Scalable Synthesis of Trifluoromethanesulfonyl Isoxazoles for Advanced Pharmaceutical Applications
Scalable Synthesis of Trifluoromethanesulfonyl Isoxazoles for Advanced Pharmaceutical Applications
The pharmaceutical and agrochemical industries are increasingly recognizing the critical value of fluorine-containing heterocyclic compounds, particularly those incorporating the trifluoromethanesulfonyl group due to its unique electron-withdrawing and lipophilic properties. Patent CN111196786B introduces a groundbreaking synthesis method for trifluoromethanesulfonyl-substituted isoxazoles, addressing significant limitations in existing manufacturing protocols. This innovation utilizes copper nitrate trihydrate as a nitrogen-oxygen source in butyl butyrate, enabling a direct, environmentally friendly cyclization of alkynes. The resulting compounds serve as vital intermediates for high-performance applications, including TRPV1 receptor antagonists and advanced material science components. By shifting away from harsh oxidants and complex multi-step sequences, this technology offers a robust pathway for producing high-purity pharmaceutical intermediates with enhanced structural diversity.
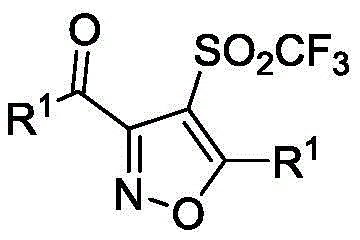
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of trifluoromethanesulfonyl groups into heterocyclic systems has relied on chemically aggressive and operationally complex strategies that hinder efficient commercial production. Traditional approaches often involve the oxidation of trifluoromethyl sulfides using meta-chloroperoxybenzoic acid (mCPBA), a process that generates substantial waste and requires careful handling of explosive peroxides. Alternatively, direct sulfonylation using triflic anhydride (Tf2O) typically necessitates cryogenic conditions and stoichiometric amounts of strong organolithium bases, posing severe safety risks and scalability challenges. Furthermore, Friedel-Crafts acylation strategies often require specific substrates and additives, limiting the scope of accessible derivatives. These conventional methods frequently suffer from low atom economy, harsh reaction environments, and difficult purification processes, making them less ideal for the cost-sensitive and safety-regulated landscape of modern fine chemical manufacturing.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN111196786B presents a streamlined, one-pot cyclization strategy that significantly simplifies the synthetic workflow. By employing copper nitrate trihydrate as a dual nitrogen and oxygen source, the reaction proceeds under mild thermal conditions (90-100°C) in an open air atmosphere, eliminating the need for inert gas protection or cryogenic cooling. The use of butyl butyrate as a solvent further enhances the environmental profile of the process, providing a stable medium that supports high conversion rates without the toxicity associated with chlorinated solvents. This approach not only tolerates a wide range of functional groups on the aromatic rings, such as halogens, alkyl, and alkoxy substituents, but also delivers consistent yields across diverse substrate classes. The operational simplicity and reduced hazard profile make this novel route exceptionally attractive for reliable pharmaceutical intermediate supplier networks aiming to optimize their production capabilities.
Mechanistic Insights into Copper-Catalyzed Cyclization
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the copper catalyst, which orchestrates the simultaneous construction of the isoxazole ring and the incorporation of the sulfonyl functionality. The reaction initiates with the coordination of the copper species to the alkyne moiety of the trifluoromethanesulfonyl-substituted starting material, activating the triple bond towards nucleophilic attack. Subsequent interaction with the nitrate anion provides the necessary nitrogen and oxygen atoms to close the heterocyclic ring, a process that is thermodynamically driven by the formation of the stable isoxazole core. This intramolecular cyclization avoids the formation of unstable intermediates often seen in stepwise syntheses, thereby minimizing side reactions and impurity generation. The catalytic cycle is sustained by the regeneration of the active copper species, ensuring efficient turnover throughout the reaction duration.
