Advanced Metal-Free Synthesis of 2-Iodo Aryl Ethers for Commercial Pharmaceutical Production
Introduction to Next-Generation 2-Iodo Aryl Ether Synthesis
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for greener, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is detailed in Chinese patent CN112979400A, published in June 2021, which discloses a novel method for preparing 2-iodo aryl ethers under the action of alkali metal hydrides. This technology represents a paradigm shift from traditional transition-metal-catalyzed cross-coupling reactions, offering a streamlined pathway to access these critical building blocks. 2-Iodo aryl ethers are indispensable precursors in the construction of complex oxygen-containing heterocycles and serve as versatile handles for further ortho-substituted aryl ether derivatives via various downstream conversions. The ability to synthesize these compounds without the burden of expensive catalysts or harsh thermal conditions addresses long-standing pain points in process chemistry, particularly for large-scale manufacturing where cost and purity are paramount.
The core innovation lies in the utilization of readily available alkali metal hydrides, such as sodium hydride or potassium hydride, to facilitate the coupling between 1,2-diiodoarenes and phenols. This approach not only simplifies the operational procedure but also drastically reduces the environmental footprint associated with heavy metal waste disposal. For R&D directors and process chemists, this patent offers a robust alternative to conventional methods, promising high yields and exceptional purity profiles. The reaction operates effectively at temperatures ranging from 0 to 100 degrees Celsius, with a strong preference for room temperature conditions, thereby minimizing energy consumption. As we delve deeper into the technical specifics, it becomes evident that this methodology is not merely an academic curiosity but a commercially viable solution for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the formation of carbon-oxygen bonds to create aryl ethers has relied heavily on transition metal catalysis, such as copper-mediated Ullmann condensations or palladium-catalyzed Buchwald-Hartwig couplings. While effective, these traditional pathways impose significant constraints on industrial scalability and cost efficiency. The requirement for noble metal catalysts like palladium introduces substantial raw material costs, which are subject to volatile market fluctuations. Furthermore, the presence of these metals necessitates rigorous purification steps to meet stringent regulatory limits for residual metals in active pharmaceutical ingredients (APIs). Removing trace amounts of palladium or copper often requires specialized scavengers or multiple chromatographic passes, which complicates the workflow and reduces overall throughput. Additionally, many of these metal-catalyzed reactions demand elevated temperatures and inert atmospheres, increasing energy expenditures and operational complexity. The use of complex ligands to stabilize the metal center further adds to the cost and potential impurity profile, creating a bottleneck for manufacturers seeking to optimize their supply chains for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast to the metal-dependent status quo, the method disclosed in CN112979400A leverages the intrinsic reactivity of 1,2-diiodoarenes activated by alkali metal hydrides. This transition-metal-free protocol allows for the direct coupling of phenols with diiodo-substituted aromatics under remarkably mild conditions. By eliminating the need for exogenous metal catalysts, the process inherently avoids metal pollution, resulting in a cleaner crude product that requires less intensive downstream processing. The reaction is typically conducted in a solvent system comprising tetrahydrofuran (THF) and dimethylacetamide (DMA), which provides an optimal medium for the solubility of both the inorganic base and the organic substrates. The operational simplicity is further enhanced by the fact that the reaction can proceed at room temperature, removing the need for energy-intensive heating cycles. This novel approach not only accelerates the reaction kinetics but also expands the scope of compatible functional groups, enabling the synthesis of diverse 2-iodo aryl ether derivatives that might be unstable under harsher thermal conditions required by older methods.
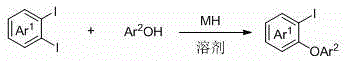
Mechanistic Insights into Alkali Metal Hydride Mediated Coupling
The mechanistic underpinning of this transformation is believed to involve the generation of a highly reactive aryne intermediate, facilitated by the elimination of hydrogen iodide from the 1,2-diiodoarene substrate upon treatment with the strong base. The alkali metal hydride, preferably sodium hydride, acts as a potent deprotonating agent that initiates the sequence by abstracting a proton or facilitating the elimination of the ortho-iodine atom. This generates a transient benzyne species which is then rapidly intercepted by the nucleophilic phenoxide anion, formed in situ from the reaction of the phenol with the hydride base. This addition-elimination pathway ensures high regioselectivity and efficiency, leading to the exclusive formation of the 2-iodo aryl ether product without significant over-coupling by-products. The absence of a metal cycle means there are no oxidative addition or reductive elimination steps that typically limit turnover numbers in catalytic systems; instead, the stoichiometric use of the hydride drives the reaction to completion through thermodynamic favorability. This mechanism is particularly advantageous for substrates containing electron-withdrawing groups, which can stabilize the transition states involved in the nucleophilic attack, thereby enhancing reaction rates and yields.
From an impurity control perspective, the metal-free nature of this chemistry is a game-changer for quality assurance teams. In traditional catalytic processes, metal residues can catalyze unwanted side reactions during storage or subsequent synthetic steps, leading to degradation products that are difficult to remove. By completely bypassing the use of transition metals, this method ensures that the impurity profile is dominated by organic by-products that are generally easier to separate via standard crystallization or extraction techniques. The high functional group compatibility observed in the patent examples, including tolerance towards nitro, cyano, and methoxy groups, underscores the mildness of the reaction environment. This selectivity minimizes the formation of structural analogs or decomposition products, thereby simplifying the purification strategy. For procurement managers, this translates to a more predictable and consistent supply of high-purity 2-iodo aryl ether, reducing the risk of batch failures due to out-of-specification metal content or complex impurity profiles that plague metal-catalyzed routes.
