Advanced Ruthenium-Catalyzed Synthesis of 7-Chloro-2-(3-Chlorophenyl)Quinazoline for Commercial Pharmaceutical Applications
Advanced Ruthenium-Catalyzed Synthesis of 7-Chloro-2-(3-Chlorophenyl)Quinazoline for Commercial Pharmaceutical Applications
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the urgent need for greener, more efficient synthetic pathways that align with sustainable development goals. Patent CN109180592B introduces a groundbreaking methodology for the synthesis of 7-chloro-2-(3-chlorophenyl)quinazoline, a critical scaffold in medicinal chemistry known for its potential in oncology and other therapeutic areas. This technology leverages a sophisticated NNN-type pincer ruthenium(II) complex to facilitate a direct dehydrogenative coupling between an alcohol and a nitrile, fundamentally shifting the paradigm from traditional oxidant-dependent protocols. By utilizing air as the terminal oxidant source and avoiding stoichiometric heavy metal oxidants, this process offers a compelling value proposition for reliable pharmaceutical intermediate suppliers seeking to optimize their production portfolios. The structural integrity and chemical identity of the target molecule are paramount for downstream applications, as illustrated below.
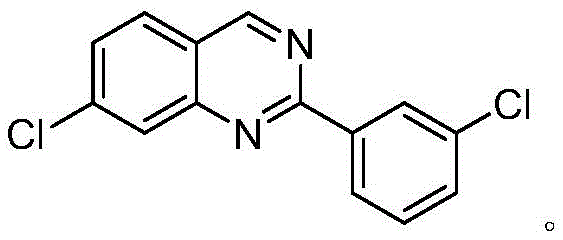
This innovation represents a significant leap forward in cost reduction in pharma manufacturing, addressing both economic and environmental pain points simultaneously. For R&D directors and process chemists, the ability to construct the quinazoline core in a single step with high atom economy is a major technical achievement. The method circumvents the generation of substantial inorganic waste streams associated with conventional oxidation methods, thereby simplifying the purification workflow and reducing the burden on waste treatment facilities. Furthermore, the operational simplicity of running the reaction under an air atmosphere, rather than requiring rigorous inert gas conditions, lowers the barrier for commercial scale-up of complex pharmaceutical intermediates. This report delves deep into the mechanistic nuances, comparative advantages, and commercial implications of adopting this patented technology for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazoline heterocyclic skeleton has relied heavily on the condensation of anthranilic acid derivatives with amidines or nitriles, often necessitating harsh reaction conditions and stoichiometric amounts of strong oxidants. Traditional protocols frequently employ reagents such as hypervalent iodine compounds, manganese dioxide, or copper salts in excess to drive the oxidative cyclization to completion. These conventional approaches suffer from significant drawbacks, including poor atom economy, the generation of toxic heavy metal waste, and the formation of difficult-to-remove byproducts that complicate downstream purification. The reliance on pre-functionalized substrates, such as aldehydes or acids, also adds steps to the synthetic sequence, increasing the overall cost of goods sold (COGS) and extending the production lead time. Moreover, the use of stoichiometric oxidants often poses safety hazards on a large scale due to exothermic risks and the potential for runaway reactions, making these methods less desirable for modern high-purity pharmaceutical intermediate manufacturing where safety and sustainability are critical KPIs.
The Novel Approach
In stark contrast, the methodology disclosed in CN109180592B utilizes a transition-metal catalyzed dehydrogenative coupling strategy that transforms simple, commercially available alcohols directly into the target quinazoline. By employing (2-amino-4-chlorophenyl)methanol and m-chlorobenzonitrile as starting materials, the process bypasses the need for pre-oxidation of the alcohol to an aldehyde, effectively merging the oxidation and condensation steps into a single catalytic cycle. The use of the NNN-type pincer ruthenium(II) catalyst allows the reaction to proceed efficiently under mild air atmosphere conditions at 130°C, eliminating the need for expensive and hazardous external oxidants. This "borrowing hydrogen" or dehydrogenative approach not only streamlines the synthetic route but also drastically reduces the E-factor (environmental factor) of the process by minimizing waste generation. The operational robustness of this system, capable of functioning in open air without sensitive inert gas protection, makes it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates in a fast-paced commercial environment.
