Advanced Racemization Strategy for Larotrectinib Intermediates Enhancing Commercial Viability
The pharmaceutical landscape for oncology treatments has been significantly reshaped by the advent of Tropomyosin Receptor Kinase (TRK) inhibitors, with Larotrectinib standing out as a paradigm-shifting therapy for solid tumors harboring NTRK gene fusions. As detailed in patent CN112457231A, the efficient synthesis of key chiral intermediates remains a critical bottleneck in meeting the global demand for this life-saving medication. The core challenge lies in the stereoselective construction of the (2R)-2-(2,5-difluorophenyl)pyrrolidine moiety, which is essential for the drug's biological activity. Traditional synthetic routes often struggle with the inherent inefficiency of chiral resolution, where nearly half of the synthesized material is discarded as the unwanted S-enantiomer. This patent introduces a groundbreaking racemization methodology that transforms this liability into an asset, enabling the continuous recycling of the S-isomer back into the production cycle. By addressing the fundamental atom economy issues associated with chiral resolution, this technology offers a robust pathway for reliable pharmaceutical intermediate supplier operations to enhance overall process efficiency.
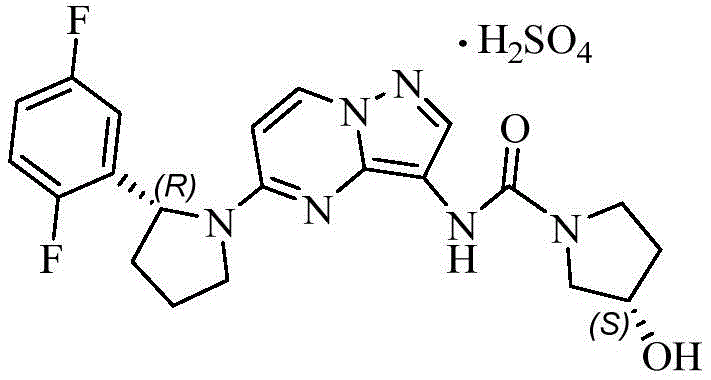
The structural complexity of Larotrectinib necessitates precise stereochemical control, as the therapeutic efficacy is strictly dependent on the (R)-configuration of the pyrrolidine ring. The innovation described in CN112457231A provides a mechanism to bypass the theoretical 50% yield ceiling imposed by classical resolution techniques. Instead of treating the S-enantiomer as waste, the disclosed method subjects it to base-catalyzed equilibration, effectively resetting the stereochemical clock and generating a racemic mixture that can be subjected to resolution once more. This approach not only maximizes the utility of starting materials but also aligns with modern green chemistry principles by drastically reducing the volume of chemical waste generated per kilogram of active pharmaceutical ingredient produced.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of the critical (2R)-2-(2,5-difluorophenyl)pyrrolidine fragment has relied on methodologies that are fraught with economic and operational disadvantages for large-scale manufacturing. As illustrated in prior art such as CN102264736B, early synthetic strategies employed palladium-catalyzed coupling reactions which, while effective, introduce significant complications regarding catalyst cost and residual metal removal. The reliance on expensive ligands like (-)-cytisine and palladium acetate creates a volatile cost structure that is highly sensitive to fluctuations in the precious metals market. Furthermore, the purification requirements to meet stringent regulatory limits for heavy metals in APIs add multiple downstream processing steps, increasing both the lead time and the environmental footprint of the manufacturing process.
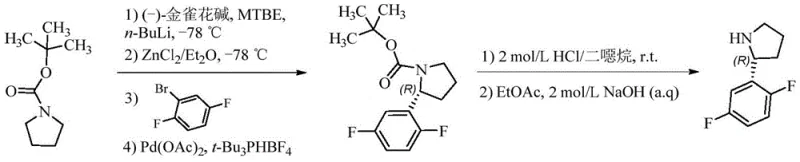
Beyond the issues of metal catalysis, alternative routes involving iridium complexes or stoichiometric chiral auxiliaries like tert-butylsulfinamide present their own set of challenges. These methods often require cryogenic conditions, such as temperatures as low as -30°C, which impose severe energy demands and limit the scalability of the reaction in standard industrial reactors. Additionally, routes utilizing strong reducing agents like lithium aluminum hydride pose significant safety hazards and complicate the workup procedures due to the formation of difficult-to-remove aluminum salts. When combined with the inherent 50% loss of material during the initial chiral resolution step, these conventional methods result in a cumulative yield that is commercially suboptimal, driving up the cost reduction in pharmaceutical intermediates manufacturing to unsustainable levels for generic competition.
