Advanced Visible Light Photocatalysis for Commercial Scale-up of Complex Azaspirocyclohexadienones
Advanced Visible Light Photocatalysis for Commercial Scale-up of Complex Azaspirocyclohexadienones
The pharmaceutical industry is constantly seeking innovative synthetic methodologies that balance structural complexity with process efficiency, and the technology disclosed in patent CN108117507B represents a significant leap forward in this domain. This patent details a novel visible light-induced dearomatization cyclization strategy for synthesizing aryl-substituted azaspirocyclohexadienones, a privileged scaffold found in numerous bioactive natural products and drug candidates. By leveraging a ruthenium-based photocatalyst system under mild blue LED irradiation, this method overcomes the kinetic barriers associated with traditional thermal cyclizations, offering a robust pathway for generating high-purity pharmaceutical intermediates. For R&D directors and process chemists, this approach provides a versatile tool for accessing spirocyclic cores that are notoriously difficult to construct using conventional logic, thereby expanding the chemical space available for medicinal chemistry campaigns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of nitrogen-containing spirocyclic frameworks has relied heavily on harsh oxidative conditions or expensive transition metal catalysts that operate at elevated temperatures. These traditional protocols often suffer from poor atom economy and limited functional group tolerance, leading to significant downstream purification challenges and reduced overall yields. Furthermore, the reliance on stoichiometric oxidants generates substantial chemical waste, creating environmental burdens that conflict with modern green chemistry principles. For procurement managers, these inefficiencies translate into higher raw material costs and extended lead times due to the complexity of isolating the desired product from complex reaction mixtures containing multiple side products. The lack of selectivity in many thermal processes also necessitates rigorous quality control measures, further straining resources and delaying project timelines in competitive drug development pipelines.
The Novel Approach
In stark contrast, the visible light-mediated protocol described in the patent utilizes mild reaction conditions at room temperature, effectively preserving sensitive functional groups that would otherwise decompose under thermal stress. By employing aryl diazonium tetrafluoroborates as inexpensive radical precursors, the method achieves high conversion rates with exceptional regioselectivity, minimizing the formation of unwanted by-products. This shift from thermal to photochemical activation not only simplifies the operational procedure but also aligns with sustainability goals by reducing energy consumption and waste generation. For supply chain heads, the simplicity of the reaction setup—requiring only standard LED arrays and common solvents like DMF—ensures that the process can be rapidly implemented across different manufacturing sites without specialized high-pressure equipment. This technological advancement positions the synthesis of azaspirocyclohexadienones as a more reliable and cost-effective operation, directly addressing the pain points of scalability and reproducibility in fine chemical manufacturing.
Mechanistic Insights into Visible Light-Induced Dearomatization Cyclization
The core of this transformative synthesis lies in the photoredox catalytic cycle driven by the Ru(bpy)3Cl2 complex, which facilitates the generation of aryl radicals under visible light irradiation. Upon absorption of blue photons, the ruthenium catalyst enters an excited state capable of engaging in single-electron transfer (SET) processes with the diazonium salt substrate. This interaction triggers the release of nitrogen gas and the formation of a highly reactive aryl radical species, which subsequently adds to the electron-deficient alkene moiety of the N-benzyl acrylamide precursor. The resulting carbon-centered radical undergoes an intramolecular cyclization onto the aromatic ring, followed by oxidation and deprotonation steps to restore aromaticity in the newly formed spirocyclic system. This elegant cascade sequence allows for the rapid assembly of complex molecular architectures from simple starting materials, demonstrating the power of photocatalysis in modern organic synthesis.
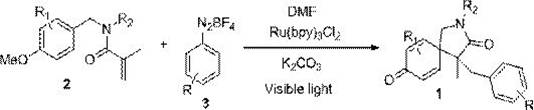
Understanding the mechanistic nuances is critical for controlling impurity profiles and ensuring batch-to-batch consistency in a commercial setting. The use of potassium carbonate as a base plays a pivotal role in neutralizing acidic by-products and facilitating the final rearomatization step, while the choice of DMF as a solvent ensures optimal solubility for both polar and non-polar intermediates. The mild nature of the radical generation prevents uncontrolled polymerization or degradation of the acrylamide double bond, a common issue in free-radical reactions. For technical teams, mastering these parameters allows for fine-tuning the reaction to accommodate diverse substituents on the aromatic rings, thereby enabling the synthesis of a broad library of analogues for structure-activity relationship (SAR) studies. This level of control is essential for producing high-purity intermediates that meet the rigorous standards required for subsequent biological evaluation and clinical development.
