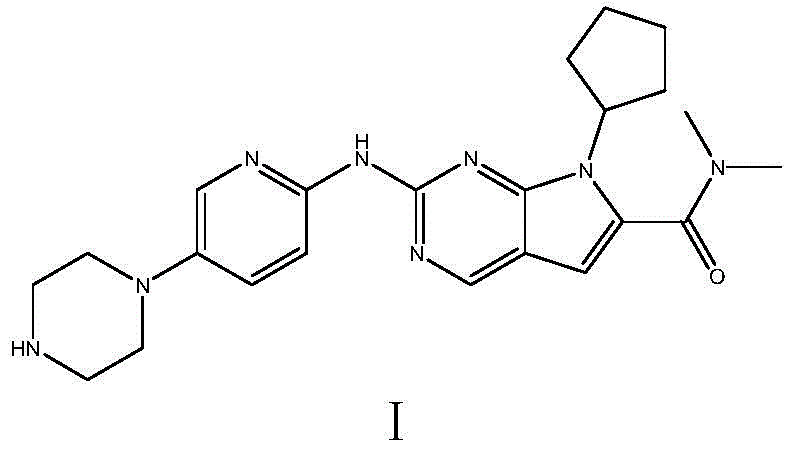
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, with CDK4/6 inhibitors like Ribociclib (Kisqali) playing a pivotal role in treating advanced breast cancer. However, the complexity of synthesizing these potent molecules has often been a bottleneck for global supply chains. A groundbreaking industrial preparation method detailed in patent CN109928975B offers a transformative solution to these challenges. This patent discloses a novel synthetic pathway that bypasses the reliance on expensive noble metal catalysts and toxic reagents traditionally associated with Ribociclib production. By shifting the paradigm from complex cross-coupling reactions to a streamlined condensation and N-substitution strategy, this technology promises to redefine the economics of kinase inhibitor manufacturing. For R&D directors and procurement managers alike, understanding this shift is critical for securing a reliable Ribociclib intermediate supplier capable of meeting future demand without compromising on purity or cost-efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ribociclib has relied heavily on routes described in patents such as WO2010020675 and US20120115878. These conventional pathways typically utilize 2,4-dichloro-5-bromopyrimidine as a starting material, necessitating a Sonogashira coupling reaction with propargyl alcohol. While chemically valid, this approach introduces significant logistical and financial burdens. The requirement for palladium catalysts not only inflates raw material costs but also necessitates rigorous downstream processing to remove trace heavy metals to meet stringent pharmaceutical specifications. Furthermore, these routes often employ manganese dioxide oxidants and highly toxic cyanides, creating substantial safety hazards and generating complex waste streams that are difficult and costly to treat. The cumulative effect is a process that is fragile, expensive, and environmentally taxing, posing risks to cost reduction in kinase inhibitor manufacturing initiatives.
