Advanced One-Pot Synthesis of Chiral Cyclopentenone Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex chiral scaffolds, particularly those serving as critical precursors for bioactive natural products and active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in Chinese Patent CN109180406B, which discloses a highly efficient synthesis method for (4R*, 5R*)-5-benzyl-4-hydroxy-2-cyclopentene-1-ketone compounds. This technology represents a paradigm shift from traditional multi-step sequences to a streamlined one-pot multi-step tandem reaction, addressing long-standing challenges in stereoselectivity and operational complexity. The core innovation lies in the direct transformation of simple allenoic alcohols and iodo-aromatics into valuable cyclopentenone derivatives through a palladium-catalyzed carbonylative cyclization. By leveraging carbon monoxide insertion and intramolecular cyclization in a single vessel, this method not only enhances atom economy but also drastically reduces the environmental footprint associated with intermediate isolation and purification steps. For R&D directors and process chemists, this patent offers a compelling route to access high-purity intermediates with defined stereochemistry, essential for downstream drug development.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 4-hydroxy-2-cyclopenten-1-one core has relied on several classical strategies, each fraught with significant limitations that hinder large-scale manufacturing. Traditional approaches often involve the cycloaddition reaction of 1,2-diketones with 2,3-allenoic acid esters, which frequently requires harsh reaction conditions and specialized reagents that are not readily available commercially. Another common pathway involves the intramolecular coupling-desilylation reaction of transition metal catalyzed 3-(triethylsiloxy)-5-(tributyltin)-4(Z)-thiopentenoic acid ethyl ester; however, the use of organotin reagents poses severe toxicity and environmental disposal challenges, making it undesirable for green chemistry initiatives. Furthermore, condensation reactions catalyzed by N-heterocyclic carbenes often struggle with controlling the stereochemical outcome, leading to the easy formation of R/S configuration mixtures that require costly and yield-reducing chiral separation processes. These inherent drawbacks, including complicated reaction steps, difficult raw material preparation, and poor stereoselectivity, greatly limit the practical utility of existing methods for producing high-value pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN109180406B introduces a novel economical and practical synthesis route that directly obtains the target (4R, 5R)-5-benzyl-4-hydroxy-2-cyclopentene-1-ketone compounds through a one-pot series reaction. This approach utilizes simple and easily prepared raw materials, specifically 1-phenyl-4,5-hexadiene-3-alcohol and various iodo aromatic compounds, which are significantly more accessible than the specialized precursors required by older methods. The reaction proceeds under remarkably mild conditions, typically maintained below 100°C, which minimizes thermal degradation of sensitive functional groups and reduces energy consumption during the manufacturing process. Most critically, this new method demonstrates high product configuration selectivity, effectively suppressing the formation of unwanted stereoisomers and thereby simplifying the downstream purification workflow. The operational simplicity of a one-pot process means fewer unit operations, less solvent usage, and a reduced overall processing time, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The success of this transformation hinges on a sophisticated palladium-catalyzed mechanism that orchestrates multiple bond-forming events in a single sequence. The catalytic cycle initiates with the oxidative addition of the iodo aromatic compound to the active palladium(0) species generated in situ from the palladium acetate precursor and the phosphine ligand. Following this activation, carbon monoxide inserts into the palladium-carbon bond to form an acyl-palladium intermediate, a crucial step that introduces the carbonyl functionality into the final ring structure. Subsequently, the allenoic alcohol substrate coordinates to the metal center, facilitating an intramolecular nucleophilic attack or migratory insertion that closes the five-membered ring. The presence of the base, such as triethylamine, plays a vital role in neutralizing the hydrogen iodide byproduct and regenerating the active catalyst species, ensuring the turnover of the catalytic cycle. The choice of ligand, particularly trifuranylphosphine, is instrumental in modulating the electronic properties of the palladium center, enhancing both the rate of oxidative addition and the stability of the intermediate complexes.
From an impurity control perspective, the high stereoselectivity observed in this reaction is attributed to the specific coordination geometry enforced by the chiral or sterically demanding environment around the palladium center during the cyclization step. The reaction conditions are finely tuned to favor the formation of the (4R, 5R) diastereomer, minimizing the generation of the (4S, 5S) or meso counterparts that would otherwise complicate the purity profile. By maintaining the reaction temperature at 80°C and utilizing acetonitrile as the solvent, the system achieves an optimal balance between reaction kinetics and thermodynamic control, preventing side reactions such as polymerization of the allene moiety or over-carbonylation. This precise control over the reaction pathway ensures that the final crude product contains a minimal burden of structural impurities, reducing the load on subsequent chromatographic purification steps and improving the overall mass balance of the process.