Mechanistic Insights into NNN-Pincer Ruthenium(II) Catalyzed Cyclization
The heart of this technological breakthrough lies in the unique electronic and steric properties of the NNN-type pincer ruthenium(II) catalyst, which orchestrates the multi-step transformation with remarkable precision. The catalyst, characterized by a symmetrical 2,6-bis(6-methylimidazo[1,2-a]pyridin-2-yl)pyridine ligand framework, stabilizes the ruthenium center and facilitates the reversible dehydrogenation of the benzylic alcohol substrate. The mechanism initiates with the coordination of the alcohol to the metal center, followed by beta-hydride elimination to generate the reactive aldehyde intermediate in situ, releasing hydrogen which is subsequently scavenged by oxygen from the air to form water. This transient aldehyde then undergoes nucleophilic attack by the amine group of the adjacent aniline moiety (after condensation with the nitrile), leading to the formation of a dihydroquinazoline intermediate. The final aromatization step, driven by the removal of further hydrogen equivalents, yields the fully conjugated quinazoline system. The specific geometry of the pincer ligand prevents catalyst decomposition and ensures high turnover numbers, which is critical for maintaining low catalyst loading levels in commercial batches.
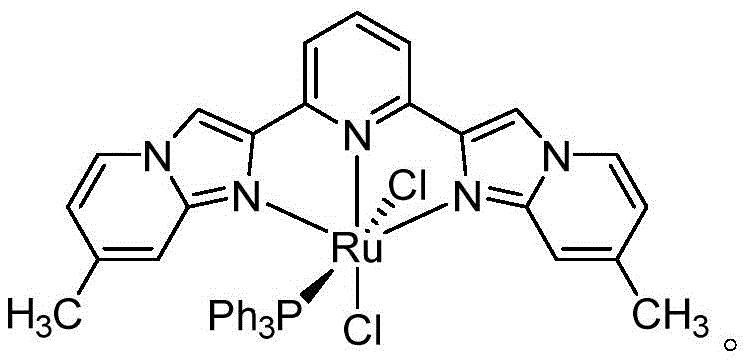
From an impurity control perspective, this catalytic cycle offers superior selectivity compared to radical-based oxidative methods. Because the oxidation is mediated by the metal center rather than free radical species generated by stoichiometric oxidants, there is a marked reduction in non-specific oxidation byproducts, such as over-oxidized carboxylic acids or polymerized tars. The mild basic conditions employed (using bases like potassium tert-butoxide or sodium methoxide) further suppress acid-catalyzed degradation pathways that often plague quinazoline synthesis. The result is a cleaner reaction profile that simplifies the isolation of the target API intermediate, ensuring that the final product meets stringent purity specifications required for clinical supply. Understanding this mechanism allows process chemists to fine-tune parameters such as base strength and solvent polarity to maximize yield while minimizing trace impurities, providing a robust platform for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 7-Chloro-2-(3-Chlorophenyl)Quinazoline Efficiently
The practical implementation of this synthesis involves a straightforward one-pot procedure that is amenable to standard laboratory and pilot plant equipment. The process begins by charging a dry, clean high-pressure reactor with the alcohol substrate, the nitrile coupling partner, the specialized ruthenium catalyst, a suitable base, and a high-boiling solvent such as tert-amyl alcohol or 1,4-dioxane. The reaction mixture is then heated to 130°C under magnetic stirring in an air atmosphere, where it typically reaches completion within 2 hours as monitored by TLC. Following the reaction, the solvent is removed via vacuum rotary evaporation, and the crude product is purified using standard chromatographic techniques to afford the target quinazoline in high yield. This streamlined workflow eliminates multiple unit operations associated with traditional methods, such as separate oxidation steps and extensive aqueous workups to remove metal oxidant residues.
- Charge a dry high-pressure reactor with (2-amino-4-chlorophenyl)methanol, m-chlorobenzonitrile, NNN-pincer Ru(II) catalyst (1 mol%), base (e.g., t-BuOK), and solvent (e.g., tert-amyl alcohol).
