Scalable Production of 3,3'-Bis(Trifluoromethyl) Benzophenone via Advanced Photo-Chlorination
The global demand for high-performance fluorinated intermediates continues to surge, driven by their critical applications in pharmaceuticals, agrochemicals, and advanced polymer materials. A pivotal development in this sector is detailed in patent CN101973862B, which discloses a robust and economically viable method for preparing 3,3'-bis(trifluoromethyl) benzophenone. This compound serves as a vital UV-absorbing agent and a key building block for fluoro-containing macromolecular materials, necessitating a synthesis route that balances high yield with environmental compliance. The patented technology introduces a novel two-step sequence involving photo-chlorination followed by alkaline hydrolysis, offering a distinct advantage over legacy methods that often suffer from excessive waste generation or operational complexity. For R&D directors and procurement strategists seeking a reliable fluorinated pharmaceutical intermediate supplier, understanding the mechanistic superiority of this route is essential for securing long-term supply chain stability.
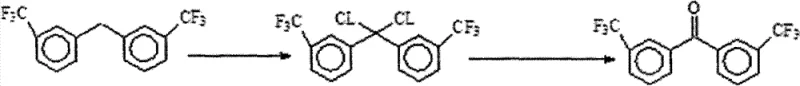
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,3'-bis(trifluoromethyl) benzophenone has relied on methods that present significant hurdles for modern industrial application. One prominent prior art method, documented in DE3411326 and EP0156278, utilizes 65% nitric acid to oxidize 3,3'-bis(trifluoromethyl) diphenylmethane under reflux conditions. While this pathway can achieve the target transformation, it is plagued by the production of substantial quantities of spent acid waste, creating a heavy burden on wastewater treatment facilities and escalating overall operational expenditures. Furthermore, the reported yield for this nitric acid oxidation route hovers around 65%, which is suboptimal for high-value fine chemical manufacturing where atom economy is paramount. Another alternative involves Grignard reactions using m-trifluoromethylphenyl magnesium bromide and m-trifluoromethyl cyanobenzene; however, the stringent requirement for anhydrous conditions and the difficulty in controlling exothermic reactions make this approach notoriously difficult to industrialize safely and consistently.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN101973862B leverages a free-radical photo-chlorination strategy that fundamentally simplifies the process architecture. By utilizing 3,3'-bis(trifluoromethyl) diphenylmethane as the starting material and subjecting it to irradiation from an incandescent lamp in the presence of chlorine gas and a phosphorus pentachloride catalyst, the process efficiently generates the dichloride intermediate. This intermediate is subsequently converted to the target ketone through a straightforward alkaline hydrolysis step. This approach not only circumvents the generation of hazardous nitrogen oxide byproducts but also operates under relatively mild thermal conditions (60-80°C), significantly lowering energy consumption. For stakeholders focused on cost reduction in fluorinated intermediate manufacturing, this shift from oxidative degradation to constructive functionalization represents a major leap forward in process efficiency and environmental sustainability.
Mechanistic Insights into Photo-Chlorination and Hydrolysis
The core of this synthetic innovation lies in the controlled radical substitution at the benzylic position. Under the irradiation of incandescent light, chlorine molecules undergo homolytic cleavage to generate chlorine radicals, which then abstract hydrogen atoms from the methylene bridge of the diphenylmethane substrate. The presence of phosphorus pentachloride acts as a crucial catalyst, likely facilitating the propagation of the radical chain reaction and ensuring high selectivity for the dichloride species over mono-chlorinated or over-chlorinated byproducts. The reaction is typically conducted in solvents such as carbon tetrachloride or chloroform, which provide an inert medium that supports the radical mechanism without interfering with the reactive intermediates. Careful monitoring via Gas Chromatography (GC) and Nuclear Magnetic Resonance (NMR) allows for precise endpoint determination, ensuring that the reaction proceeds to the desired dichloride stage without significant degradation of the aromatic rings or the trifluoromethyl groups.
Following the chlorination, the transformation to the ketone is achieved through nucleophilic substitution and elimination during the hydrolysis phase. The dichloride intermediate reacts with aqueous alkali (sodium hydroxide or potassium hydroxide) in a mixed solvent system containing acetonitrile. Acetonitrile plays a vital role here by enhancing the solubility of the organic intermediate in the aqueous phase, thereby increasing the interfacial contact area and accelerating the hydrolysis rate. The mechanism involves the displacement of chloride ions by hydroxide groups, followed by the elimination of water to form the carbonyl double bond characteristic of the benzophenone structure. This step is highly efficient, converting the colorless oil intermediate into white crystalline product with a sharp melting point of 102-104°C, indicating a high degree of structural integrity and purity suitable for downstream applications in high-purity OLED material or pharmaceutical synthesis.