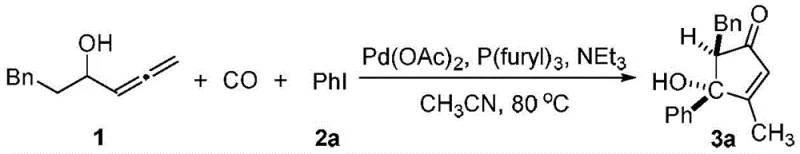
How to Synthesize (4R,5R)-5-Benzyl-4-hydroxy-2-cyclopenten-1-ketone Efficiently
To implement this synthesis effectively, process chemists should adhere to the optimized parameters outlined in the patent examples, which demonstrate the robustness of the method across various substrates. The standard protocol involves charging a reaction vessel with the allenoic alcohol substrate and the iodoarene in a polar aprotic solvent, followed by the sequential addition of the palladium catalyst, phosphine ligand, and organic base. The reaction is then conducted under a carbon monoxide atmosphere, typically at 1 atm pressure, ensuring sufficient CO concentration for the insertion step without requiring high-pressure equipment. As illustrated in the specific example below, the use of trifuranylphosphine as the ligand and triethylamine as the base yields superior results compared to other combinations, highlighting the importance of ligand selection in maximizing conversion and selectivity.
- Dissolve 1-phenyl-4,5-hexadiene-3-alcohol and an iodo aromatic compound in acetonitrile solvent within a reaction flask.
- Add palladium acetate catalyst, trifuranylphosphine ligand, and triethylamine base to the mixture under a carbon monoxide atmosphere.
- Stir the reaction at 80°C for 8 hours, then quench with saturated ammonium chloride and extract with ethyl acetate to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology offers tangible strategic benefits that extend beyond mere technical feasibility. The shift towards a one-pot methodology fundamentally alters the cost structure of manufacturing by eliminating the need for isolating and purifying reactive intermediates, which traditionally accounts for a significant portion of production costs and time. The reliance on readily available starting materials, such as substituted iodobenzenes and simple allenoic alcohols, mitigates supply chain risks associated with sourcing exotic or custom-synthesized reagents, ensuring a more stable and continuous supply of raw materials. Furthermore, the mild reaction conditions reduce the demand for specialized high-temperature or high-pressure reactors, allowing for production in standard glass-lined or stainless steel vessels commonly found in multipurpose chemical plants.
- Cost Reduction in Manufacturing: The elimination of multiple isolation steps and the use of a catalytic amount of palladium significantly lower the operational expenditures associated with solvent consumption and waste treatment. By avoiding the use of toxic organotin reagents found in older methods, the facility saves substantially on hazardous waste disposal fees and environmental compliance costs. The high selectivity of the reaction reduces the loss of valuable material during purification, leading to improved overall yields and better utilization of raw materials. Additionally, the ability to run the reaction at atmospheric pressure with carbon monoxide avoids the capital expenditure required for high-pressure autoclaves, further reducing the barrier to entry for manufacturing this intermediate.
- Enhanced Supply Chain Reliability: The broad substrate scope of this reaction, which tolerates various functional groups including chloro, nitrile, and methyl substituents on the aromatic ring, allows for flexibility in sourcing raw materials from multiple vendors. This versatility ensures that supply chain disruptions for a specific substituted iodoarene can be managed by switching to alternative analogs without redesigning the entire synthetic route. The robustness of the catalytic system, which functions effectively with common bases like triethylamine and solvents like acetonitrile, means that procurement teams are not locked into single-source suppliers for specialized reagents. This flexibility enhances the resilience of the supply chain against market volatility and geopolitical uncertainties affecting chemical availability.
- Scalability and Environmental Compliance: The one-pot nature of the process inherently simplifies scale-up, as there are fewer transfer operations and unit processes that could introduce variability or safety hazards during batch enlargement. The reaction generates minimal hazardous byproducts, primarily relying on benign salts and organic solvents that can be recovered and recycled, aligning with modern green chemistry principles and regulatory expectations. The absence of heavy metal contaminants like tin in the final product streamlines the quality control process, reducing the need for extensive metal scavenging steps that can delay batch release. Consequently, this method supports a faster time-to-market for new drug candidates while maintaining a sustainable and compliant manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, aiming to clarify the operational nuances for potential partners. Understanding these details is crucial for assessing the feasibility of integrating this technology into existing production lines or new product development pipelines.
Q: What are the primary advantages of this synthesis method over conventional cycloaddition routes?
A: Unlike conventional methods that often suffer from difficult raw material preparation and the formation of R/S configuration mixtures, this novel one-pot process utilizes easily available starting materials and achieves high product configuration selectivity under mild conditions below 100°C.
Q: Which catalyst system provides the optimal yield for this transformation?
A: Experimental data indicates that the combination of Palladium acetate (Pd(OAc)2) as the catalyst and Trifuranylphosphine (P(furyl)3) as the ligand, specifically in a molar ratio of roughly 1:4 relative to the catalyst, provides superior yields compared to triphenylphosphine or other additives.
Q: Is this process scalable for industrial production of API intermediates?
A: Yes, the process is highly suitable for industrial scale-up due to its one-pot nature which simplifies operation, the use of common solvents like acetonitrile, and the ability to tolerate various substituted iodoarenes including those with chloro, nitrile, or methyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (4R,5R)-5-Benzyl-4-hydroxy-2-cyclopenten-1-ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and potency. Our expertise in palladium-catalyzed transformations allows us to optimize this specific synthesis route for maximum yield and minimal impurity formation, providing our clients with a reliable source of this complex cyclopentenone scaffold.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis technology for their specific projects. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
