Advanced [3+2] Cycloaddition Strategy for Scalable Benzoxazolo[3,2-c]oxazole Intermediates
Introduction to Next-Generation Benzoxazole Synthesis
The pharmaceutical industry continuously demands efficient routes to complex heterocyclic scaffolds, particularly those exhibiting significant biological activity such as anti-cancer, anti-viral, and anti-hypertensive properties. Patent CN109485661B introduces a groundbreaking methodology for the synthesis of benzoxazolo[3,2-c]oxazole compounds through a highly selective [3+2] cycloaddition reaction. This innovation addresses critical bottlenecks in constructing hydrogenated polycyclic benzoxazole backbones, which are prevalent in numerous natural products and active pharmaceutical ingredients. By leveraging substituted benzothiazoles and donor-acceptor (D-A) propylene oxides as primary building blocks, this process achieves remarkable atom economy and structural complexity in a single operational step.
The core of this technological advancement lies in the utilization of specific Lewis acid catalysts, most notably Scandium triflate (Sc(OTf)3), combined with molecular sieve additives to drive the dearomatization and cyclization sequence. This approach not only simplifies the synthetic pathway but also ensures exceptional control over stereochemistry, yielding products with two distinct chiral centers and high diastereoselectivity. For R&D teams focused on rapid library generation and process chemists aiming for robust manufacturing protocols, this patent represents a significant leap forward in heterocyclic chemistry.
![General reaction scheme for [3+2] cycloaddition of benzothiazoles and D-A epoxides](/insights/img/benzoxazolo-oxazole-synthesis-lewis-acid-pharma-supplier-20260305092822-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of hydrogenated benzoxazole frameworks has been fraught with challenges related to multi-step sequences, harsh reaction conditions, and poor stereocontrol. Conventional dearomatization strategies often necessitate the use of stoichiometric amounts of expensive oxidants or transition metal catalysts that are difficult to remove from the final API, posing significant regulatory hurdles for pharmaceutical manufacturers. Furthermore, existing methods frequently suffer from low chemical selectivity, leading to complex mixtures of regioisomers and diastereomers that require tedious and yield-eroding purification processes. The inability to efficiently introduce diverse functional groups at specific positions on the heterocyclic core has historically limited the exploration of structure-activity relationships (SAR) in drug discovery campaigns targeting benzoxazole-based therapeutics.
The Novel Approach
In stark contrast, the novel [3+2] cycloaddition strategy detailed in the patent data offers a streamlined, one-pot solution that bypasses these traditional inefficiencies. By employing a catalytic amount of Lewis acid, specifically Sc(OTf)3, the reaction proceeds under mild conditions, typically at room temperature or slightly elevated temperatures up to 60°C, thereby minimizing thermal degradation of sensitive functional groups. The integration of 4Å molecular sieves plays a crucial role in sequestering moisture and driving the equilibrium towards product formation, ensuring consistently high yields ranging from 40% to an impressive 96% across a broad substrate scope. This methodology transforms the synthesis of these complex architectures from a laborious multi-step endeavor into a highly efficient, scalable process suitable for industrial application.
Mechanistic Insights into Sc(OTf)3-Catalyzed Cycloaddition
The mechanistic elegance of this transformation relies on the potent Lewis acidity of the Scandium center, which coordinates with the oxygen atoms of the donor-acceptor epoxide. This coordination significantly enhances the electrophilicity of the epoxide ring, facilitating a nucleophilic attack by the nitrogen atom of the benzothiazole substrate. This initial interaction triggers a cascade of bond reorganizations, including ring-opening of the epoxide and subsequent intramolecular cyclization, ultimately restoring the aromaticity of the benzene ring while forming the new oxazole fused system. The presence of the electron-withdrawing ester groups on the epoxide further stabilizes the developing negative charge during the transition state, lowering the activation energy and accelerating the reaction kinetics without the need for extreme thermal input.
From an impurity control perspective, the high diastereoselectivity observed (often exceeding a 20:1 ratio) is attributed to the rigid transition state imposed by the catalyst-substrate complex. This stereochemical fidelity is paramount for pharmaceutical manufacturing, as it drastically reduces the burden of chiral separation and ensures that the final product meets stringent enantiomeric purity specifications required by regulatory bodies. Furthermore, the compatibility of this catalytic system with various substituents, including halogens and nitro groups, demonstrates its robustness against electronic variations, making it a versatile tool for synthesizing diverse analogues for SAR studies.
