Advanced Udenafil Manufacturing: High-Purity Synthesis for Global Supply Chains
The global pharmaceutical landscape is continuously evolving to meet the escalating demand for effective treatments for erectile dysfunction (ED), with PDE-5 inhibitors remaining the cornerstone of therapeutic intervention. Within this competitive sector, the chemical synthesis of key active pharmaceutical ingredients like Udenafil requires constant innovation to address the limitations of legacy manufacturing protocols. Patent CN107759603B introduces a transformative preparation method for heterocyclic compounds represented by Udenafil, specifically designed to overcome the critical bottlenecks of high production costs and low yields that have historically plagued this chemical class. This technical insight report analyzes the strategic advantages of this novel route, which prioritizes a cyclization-first strategy to enhance reaction selectivity and overall process efficiency. By fundamentally reordering the synthetic steps, this methodology ensures that the final product achieves exceptional purity levels while significantly mitigating the environmental and safety hazards associated with traditional sulfonation processes. 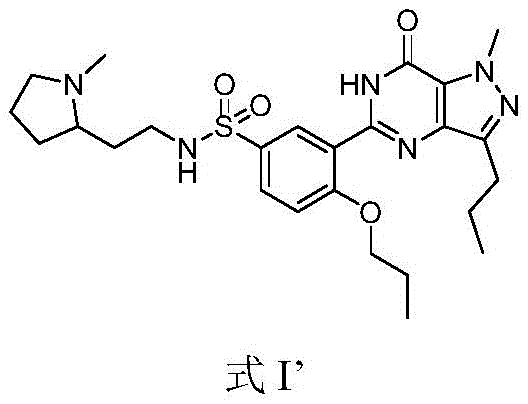
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Udenafil has been constrained by inefficient reaction sequences that introduce significant economic and operational burdens on the supply chain. Prior art methods, such as those disclosed in earlier patents, typically initiate the synthesis by constructing the sulfonamide moiety at a very early stage using chlorosulfonic acid. This approach creates a highly water-soluble sulfonamide intermediate that is notoriously difficult to separate and purify from the reaction mixture, leading to substantial product loss during workup procedures. Furthermore, these conventional routes often necessitate the use of expensive coupling reagents or activators, such as EEDQ or trichlorobenzoyl chloride, to facilitate the formation of the carboxamide bond, which drastically inflates the raw material costs. The early introduction of expensive starting materials like N-methyl-2-(2-aminoethyl)-pyrrolidine also means that any yield loss in subsequent steps results in a disproportionate financial impact. Additionally, the heavy reliance on chlorosulfonic acid in the initial steps generates large volumes of acidic waste gas and wastewater, creating severe environmental compliance challenges and safety hazards for large-scale workshop production. 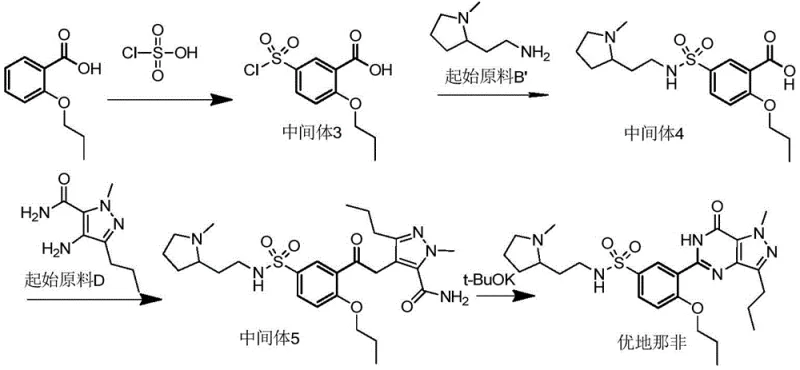
The Novel Approach
In stark contrast to the legacy methodologies, the novel approach detailed in patent CN107759603B ingeniously reverses the traditional order of operations to maximize efficiency and minimize waste. The core innovation lies in performing the intramolecular cyclization reaction as the very first step, converting the starting material directly into the pyrazolopyrimidinone intermediate before any sulfonation occurs. This strategic reordering effectively passivates the chemical activity of the heterocyclic ring, which subsequently enhances the selectivity of the sulfonation reaction in the second step, thereby reducing the formation of unwanted by-products. By delaying the introduction of the expensive pyrrolidinamine starting material until the final step, the process ensures that this high-value reagent is not subjected to the harsh conditions of the earlier cyclization and sulfonation stages, preserving its integrity and maximizing its conversion into the final product. This streamlined three-step sequence not only simplifies the purification process but also creates a robust manufacturing protocol that is inherently safer and more cost-effective for commercial implementation. 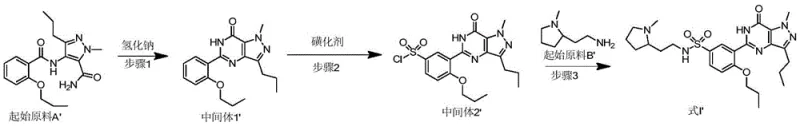