The Novel Approach
In stark contrast to these complex and resource-intensive pathways, the novel racemization method disclosed in the patent offers a streamlined solution that leverages simple thermal activation and basic catalysis. The core of this innovation is the discovery that the S-enantiomer, previously considered a byproduct, can be efficiently converted into the racemic compound 2-(2,5-difluorophenyl)-1-pyrrolidine (Compound II) under relatively mild conditions. By utilizing common inorganic bases such as potassium hydroxide or sodium alkoxides in polar aprotic solvents like dimethyl sulfoxide (DMSO), the process eliminates the need for any transition metal catalysts entirely. This shift from noble metal catalysis to organo-basic catalysis represents a fundamental simplification of the synthetic tree, removing entire categories of impurities and reducing the complexity of the supply chain required to source specialized reagents.
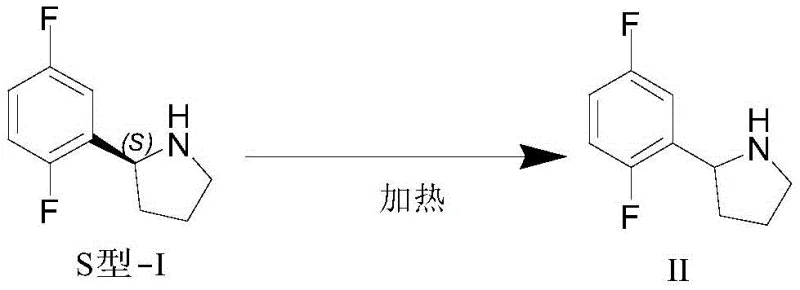
The operational simplicity of this new approach cannot be overstated, as it allows for the integration of a recycling loop directly into the existing resolution workflow. Once the desired R-enantiomer is crystallized out via salt formation with D-malic acid, the mother liquor containing the enriched S-isomer is not discarded but is instead fed directly into the racemization reactor. After heating at temperatures between 100°C and 140°C for a short duration of 1 to 8 hours, the material is returned to a racemic state, ready for another round of resolution. This closed-loop system effectively decouples the yield of the final product from the theoretical limits of a single resolution pass, offering a pathway to near-quantitative utilization of the carbon skeleton introduced at the beginning of the synthesis.
Mechanistic Insights into Base-Catalyzed Racemization
The chemical mechanism underpinning this transformation relies on the acidity of the proton at the chiral center (the 2-position of the pyrrolidine ring), which is activated by the adjacent electron-withdrawing 2,5-difluorophenyl group. Under the influence of a strong base in a high-boiling polar solvent, this alpha-proton is abstracted to form a planar carbanionic intermediate or an enamine-like species that loses its stereochemical information. Upon reprotonation by the solvent or conjugate acid present in the medium, the proton can attack from either face of the planar intermediate with equal probability, resulting in a statistical 1:1 mixture of the R and S enantiomers. The choice of solvent is critical here; DMSO is particularly effective not only because of its high boiling point which facilitates the required thermal energy but also because its polar nature stabilizes the charged intermediate, accelerating the rate of equilibration without promoting degradation side reactions.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. The absence of palladium or iridium eliminates the risk of forming organometallic byproducts that are notoriously difficult to purge to parts-per-million levels required by pharmacopeial standards. Furthermore, the use of inorganic bases like KOH generates inorganic salts upon quenching, which are easily removed via aqueous extraction, leaving the organic phase remarkably clean. The patent data indicates that chemical purity exceeding 99% is readily achievable with simple workup procedures involving extraction and washing. This high level of purity is crucial for the subsequent resolution step, as impurities can often interfere with the crystallization kinetics of the diastereomeric salt, potentially compromising the optical purity of the final R-enantiomer.
How to Synthesize (2R)-2-(2,5-difluorophenyl)pyrrolidine Efficiently
The implementation of this racemization strategy requires careful attention to reaction parameters to ensure complete equilibration while minimizing thermal degradation. The process begins with the isolation of the S-enriched fraction from the resolution mother liquors, which is then dissolved in a suitable solvent such as DMSO or DMF. A stoichiometric excess of base, typically ranging from 1 to 4 equivalents relative to the substrate, is added to drive the equilibrium. The mixture is then heated to the optimal temperature window identified in the patent examples, generally between 100°C and 140°C, and maintained under agitation until chiral HPLC analysis confirms that the enantiomeric excess has dropped to near zero, indicating successful racemization. Detailed standardized synthesis steps see the guide below.