How to Synthesize Benzoxazolo[3,2-c]oxazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters identified during the patent's development phase. The protocol emphasizes the importance of anhydrous conditions and the precise loading of the catalyst to maximize turnover frequency while minimizing metal residue. The following section outlines the generalized procedure derived from the exemplary data, serving as a foundational guide for process replication and scale-up activities.
- Prepare the reaction vessel by adding Sc(OTf)3 catalyst (10-20 mol%) and activated 4Å molecular sieves under an inert nitrogen atmosphere.
- Dissolve the substituted benzothiazole and diethyl phenyloxirane dicarboxylate in 1,2-dichloroethane (DCE) and add the solutions to the catalyst mixture at room temperature.
- Stir the reaction mixture at room temperature or 60°C for 48-72 hours, then purify the crude product via preparative thin layer chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Sc(OTf)3-catalyzed route presents compelling economic and logistical advantages over legacy synthetic pathways. The shift towards a single-step cycloaddition significantly compresses the manufacturing timeline, reducing the number of unit operations and the associated labor and utility costs. By eliminating the need for precious metal catalysts like palladium or platinum, which are subject to volatile market pricing and supply chain disruptions, this method utilizes earth-abundant scandium salts that offer a more stable and predictable cost structure. Additionally, the high yields and selectivity minimize raw material waste, directly contributing to a lower cost of goods sold (COGS) and a reduced environmental footprint.
- Cost Reduction in Manufacturing: The elimination of multi-step protection and deprotection sequences, which are common in traditional heterocycle synthesis, leads to substantial savings in reagent consumption and solvent usage. The ability to run the reaction at ambient or near-ambient temperatures further reduces energy expenditures associated with heating and cooling large-scale reactors. Moreover, the simplified workup procedure, primarily involving filtration of molecular sieves and standard chromatography, lowers the operational complexity and equipment maintenance costs, resulting in a more lean and cost-effective production model.
- Enhanced Supply Chain Reliability: The starting materials, specifically substituted benzothiazoles and donor-acceptor epoxides, are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions allows for flexibility in sourcing raw materials with varying grades, as the catalytic system tolerates minor impurities without significant loss in performance. This resilience ensures consistent production schedules and reliable delivery timelines for downstream API manufacturers, safeguarding against potential supply disruptions.
- Scalability and Environmental Compliance: The use of 1,2-dichloroethane (DCE) as a solvent, while requiring careful handling, is well-established in industrial settings with existing recovery and recycling infrastructure. The high atom economy of the [3+2] cycloaddition minimizes the generation of hazardous byproducts, simplifying waste treatment and disposal protocols. As the process scales from kilogram to tonnage levels, the linear relationship between catalyst loading and conversion efficiency suggests that the method can be readily adapted for continuous flow chemistry, further enhancing throughput and safety profiles in a GMP environment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating this route for their specific projects.
Q: What is the optimal catalyst for this [3+2] cycloaddition?
A: According to patent CN109485661B, Scandium triflate (Sc(OTf)3) demonstrates superior performance compared to other Lewis acids like Yb(OTf)3 or Ni(ClO4)2, providing yields up to 96% with excellent diastereoselectivity.
Q: Can the ester groups in the product be further modified?
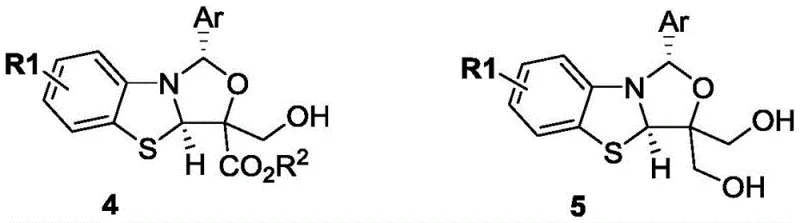
A: Yes, the resulting benzoxazolo[3,2-c]oxazole compounds containing ester groups can be selectively reduced using agents like NaBH4 to produce mono- or bis-hydroxymethyl derivatives, expanding their utility in drug discovery.
Q: Is this process suitable for substrates with electron-withdrawing groups?
A: The methodology is highly robust and tolerates various substituents including halogens (Cl, Br), nitro groups, and alkyl groups on the benzothiazole ring, maintaining high chemical selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxazolo[3,2-c]oxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates for the development of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and assay every batch.
We invite you to collaborate with us to leverage this advanced [3+2] cycloaddition technology for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your project timelines and optimize your supply chain economics.