The Novel Approach
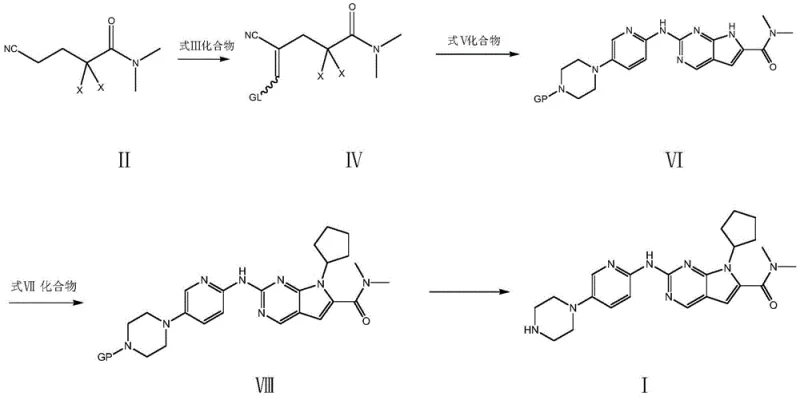
In stark contrast, the method disclosed in CN109928975B introduces a robust alternative that prioritizes industrial feasibility and safety. This innovative route begins with the condensation of N,N-dimethyl-2,2-dihalo-4-cyano-N-butylamide (Compound II) with a methylene reagent (Compound III) to form an intermediate (Compound IV). This intermediate is then condensed with a substituted guanidine (Compound V) to construct the core pyrrolo[2,3-d]pyrimidine scaffold (Compound VI) in a one-pot fashion. Subsequent N-substitution with halogenated cyclopentane and final deprotection yields the target molecule. This strategy completely eliminates the need for noble metals and toxic cyanides, utilizing cheap and easily obtained raw materials instead. The result is a process that is not only safer to operate but also inherently more scalable, addressing the critical need for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into the Condensation and Cyclization Strategy
The core innovation of this patent lies in the efficient construction of the fused heterocyclic system through a sequential condensation mechanism. The process initiates with the activation of the dihalo-amide precursor using a methyleneating agent, such as N,N-dimethylformamide dimethyl acetal or tri-orthoformates, in the presence of a catalyst like piperidine or zinc chloride. This step generates a reactive enamine or imine intermediate (Compound IV) which possesses the necessary electronic configuration for subsequent cyclization. When this intermediate reacts with the guanidine derivative (Compound V) at elevated temperatures (80-150°C), a cascade of nucleophilic attacks and eliminations occurs, effectively closing the pyrimidine ring onto the pre-existing carbon chain. This mechanistic pathway is highly advantageous because it avoids the harsh conditions and regioselectivity issues often encountered in direct pyrimidine functionalization, ensuring the formation of the correct 7H-pyrrolo[2,3-d]pyrimidine core with high fidelity.
Following the core formation, the N-substitution step introduces the cyclopentyl moiety, a critical structural feature for CDK4/6 inhibitory activity. This reaction proceeds via a standard nucleophilic substitution mechanism where the nitrogen atom of the pyrrole ring attacks the halogenated cyclopentane (Compound VII) in the presence of a base such as potassium carbonate. The choice of solvent, ranging from polar aprotic solvents like DMF and DMSO to alcohols like N-butanol, plays a crucial role in solubilizing the reactants and stabilizing the transition state. The patent data indicates that this step can achieve liquid phase purities exceeding 99%, demonstrating exceptional control over side reactions and impurities. This high level of purity at the intermediate stage significantly simplifies the final purification steps, directly contributing to reducing lead time for high-purity API intermediates and ensuring a consistent supply of high-purity CDK4/6 inhibitor materials.
How to Synthesize Ribociclib Efficiently
The synthesis of Ribociclib via this novel route is designed for operational simplicity and high yield, making it an ideal candidate for technology transfer and large-scale manufacturing. The process is divided into three distinct stages: the formation of the heterocyclic core, the installation of the cyclopentyl group, and the final deprotection. Each step utilizes common industrial reagents and standard equipment, minimizing the need for specialized infrastructure. The following guide outlines the standardized synthesis steps derived from the patent examples, providing a clear roadmap for production teams aiming to implement this superior methodology.
- Condense N,N-dimethyl-2,2-dihalo-4-cyano-N-butylamide with a methylene reagent and subsequently with a protected guanidine derivative to form the pyrrolo[2,3-d]pyrimidine core.
- Perform N-substitution on the intermediate using halogenated cyclopentane and an inorganic base in a polar aprotic solvent.
- Remove the protecting group (PG) via acid hydrolysis or hydrogenolysis to obtain the final Ribociclib API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic advantages beyond mere chemical elegance. The primary benefit is the drastic simplification of the supply chain risk profile. By removing dependence on volatile noble metal markets and hazardous reagents like cyanides, manufacturers can secure a more stable and predictable flow of raw materials. This stability is crucial for maintaining continuous production schedules and avoiding the disruptions that often plague complex pharmaceutical supply chains. Furthermore, the elimination of heavy metal catalysts removes the need for expensive and time-consuming scavenging processes, which traditionally add significant cost and time to the manufacturing cycle. This streamlined approach allows for faster batch turnover and more efficient resource utilization.