Mechanistic Insights into Base-Catalyzed Cyclization and Selective Sulfonation
The mechanistic foundation of this improved synthesis rests on the precise control of reactivity through the initial cyclization step, which fundamentally alters the electronic properties of the intermediate. In the first stage, the starting material undergoes a base-catalyzed intramolecular condensation in an organic solvent, facilitated by alkali metal salts or hydroxides, to form the stable pyrazolopyrimidinone ring system. This cyclization is critical because it locks the molecular conformation and reduces the electron density on the aromatic ring, which would otherwise be highly susceptible to non-selective electrophilic attack during sulfonation. When the resulting intermediate is subsequently treated with a sulfonating agent, such as chlorosulfonic acid or a chlorosulfonic acid-thionyl chloride mixture, the reaction proceeds with high regioselectivity at the desired position on the benzene ring. This high selectivity is a direct consequence of the deactivating effect of the newly formed heterocyclic ring, which prevents over-sulfonation or the formation of isomeric impurities that are common in the prior art methods. The ability to control the sulfonation so precisely means that the resulting sulfonyl chloride intermediate is of significantly higher quality, requiring less rigorous purification before moving to the final coupling step.
Furthermore, the impurity control mechanism is deeply integrated into the reaction design, ensuring that the final Udenafil product meets stringent pharmaceutical quality standards. By avoiding the use of coupling reagents in the amide bond formation, the process eliminates a major source of difficult-to-remove organic impurities that often persist through multiple recrystallization steps in conventional synthesis. The final reaction between the sulfonyl chloride intermediate and the amine starting material is conducted under mild conditions in aprotic solvents, which minimizes thermal degradation and side reactions. The result is a crude product with exceptionally high purity, often exceeding ninety-nine percent, which significantly reduces the burden on downstream purification units. This mechanistic advantage translates directly into a more reliable supply of high-purity pharmaceutical intermediates, as the process is less sensitive to minor fluctuations in reaction parameters, ensuring consistent batch-to-batch quality that is essential for regulatory compliance in the global pharmaceutical market.
How to Synthesize Udenafil Efficiently
The implementation of this synthesis route requires careful attention to reaction conditions and stoichiometry to fully realize the benefits of the cyclization-first strategy. The process begins with the preparation of the cyclized intermediate using a suitable base and solvent system, followed by a controlled sulfonation step that manages exothermicity and reagent consumption. The final coupling step is designed to be high-yielding and clean, leveraging the high reactivity of the sulfonyl chloride intermediate. Detailed standardized synthesis steps see the guide below.
- Cyclize starting material A with base in organic solvent to form intermediate 1.
- React intermediate 1 with sulfonating agent to generate intermediate 2.