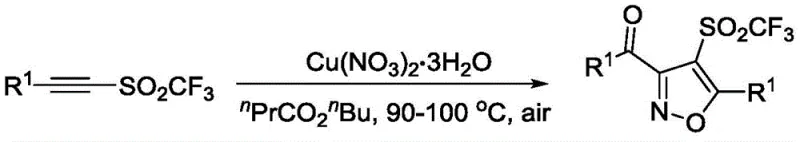
From an impurity control perspective, this mechanism offers distinct advantages by reducing the reliance on external oxidants that often lead to over-oxidation byproducts. The mild oxidative potential of the nitrate source under thermal conditions ensures selective transformation of the alkyne without degrading sensitive functional groups on the aryl rings, such as bromo or iodo substituents which are crucial for downstream cross-coupling reactions. Furthermore, the absence of strong bases prevents base-mediated decomposition or polymerization of the reactive alkyne precursors. The result is a cleaner crude reaction profile that simplifies downstream processing, requiring less aggressive purification steps to achieve the stringent purity specifications demanded by regulatory bodies. This level of control over the reaction trajectory is essential for maintaining batch-to-batch consistency in the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Trifluoromethanesulfonyl Isoxazoles Efficiently
Implementing this synthesis protocol requires precise adherence to the optimized reaction parameters to maximize yield and purity while ensuring operational safety. The process begins with the accurate weighing of the trifluoromethanesulfonyl alkyne precursor and copper nitrate trihydrate, maintaining a specific molar ratio to drive the reaction to completion. The choice of butyl butyrate as the solvent is critical, as it provides the necessary boiling point range to sustain the reaction temperature of 90-100°C without excessive pressure buildup. Monitoring the reaction progress via thin-layer chromatography allows for real-time assessment of raw material consumption, ensuring the reaction is stopped at the optimal point to prevent degradation. Detailed standardized synthesis steps see the guide below.
- Mix trifluoromethanesulfonyl alkyne compound and copper nitrate trihydrate in butyl butyrate solvent.
- Stir the mixture at 90-100°C under air atmosphere until raw materials disappear.
- Extract with ethyl acetate, wash with brine, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology translates into tangible strategic benefits regarding cost stability and supply continuity. The shift from exotic reagents like Tf2O to commodity chemicals like copper nitrate drastically reduces the volatility of raw material costs, shielding the supply chain from market fluctuations associated with specialized fluorinating agents. Additionally, the elimination of cryogenic requirements and inert atmosphere setups lowers the capital expenditure needed for reactor infrastructure, allowing for more flexible manufacturing scheduling. The simplified workup procedure, involving standard extraction and crystallization, reduces solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable production model. These factors collectively enhance the overall resilience of the supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive triflic anhydride and organolithium bases with inexpensive copper nitrate trihydrate results in substantial cost savings on reagent procurement. By avoiding the need for specialized low-temperature equipment and rigorous moisture exclusion systems, operational expenditures related to energy consumption and maintenance are significantly lowered. The high atom economy of the cyclization reaction minimizes raw material waste, further driving down the cost per kilogram of the final active pharmaceutical ingredient intermediate. This economic efficiency allows for more competitive pricing structures without compromising on the quality or purity of the delivered chemical products.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as substituted phenylacetylenes and common metal salts ensures a robust supply chain that is less susceptible to geopolitical disruptions or vendor shortages. The tolerance of the reaction to air and moisture simplifies logistics and storage requirements, reducing the risk of material degradation during transport. This reliability is crucial for maintaining consistent production schedules and meeting tight delivery deadlines for global clients. The ability to source raw materials from multiple suppliers enhances negotiation leverage and secures long-term supply continuity for critical drug development programs.
- Scalability and Environmental Compliance: The use of butyl butyrate, a solvent with a favorable environmental profile, aligns with increasingly stringent global regulations regarding volatile organic compound (VOC) emissions and waste management. The mild reaction conditions facilitate safe scale-up from laboratory benchtop to multi-ton industrial reactors without the exponential increase in safety risks associated with exothermic oxidations. This scalability ensures that production capacity can be rapidly expanded to meet surging market demand for novel therapeutic agents. Furthermore, the reduced generation of hazardous byproducts simplifies effluent treatment processes, supporting corporate sustainability goals and regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these specialized heterocyclic compounds. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing development pipelines. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industrial application. Clients are encouraged to review these insights to better understand the operational benefits and chemical versatility offered by this novel synthetic route.
Q: What are the key advantages of the copper nitrate method over traditional Tf2O routes?
A: The copper nitrate method eliminates the need for expensive and hazardous triflic anhydride (Tf2O) and strong bases like n-BuLi, offering a safer, one-pot protocol with milder conditions.
Q: What is the typical yield range for this synthesis?
A: According to patent data, yields generally range from average to good, with specific examples demonstrating isolated yields between 44% and 73% depending on the substituents.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of simple raw materials, air atmosphere, and standard solvents like butyl butyrate makes the process highly amenable to commercial scale-up without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethanesulfonyl Isoxazoles Supplier
At NINGBO INNO PHARMCHEM, we leverage cutting-edge synthetic methodologies like the copper-catalyzed cyclization described in CN111196786B to deliver superior chemical solutions to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from discovery to market. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of trifluoromethanesulfonyl-substituted isoxazoles meets the highest industry standards for pharmaceutical and agrochemical applications. Our commitment to technical excellence and operational efficiency makes us the preferred partner for complex intermediate manufacturing.
We invite you to collaborate with us to explore how this advanced synthesis technology can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and supply chain planning. Let us help you accelerate your development timeline with reliable, high-quality chemical intermediates.
