Advanced Manufacturing of High-Purity Glycopyrronium Bromide via Novel Triazine Activation
Introduction to Next-Generation Glycopyrronium Bromide Synthesis
The pharmaceutical landscape for antimuscarinic agents has long been dominated by Glycopyrronium Bromide, a critical compound utilized extensively in preanesthetic medication and the treatment of gastrointestinal disorders. However, the historical manufacturing pathways for this essential API have been plagued by significant safety hazards and environmental inefficiencies. Patent CN102627595A introduces a transformative synthetic methodology that fundamentally reengineers the production workflow, shifting away from dangerous pyrophoric reagents toward a controlled, triazine-mediated activation strategy. This innovation addresses the urgent industry demand for safer, greener, and more cost-effective manufacturing protocols that do not compromise on the stringent purity requirements necessary for injectable formulations. By leveraging a novel intermediate activation step, this technology ensures consistent high-quality output while mitigating the operational risks associated with legacy processes.
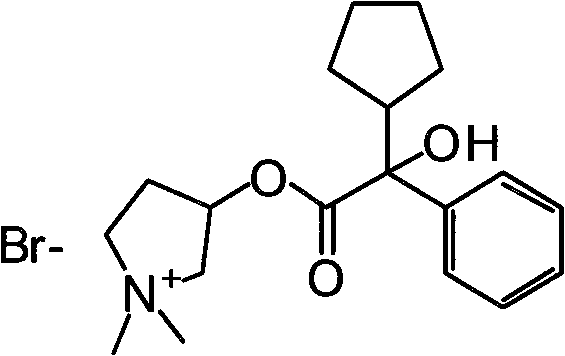
Furthermore, the strategic implementation of this synthesis route allows manufacturers to bypass the regulatory and logistical bottlenecks associated with handling highly toxic methylating agents and volatile hydrocarbon solvents. The core of this technological breakthrough lies in the precise activation of the carboxylic acid precursor, which facilitates a smoother transesterification reaction with the pyrrolidine moiety. For R&D directors and process chemists, this represents a significant opportunity to optimize impurity profiles and reduce the burden on downstream purification units. As we delve deeper into the technical specifics, it becomes evident that this patent offers a robust framework for the reliable supply of high-purity pharmaceutical intermediates, aligning perfectly with modern Good Manufacturing Practice (GMP) standards and sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Glycopyrronium Bromide has relied heavily on methodologies described in legacy patents such as US 2956062 and various academic publications from the mid-20th century. These conventional approaches typically necessitate the use of metallic sodium dispersed in low-molecular-weight hydrocarbons like n-heptane to drive the transesterification reaction. This reliance on pyrophoric materials introduces severe safety liabilities, including the risk of spontaneous combustion and explosive hazards during the reaction phase, thereby complicating the engineering controls required for large-scale manufacturing facilities. Additionally, alternative routes disclosed in documents like US 2009/0005577 often employ dimethyl sulfate as a methylating agent, a substance known for its extreme toxicity and carcinogenic potential, which imposes heavy burdens on waste treatment systems and worker safety protocols. The cumulative effect of these hazardous reagents is a process that is not only dangerous to operate but also generates substantial volumes of toxic waste, leading to inflated operational expenditures and complex environmental compliance challenges.
The Novel Approach
In stark contrast to these perilous legacy methods, the novel approach detailed in the patent utilizes a sophisticated activation strategy centered around 4-chloro-2,6-dimethoxy-triazine. This reagent serves as a potent coupling agent that activates the carboxylic acid group of the mandelic acid derivative under mild conditions, eliminating the need for aggressive bases or dangerous metals. The reaction proceeds smoothly in common organic solvents such as toluene or isopropanol at temperatures ranging from ambient to a moderate 60°C, significantly lowering the energy footprint of the process. By avoiding the use of sodium metal and dimethyl sulfate, this method inherently enhances the safety profile of the manufacturing plant, allowing for simpler reactor designs and reduced insurance premiums. Moreover, the streamlined workflow results in a dramatic improvement in overall yield, with experimental data demonstrating conversion rates nearing 90%, which directly translates to superior atom economy and reduced raw material consumption per kilogram of finished API.