How to Synthesize 3,3'-Bis(Trifluoromethyl) Benzophenone Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of reagents and the control of reaction parameters to maximize yield and safety. The process begins with the preparation of the chlorination mixture, where the molar ratio of chlorine to substrate is maintained between 2:1 and 6:1 to ensure complete conversion to the dichloride. The catalyst loading is optimized at a weight ratio of 0.2 to 0.5 relative to the substrate, balancing reaction speed with cost. Following the isolation of the intermediate, the hydrolysis step utilizes a specific weight ratio of water to intermediate (3:1 to 8:1) and alkali (2:1 to 4:1 molar ratio) to drive the reaction to completion within 3 to 5 hours under reflux. Detailed standardized operating procedures for scaling this chemistry from laboratory to production volumes are outlined below.
- Mix 3,3'-bis(trifluoromethyl) diphenylmethane with a solvent (carbon tetrachloride or chloroform) and phosphorus pentachloride catalyst.
- Heat the mixture to 60-80°C under reflux while irradiating with incandescent light and introducing chlorine gas for 12-20 hours to form the dichloride intermediate.
- Subject the intermediate to alkaline hydrolysis using sodium or potassium hydroxide in a water-acetonitrile system under reflux for 3-5 hours to obtain the final benzophenone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photo-chlorination technology offers tangible strategic benefits beyond mere technical feasibility. The elimination of nitric acid from the process flow removes a major bottleneck related to hazardous waste disposal and regulatory compliance, directly translating to lower overhead costs and reduced risk of production stoppages due to environmental violations. Furthermore, the use of common industrial solvents and standard lighting equipment means that existing manufacturing infrastructure can often be adapted for this process with minimal capital expenditure, accelerating the time-to-market for new product launches. This operational flexibility ensures a more resilient supply chain capable of responding rapidly to fluctuations in market demand for fluorinated specialty chemicals.
- Cost Reduction in Manufacturing: The economic profile of this method is significantly improved by the removal of expensive and hazardous reagents associated with older oxidation routes. By avoiding the generation of large volumes of spent acid, manufacturers save substantially on neutralization agents and wastewater treatment fees, which are often hidden costs in traditional chemical processing. Additionally, the higher overall yield (reported up to 85% after recrystallization) means that less raw material is required per kilogram of finished product, directly improving the gross margin. The simplicity of the work-up procedure, involving standard extraction and filtration, further reduces labor hours and utility consumption, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically 3,3'-bis(trifluoromethyl) diphenylmethane, are readily accessible through established supply channels, reducing the risk of raw material shortages that can plague more exotic synthetic routes. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high vacuum systems, minimizes the likelihood of equipment failure or batch loss due to sensitive process deviations. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of global pharmaceutical and agrochemical clients who depend on consistent quality and availability.
- Scalability and Environmental Compliance: From a sustainability perspective, this process aligns well with modern green chemistry principles by reducing the E-factor (mass of waste per mass of product). The absence of heavy metal catalysts or stoichiometric oxidants simplifies the purification of the final product, ensuring that residual impurities are kept to trace levels without complex chromatographic separations. This ease of purification facilitates the commercial scale-up of complex fluorinated intermediates, allowing manufacturers to confidently increase batch sizes from pilot plant scales to multi-ton annual production capacities while maintaining strict adherence to environmental regulations and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3,3'-bis(trifluoromethyl) benzophenone using this patented methodology. These insights are derived directly from the experimental data and process descriptions found in the source intellectual property, providing a transparent view of the technology's capabilities. Understanding these details helps potential partners evaluate the fit of this material within their specific formulation or synthesis pipelines.
Q: What are the primary advantages of this photo-chlorination method over traditional nitric acid oxidation?
A: The photo-chlorination method eliminates the generation of large quantities of spent acid waste associated with nitric acid oxidation, significantly reducing environmental treatment costs and simplifying the purification process while achieving higher yields (up to 85%).
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes standard industrial equipment such as reflux condensers and incandescent light sources, avoiding complex conditions like cryogenic temperatures required for Grignard reactions, making it highly amenable to commercial scale-up.
Q: What is the expected purity and physical state of the final product?
A: The final product is obtained as white crystals with a melting point of 102-104°C. Through recrystallization from ethanol, high purity specifications suitable for pharmaceutical and electronic applications can be consistently achieved.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Bis(Trifluoromethyl) Benzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of next-generation pharmaceuticals and advanced materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our dedication to process optimization allows us to offer competitive pricing without compromising on the quality or consistency that our global partners expect.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific applications. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and quality needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