How to Synthesize 2-Iodo Aryl Ether Efficiently
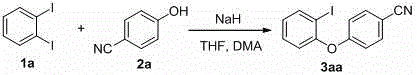
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the handling of alkali metal hydrides, which are moisture-sensitive reagents. The general protocol involves suspending the hydride in a dry solvent, followed by the sequential addition of the phenol and the diiodoarene. The reaction progress can be monitored via standard analytical techniques such as TLC or HPLC, with typical completion times ranging from thirty minutes to two hours at ambient temperature. The workup procedure is straightforward, involving quenching with water, extraction with organic solvents like ethyl acetate, and purification via column chromatography or recrystallization. This streamlined process significantly reduces the technical barrier for adoption, making it accessible for both small-scale discovery chemistry and larger-scale production campaigns. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized synthesis steps outlined below.
- Suspend alkali metal hydride (preferably sodium hydride) in a solvent mixture such as THF and DMA under standard stirring conditions.
- Add the phenol substrate to the suspension and stir for approximately 10 minutes to ensure complete deprotonation and phenoxide formation.
- Introduce the 1,2-diiodoarene solution to the reaction mixture and maintain stirring at room temperature for 0.5 to 2 hours until conversion is complete.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this alkali metal hydride-mediated synthesis offers profound commercial advantages that resonate deeply with procurement and supply chain stakeholders. By shifting away from precious metal catalysts, manufacturers can achieve substantial cost savings on raw materials, as sodium hydride is significantly cheaper and more abundant than palladium or copper complexes. This reduction in material costs is compounded by the elimination of expensive metal scavenging resins and the associated waste disposal fees, leading to a leaner and more economical production model. Furthermore, the ability to run reactions at room temperature drastically cuts down on energy consumption, contributing to lower utility bills and a smaller carbon footprint. These factors collectively enhance the overall cost competitiveness of the final intermediate, allowing suppliers to offer more attractive pricing structures to their downstream clients in the pharmaceutical and agrochemical sectors.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a major cost driver from the bill of materials. Precious metals like palladium are not only expensive to purchase but also incur high costs for recovery and disposal to meet environmental regulations. By utilizing inexpensive alkali metal hydrides, the process achieves a drastic simplification of the cost structure. Additionally, the mild reaction conditions reduce the need for specialized high-temperature reactors and extensive cooling systems, further lowering capital and operational expenditures. This economic efficiency allows for better margin management and the ability to absorb fluctuations in other raw material prices without passing excessive costs to the customer.
- Enhanced Supply Chain Reliability: Reliance on scarce transition metals can introduce supply chain vulnerabilities, especially during periods of geopolitical instability or mining disruptions. Sodium hydride and the requisite diiodoarene precursors are commodity chemicals with robust and diversified global supply chains, ensuring consistent availability. The simplified process also reduces the lead time for production, as there are no lengthy catalyst screening phases or complex metal removal steps that can delay batch release. This reliability is crucial for maintaining continuous manufacturing schedules and meeting tight delivery deadlines for critical API intermediates, thereby strengthening the partnership between suppliers and pharmaceutical companies.
- Scalability and Environmental Compliance: Scaling up metal-free reactions is inherently safer and more straightforward than managing large volumes of toxic metal catalysts. The absence of heavy metals simplifies the regulatory approval process for new drug applications, as the burden of proving low residual metal levels is removed. Moreover, the waste streams generated are less hazardous, facilitating easier treatment and compliance with increasingly strict environmental protection laws. The process generates sodium iodide as a benign by-product, which is easy to handle and dispose of compared to heavy metal sludge. This alignment with green chemistry principles enhances the sustainability profile of the manufacturing site, appealing to environmentally conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing a reliable foundation for decision-making. Understanding these nuances helps R&D and procurement teams evaluate the feasibility of integrating this technology into their existing workflows. The clarity provided here aims to mitigate risks associated with technology transfer and ensure a smooth transition from laboratory scale to commercial production.
Q: Does this synthesis method require transition metal catalysts?
A: No, the method described in patent CN112979400A operates without any transition metal catalysts, utilizing alkali metal hydrides instead, which eliminates the risk of heavy metal contamination in the final product.
Q: What are the optimal reaction conditions for this coupling?
A: The reaction proceeds efficiently at room temperature (0-100°C range, preferably ambient) using a solvent system of THF and DMA, with reaction times typically ranging from 0.5 to 2 hours depending on the substrate.
Q: Is this method suitable for substrates with sensitive functional groups?
A: Yes, the mild conditions and lack of aggressive metal catalysts provide high functional group compatibility, allowing for the successful synthesis of derivatives containing nitro, cyano, and other sensitive groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Iodo Aryl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis technology described in CN112979400A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-iodo aryl ether meets the highest industry standards. We are committed to leveraging this advanced chemistry to deliver superior products that accelerate our partners' drug development timelines while maintaining the utmost quality and safety.
We invite you to collaborate with us to explore how this cost-effective and sustainable synthesis route can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic benefits of switching to this metal-free protocol. Please contact us today to request specific COA data for our catalog items or to discuss route feasibility assessments for your custom synthesis projects. Together, we can drive innovation and efficiency in the global supply chain for fine chemical intermediates.