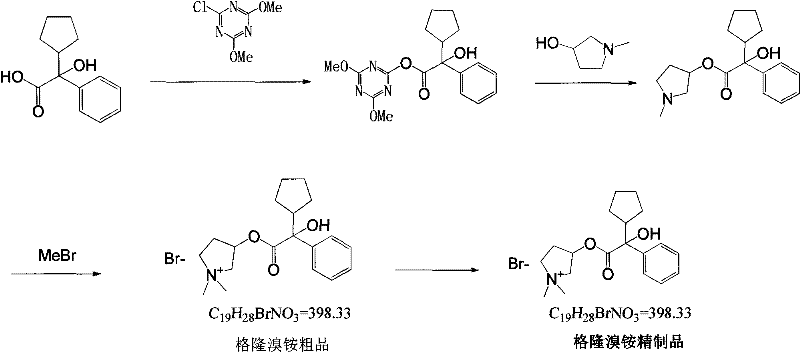
Mechanistic Insights into Triazine-Mediated Esterification
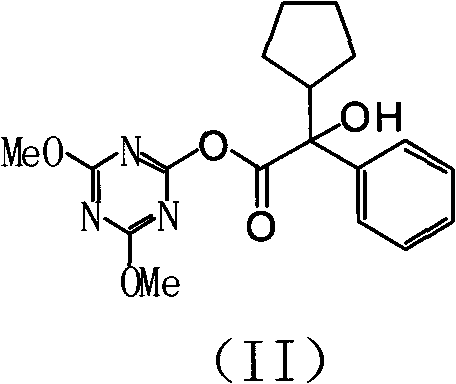
The cornerstone of this advanced synthesis is the formation of a highly reactive mixed anhydride-like intermediate, specifically the 4-chloro-2,6-dimethoxy-1,3,5-triazine activated ester. When alpha-cyclopentyl racemic mandelic acid reacts with the triazine derivative in an organic solvent, the carboxylic acid oxygen attacks the electron-deficient carbon of the triazine ring, displacing the chloride ion and forming the activated species designated as Formula II in the patent documentation. This activation step is crucial because it renders the carbonyl carbon significantly more electrophilic, thereby facilitating a rapid and selective nucleophilic attack by the hydroxyl group of 1-methyl-3-pyrrolidinol. Unlike traditional base-catalyzed transesterification which can lead to racemization or side reactions due to harsh conditions, this mild activation preserves the stereochemical integrity of the molecule while ensuring high conversion efficiency. The result is a clean reaction profile with minimal byproduct formation, simplifying the subsequent isolation and purification stages.
Following the formation of the ester linkage, the final quaternization step involves the introduction of methyl bromide to the tertiary amine nitrogen of the pyrrolidine ring. This alkylation transforms the neutral ester into the cationic quaternary ammonium salt, which is the pharmacologically active form of Glycopyrronium Bromide. The patent specifies that this step can be conducted directly in the reaction mixture after cooling, further streamlining the process by avoiding intermediate isolation. The mechanistic elegance of this route lies in its orthogonality; the activation of the acid and the subsequent quaternization occur without interfering with each other, allowing for a telescoped process that minimizes solvent usage and processing time. For process chemists, understanding this mechanism is vital for troubleshooting and optimization, as it highlights the importance of maintaining anhydrous conditions during the activation phase to prevent hydrolysis of the sensitive triazine intermediate.
How to Synthesize Glycopyrronium Bromide Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control to maximize the benefits of the triazine activation strategy. The process begins with the dissolution of the mandelic acid derivative in a dry organic solvent, followed by the slow addition of the triazine coupling agent to generate the active intermediate in situ. Once the activation is complete, the pyrrolidinol component is introduced, and the mixture is gently heated to promote the esterification. The final step involves the controlled addition of methyl bromide, a gas that must be handled with appropriate containment systems, to effect the quaternization.
- Activate alpha-cyclopentyl mandelic acid using 4-chloro-2,6-dimethoxy-triazine in an organic solvent like toluene at room temperature to form the active intermediate.
- React the activated intermediate solution with 1-methyl-3-pyrrolidinol in isopropanol, heating gently to 60°C to facilitate transesterification.