- Couple intermediate 2 with starting material B to obtain final Udenafil product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling economic and operational benefits that directly impact the bottom line. The elimination of expensive coupling reagents and activators represents a direct reduction in the bill of materials, while the strategic delay of costly amine reagents to the final step minimizes financial exposure to yield losses in earlier stages. The process is designed to utilize readily available raw materials that are sourced from stable supply chains, reducing the risk of production delays caused by raw material shortages. Furthermore, the mild reaction conditions and reduced usage of hazardous sulfonating agents lower the operational costs associated with waste treatment and safety compliance, making the manufacturing process more sustainable and economically viable in the long term.
- Cost Reduction in Manufacturing: The most significant financial advantage of this process is the complete avoidance of expensive coupling reagents like EEDQ, which are traditionally required to drive the amide bond formation in older synthesis routes. By utilizing a direct sulfonation and coupling strategy on a pre-cyclized intermediate, the method removes a major cost driver from the production budget, leading to substantial cost savings in Udenafil manufacturing. Additionally, placing the expensive pyrrolidinamine starting material at the very end of the synthesis ensures that this high-value component is not wasted on intermediates that might be discarded due to low yields in previous steps. This logical reordering of the synthetic sequence ensures that the most expensive inputs are only introduced when the probability of successful conversion to the final product is highest, optimizing the overall cost efficiency of the production line.
- Enhanced Supply Chain Reliability: From a supply chain perspective, this method relies on starting materials that are commercially available and easy to source, reducing dependency on specialized or custom-synthesized reagents that can cause bottlenecks. The robustness of the reaction conditions means that the process is less prone to failures or deviations that could interrupt production schedules, ensuring a consistent and reliable supply of the pharmaceutical intermediate. The reduction in hazardous waste generation also simplifies the logistics of waste disposal, removing a potential regulatory hurdle that could otherwise delay shipments or increase operational overhead. This stability is crucial for maintaining continuous production runs and meeting the demanding delivery timelines required by global pharmaceutical clients.
- Scalability and Environmental Compliance: The design of this synthesis route is inherently scalable, with reaction conditions that are mild enough to be safely managed in large-scale reactors without requiring extreme temperatures or pressures. The significant reduction in the usage of chlorosulfonic acid, achieved by improving reaction selectivity, directly translates to a lower volume of acidic wastewater and waste gas that requires treatment. This environmental advantage not only reduces the cost of waste management but also aligns with increasingly stringent global environmental regulations, ensuring that the manufacturing facility remains compliant and operational. The ability to scale this process from laboratory to commercial production without significant re-engineering makes it an ideal candidate for rapid capacity expansion to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the specific advantages detailed in the patent data. These answers provide clarity on how the process improvements translate into tangible benefits for production and quality control teams. Understanding these details is essential for evaluating the feasibility of adopting this route for commercial manufacturing.
Q: How does this new process improve yield compared to conventional methods?
A: By performing cyclization before sulfonation, the method passivates the compound's activity, improving selectivity and achieving yields over 74% compared to less than 30% in older routes.
Q: What are the cost advantages of this synthesis route?
A: The process eliminates expensive coupling reagents like EEDQ and delays the use of costly pyrrolidinamine to the final step, significantly reducing raw material costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild, and the strategy reduces the usage of chlorosulfonic acid, minimizing waste acid treatment and enhancing safety for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Udenafil Supplier
The technical potential of this cyclization-first synthesis route represents a significant opportunity for pharmaceutical companies to optimize their supply chains for PDE-5 inhibitors. NINGBO INNO PHARMCHEM, as a specialized CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative process to life. Our facility is equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of Udenafil produced meets the highest international standards for safety and efficacy. We understand the critical importance of consistency in pharmaceutical manufacturing and have the technical capability to adapt this patent-protected methodology to fit specific client requirements while maintaining full regulatory compliance.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply strategy. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this high-purity Udenafil production method for your commercial needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