- Cost Reduction in Manufacturing: The economic implications of this new route are profound. By replacing expensive palladium catalysts and complex oxidants with inexpensive organic bases and simple halides, the direct material cost is significantly lowered. Additionally, the ability to recover and reuse solvents, such as the hydrogen chloride tetrahydrofuran solution mentioned in the patent examples, further drives down operational expenses. The simplified workup procedures, which often involve simple filtration and washing rather than complex chromatography, reduce labor and utility costs. These factors combine to create a manufacturing process that is inherently more cost-competitive, offering substantial savings that can be passed down the supply chain or reinvested in R&D.
- Enhanced Supply Chain Reliability: Reliability is the cornerstone of a robust pharmaceutical supply chain. This synthesis method enhances reliability by utilizing raw materials that are commodity chemicals, readily available from multiple global suppliers. Unlike specialized catalysts that may have long lead times or single-source dependencies, reagents like N-butanol, potassium carbonate, and halogenated cyclopentanes are widely produced. This diversification of the supply base mitigates the risk of shortages and price spikes. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in input quality, ensuring consistent output even when sourcing from different vendors, thereby strengthening the overall resilience of the supply network.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, environmental compliance has become a key differentiator. This novel route is inherently greener, generating significantly less hazardous waste compared to traditional methods. The absence of cyanides and heavy metals simplifies wastewater treatment and reduces the burden on waste disposal systems. The process is designed for scalability, with reaction conditions that are easily managed in large reactors (e.g., temperatures below 150°C and atmospheric or low pressure). This ease of scale-up ensures that production can be ramped up quickly to meet market demand without encountering the engineering bottlenecks often associated with more hazardous chemistries, ensuring a sustainable and compliant manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial implementation of this Ribociclib synthesis method. These answers are derived directly from the technical disclosures in patent CN109928975B and provide clarity on the practical aspects of adopting this technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new synthesis route improve cost efficiency compared to traditional methods?
A: The novel route described in CN109928975B eliminates the need for expensive noble metal catalysts (like Palladium in Sonogashira couplings) and toxic oxidants (like Manganese Dioxide). By utilizing cheap, commercially available starting materials such as N,N-dimethyl-2,2-dihalo-4-cyano-N-butylamide and avoiding complex purification steps associated with heavy metal removal, the overall manufacturing cost is significantly reduced.
Q: What are the safety advantages of this industrial preparation method?
A: Traditional synthesis routes often involve highly toxic cyanides and hazardous oxidants which pose significant safety risks and generate difficult-to-treat waste streams. This new method avoids cyanides entirely and uses safer reagents like halogenated cyclopentane and standard organic bases. The simplified workflow reduces potential safety hazards and minimizes the generation of 'three wastes' (waste water, gas, and solids), facilitating easier environmental compliance.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights the method's suitability for industrialization. The reaction conditions are mild (temperatures ranging from 20°C to 130°C), and the solvents used (such as N-butanol, THF, and acetonitrile) are standard industrial solvents. Furthermore, the ability to recover and reuse solvents like hydrogen chloride tetrahydrofuran solution enhances the economic viability and scalability for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ribociclib Intermediate Supplier
The technological advancements presented in patent CN109928975B represent a significant leap forward in the industrial production of Ribociclib, offering a pathway that is safer, cleaner, and more economical than previous iterations. At NINGBO INNO PHARMCHEM, we recognize the value of such innovations and have positioned ourselves at the forefront of implementing these advanced synthetic strategies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of Ribociclib intermediate we produce adheres to the highest global standards. We are committed to translating complex patent chemistry into reliable, commercial reality.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this superior synthesis route. Whether you require custom synthesis services or bulk supply of key intermediates, our technical team is ready to assist. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to see how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall cost of goods sold.