- Perform quaternization by adding methyl bromide to the cooled reaction mixture, followed by warming and crystallization to isolate the final bromide salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers profound strategic advantages that extend far beyond simple chemical yield improvements. By eliminating the requirement for metallic sodium and dimethyl sulfate, the manufacturing process removes two of the most significant cost drivers associated with hazardous material handling, storage, and disposal. The removal of these dangerous reagents drastically simplifies the logistics of raw material sourcing, as the new inputs are stable, commercially available commodities that do not require specialized transport or high-security storage facilities. This shift not only reduces the direct cost of goods sold but also mitigates the risk of supply chain disruptions caused by regulatory crackdowns on toxic chemicals or safety incidents at supplier sites. Consequently, this technology enables a more resilient and predictable supply chain for this critical pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The transition to a triazine-mediated pathway eliminates the need for expensive safety infrastructure required for pyrophoric materials, leading to substantial capital expenditure savings. Furthermore, the high yield and selectivity of the reaction minimize the generation of waste streams, significantly lowering the operational costs associated with effluent treatment and environmental compliance. The simplified workup procedure, which relies on straightforward filtration and drying rather than complex chromatographic separations, reduces both solvent consumption and labor hours per batch. These factors combine to create a highly cost-competitive manufacturing model that delivers significant margin improvements without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous raw materials ensures a consistent and uninterrupted flow of inputs, shielding production schedules from the volatility often associated with regulated toxic substances. The robustness of the reaction conditions means that the process is less susceptible to batch failures caused by minor variations in temperature or moisture, thereby enhancing overall equipment effectiveness and throughput. This reliability is critical for maintaining continuous supply to downstream formulation partners, ensuring that market demand for Glycopyrronium Bromide injections and tablets is met without delay. The ability to scale this process from pilot to commercial tonnage with minimal re-engineering further solidifies its position as a dependable source for long-term supply agreements.
- Scalability and Environmental Compliance: From an environmental perspective, this method represents a significant leap forward in green chemistry principles by reducing the E-factor of the synthesis. The absence of heavy metal catalysts and toxic alkylating agents simplifies the waste profile, making it easier to treat and dispose of byproducts in accordance with increasingly stringent global environmental regulations. The process is inherently scalable, as the exothermic nature of the reactions is manageable even in large vessels, allowing for seamless expansion of production capacity to meet growing market needs. This alignment with sustainability goals not only future-proofs the manufacturing asset but also enhances the corporate social responsibility profile of the organization, appealing to environmentally conscious stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and feasibility of the triazine-mediated route for potential manufacturing partners. Understanding these details is essential for evaluating the fit of this technology within existing production portfolios and supply strategies.
Q: How does the triazine-mediated route improve safety compared to traditional sodium metal methods?
A: Traditional methods often utilize metallic sodium in flammable hydrocarbons, posing significant fire and explosion risks. The novel triazine-mediated route operates under mild conditions without pyrophoric reagents, drastically reducing operational hazards and EHS compliance costs.
Q: What is the expected purity and yield profile of this synthesis method?
A: The patented process demonstrates exceptional efficiency, with experimental data indicating yields approaching 90%. The resulting product exhibits a sharp melting point range of 195°C to 198°C, confirming high chemical purity suitable for clinical injection grades.
Q: Is this manufacturing process scalable for commercial API production?
A: Yes, the process utilizes readily available raw materials and avoids complex purification steps like extensive chromatography. The simple workup involving filtration and vacuum drying makes it highly amenable to multi-ton scale-up in standard stainless steel reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glycopyrronium Bromide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthesis routes to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering Glycopyrronium Bromide that meets stringent purity specifications, utilizing rigorous QC labs to verify that every batch conforms to the highest international pharmacopoeia standards. Our expertise in handling complex quaternary ammonium salts allows us to optimize the crystallization and drying parameters, guaranteeing a product with the correct polymorphic form and particle size distribution for optimal bioavailability.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this superior manufacturing technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this safer, high-yield process for your operations. We encourage you to contact us today to discuss your specific requirements, obtain specific COA data for our reference batches, and review detailed route feasibility assessments tailored to your production capabilities. Together, we can redefine the standards for quality and efficiency in the production of this vital antimuscarinic agent.