- Combine the S-enriched pyrrolidine intermediate with a strong base such as potassium hydroxide in a polar aprotic solvent like DMSO.
- Heat the reaction mixture to a temperature range of 100-140°C and maintain stirring for 1 to 8 hours to facilitate racemization.
- Quench the reaction with water, extract the organic phase with dichloromethane, and purify via standard washing and concentration techniques.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free racemization technology translates directly into enhanced margin protection and supply security. The most immediate impact is the drastic reduction in raw material costs associated with the elimination of precious metal catalysts and specialized chiral ligands. By replacing these high-value inputs with commodity chemicals like potassium hydroxide and DMSO, the variable cost per kilogram of the intermediate is significantly lowered, providing a buffer against market volatility. Moreover, the ability to recycle the S-isomer effectively doubles the theoretical yield of the desired R-enantiomer from a fixed amount of starting material, which acts as a powerful force multiplier for production capacity without requiring capital investment in new reactor volume.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, primarily driven by the removal of expensive catalytic systems and the maximization of raw material utility. In traditional routes, the cost of the discarded S-enantiomer is effectively a sunk cost that must be amortized over the yield of the R-enantiomer; by recycling this material, the effective cost basis of the starting material is halved. Additionally, the avoidance of heavy metals simplifies the purification train, reducing the consumption of scavengers, filtration media, and solvent volumes required for polishing steps. These cumulative savings contribute to a substantially lower cost of goods sold (COGS), making the final API more competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, non-specialized reagents. Unlike proprietary chiral ligands or rare earth metals which may have constrained supply chains and long lead times, bases like NaOH and solvents like DMSO are produced globally in massive quantities. This commoditization of the reagent profile reduces the risk of supply disruption due to geopolitical issues or single-source vendor failures. Furthermore, the robustness of the reaction conditions—tolerating a range of bases and solvents—provides flexibility in sourcing, allowing procurement teams to optimize purchasing based on regional availability and spot pricing without compromising process validity.
- Scalability and Environmental Compliance: From an operational standpoint, the process is inherently scalable, utilizing unit operations such as heating, stirring, and liquid-liquid extraction that are standard in any multipurpose pharmaceutical plant. The absence of pyrophoric reagents like lithium aluminum hydride or air-sensitive metal catalysts simplifies the safety profile of the plant, reducing the need for specialized containment equipment. Environmentally, the reduction in waste generation through recycling aligns with increasingly stringent regulatory frameworks regarding solvent discharge and hazardous waste disposal. The simplified waste stream, consisting largely of aqueous salt solutions and recoverable organic solvents, facilitates easier compliance with environmental permits and reduces the overhead costs associated with waste treatment and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this racemization technology in a GMP environment. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for process evaluation. Understanding these nuances is essential for technical teams assessing the feasibility of technology transfer and for commercial teams evaluating the long-term viability of the supply partnership.
Q: Why is recycling the S-isomer critical for Larotrectinib production?
A: Traditional chiral resolution typically yields a maximum of 50% of the desired R-enantiomer, discarding the S-enantiomer. Racemization allows the 'waste' S-isomer to be converted back into the racemate and re-resolved, theoretically allowing for near-quantitative conversion of raw materials into the desired API intermediate.
Q: Does this racemization method require expensive transition metal catalysts?
A: No, unlike previous methods that relied on palladium or iridium complexes, this patented process utilizes inexpensive inorganic bases like potassium hydroxide or sodium alkoxides, significantly lowering the cost of goods and eliminating heavy metal contamination risks.
Q: What represents the primary advantage of this process for supply chain stability?
A: The process relies on commodity chemicals (bases and common solvents like DMSO) rather than specialized chiral ligands or precious metals, ensuring that raw material availability is not a bottleneck for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Larotrectinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising efficiencies of this racemization technology can be fully realized at an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral chromatography systems to guarantee that every batch of (2R)-2-(2,5-difluorophenyl)pyrrolidine meets the exacting standards required for oncology drug synthesis. Our commitment to quality ensures that the benefits of this novel process are delivered consistently, batch after batch.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic benefits of switching to this metal-free, recycling-enabled process for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that balance cost, quality, and supply security in the competitive landscape of TRK inhibitor production.