How to Synthesize Azaspirocyclohexadienone Efficiently
The practical implementation of this photocatalytic route is straightforward and amenable to standard laboratory and pilot plant operations, requiring minimal specialized infrastructure beyond a light source. The process begins with the preparation of the N-benzyl acrylamide substrate, which can be synthesized via a simple three-step sequence from commercially available aldehydes and amines, ensuring a secure supply of starting materials. Once the substrate is ready, it is combined with the diazonium salt, catalyst, and base in a reaction vessel purged with nitrogen to prevent oxygen quenching of the excited catalyst state. The detailed standardized synthesis steps for optimizing yield and purity are outlined in the guide below, providing a clear roadmap for technical teams to replicate the results reported in the patent literature.
- Prepare the reaction mixture by combining N-benzyl acrylamide substrate, Ru(bpy)3Cl2 catalyst, K2CO3 base, and DMF solvent in a clean reaction vessel under nitrogen.
- Add the aryl diazonium tetrafluoroborate salt to the mixture and ensure the system is sealed and protected from ambient light sources other than the reactor.
- Irradiate the reaction with 5W blue LEDs at room temperature for 24 hours, then perform aqueous workup and column chromatography to isolate the pure spirocyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this visible light methodology offers substantial strategic benefits for organizations focused on cost reduction in pharmaceutical intermediate manufacturing. The elimination of expensive stoichiometric oxidants and the use of catalytic amounts of a readily available ruthenium complex significantly lower the direct material costs associated with production. Additionally, the mild reaction conditions reduce the energy footprint of the process, as there is no need for prolonged heating or cryogenic cooling, leading to indirect savings in utility expenses. For procurement managers, the reliance on commodity chemicals like DMF and potassium carbonate ensures stable pricing and availability, mitigating the risks associated with supply chain disruptions for exotic reagents. This economic efficiency makes the technology particularly attractive for large-scale production where margin optimization is critical for maintaining competitiveness in the global market.
- Cost Reduction in Manufacturing: The process utilizes inexpensive diazonium salts as radical sources instead of costly organometallic reagents, drastically simplifying the bill of materials. By operating at room temperature, the method eliminates the need for energy-intensive heating or cooling systems, resulting in significant operational expenditure savings. Furthermore, the high selectivity of the reaction reduces the burden on downstream purification, lowering solvent consumption and waste disposal costs associated with removing complex impurity profiles.
- Enhanced Supply Chain Reliability: The starting materials, including N-benzyl acrylamides and aryl diazonium salts, are derived from widely available commodity chemicals, ensuring a robust and continuous supply chain. The simplicity of the reaction setup means that production can be easily transferred between different manufacturing facilities without requiring specialized high-pressure or high-temperature reactors. This flexibility enhances supply security and reduces lead times for delivering high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: The use of LED light sources generates minimal heat, simplifying thermal management during scale-up and allowing for safer operation in larger reactors. The green chemistry profile of the method, characterized by high atom economy and reduced waste generation, aligns with increasingly stringent environmental regulations. This compliance reduces the regulatory burden and facilitates faster approval for commercial manufacturing processes in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology, based on the detailed data provided in the patent documentation. These insights are designed to clarify the operational feasibility and strategic value of adopting this synthetic route for your specific application needs. Understanding these aspects will help stakeholders make informed decisions about integrating this methodology into their existing production workflows.
Q: What are the primary advantages of this visible light method over traditional thermal cyclization?
A: The visible light-induced method operates at room temperature, eliminating the need for harsh thermal conditions that often degrade sensitive functional groups. It utilizes inexpensive diazonium salts as radical sources and achieves high selectivity with minimal by-product formation compared to conventional oxidative cyclization protocols.
Q: Is the Ru(bpy)3Cl2 catalyst difficult to remove from the final pharmaceutical intermediate?
A: No, the catalyst is used in very low loading (0.5 mol%) and standard aqueous workup procedures involving ethyl acetate extraction and brine washing effectively separate the organic product from the metal complex. Further purification via column chromatography ensures the final material meets stringent purity specifications required for API synthesis.
Q: Can this synthetic route be scaled for industrial production of anticancer intermediates?
A: Yes, the process demonstrates excellent scalability potential due to the use of low-energy LED light sources which generate minimal heat, simplifying thermal management in large reactors. The mild reaction conditions and readily available starting materials support reliable supply chain continuity for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azaspirocyclohexadienone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light photocatalysis in advancing the synthesis of complex pharmaceutical intermediates like azaspirocyclohexadienones. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic discoveries are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch delivered meets the highest quality standards required for drug development. We are committed to supporting our partners through every stage of the product lifecycle, from initial route scouting to full-scale commercial manufacturing.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your timeline and optimize your supply chain for high-purity azaspirocyclohexadienone derivatives.