- Stir the mixture magnetically under an air atmosphere and heat to 130°C in an oil bath for approximately 2 hours until TLC indicates complete consumption of the nitrile.
- Upon completion, perform vacuum rotary evaporation to remove solvents and purify the crude residue via thin-layer chromatography or column chromatography to isolate the target quinazoline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology translates into tangible strategic advantages beyond mere technical elegance. The shift from stoichiometric oxidants to a catalytic air-oxidation system fundamentally alters the cost structure of the manufacturing process by removing the expense of purchasing, handling, and disposing of large quantities of oxidizing agents. This reduction in raw material complexity directly contributes to cost reduction in pharma manufacturing, allowing for more competitive pricing models in a margin-sensitive market. Additionally, the use of air as the oxidant source removes the logistical burden of supplying and storing hazardous chemicals, thereby enhancing site safety and reducing insurance and compliance costs associated with dangerous goods. The simplified workup procedure, which avoids the generation of heavy metal sludge, significantly lowers waste disposal fees and accelerates the batch release cycle, enabling faster response times to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of stoichiometric oxidants and the use of cheap, readily available alcohol starting materials significantly lower the direct material costs per kilogram of product. By avoiding the purchase of expensive oxidizing reagents and the subsequent costs associated with neutralizing and disposing of their reduced byproducts, the overall variable cost of production is substantially decreased. Furthermore, the high efficiency of the catalyst means that only minute quantities of the precious metal complex are required to drive the reaction, optimizing the utilization of expensive catalytic resources and maximizing the return on investment for each batch produced.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the ability to operate under air atmosphere without the need for rigorous inert gas blanketing, simplifies the engineering requirements for production reactors. This ease of operation reduces the risk of batch failures due to oxygen ingress or equipment malfunction, ensuring a more consistent and reliable supply of the intermediate. The use of common solvents like tert-amyl alcohol and toluene, which are widely available in the global chemical supply chain, mitigates the risk of raw material shortages that can occur with specialized or niche reagents, thereby securing the continuity of supply for long-term contracts.
- Scalability and Environmental Compliance: The green chemistry credentials of this process, characterized by high atom economy and minimal waste generation, align perfectly with increasingly stringent global environmental regulations. Scaling this process from gram to tonne scale does not introduce disproportionate safety hazards related to exothermic oxidations, making the technology transfer to large-scale manufacturing seamless. The reduction in hazardous waste streams simplifies the permitting process for new manufacturing lines and enhances the corporate sustainability profile, which is becoming a critical factor in supplier selection for major multinational pharmaceutical companies committed to ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ruthenium-catalyzed synthesis. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a clear understanding of the process capabilities.
Q: What are the primary advantages of using the NNN-pincer Ruthenium catalyst over traditional oxidants?
A: The NNN-pincer Ruthenium(II) catalyst enables a dehydrogenative coupling mechanism that eliminates the need for stoichiometric external oxidants. This significantly reduces chemical waste, simplifies the workup procedure by avoiding oxidant byproducts, and improves the overall atom economy of the synthesis.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method is highly scalable. It operates under air atmosphere rather than requiring inert gas protection, uses readily available alcohol starting materials, and completes in a short reaction time (2 hours), making it suitable for large-scale commercial manufacturing.
Q: What is the typical purity profile achievable with this catalytic route?
A: The reaction demonstrates high selectivity, minimizing side reactions common in oxidative cyclizations. With standard purification techniques like chromatography, high-purity 7-chloro-2-(3-chlorophenyl)quinazoline can be obtained, meeting stringent specifications for downstream drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Chloro-2-(3-Chlorophenyl)Quinazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN109180592B for the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust, GMP-compliant commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies gained at the bench level are fully realized at the plant scale. We maintain stringent purity specifications through our state-of-the-art rigorous QC labs, guaranteeing that every batch of 7-chloro-2-(3-chlorophenyl)quinazoline meets the exacting standards required for drug substance manufacturing.
We invite global partners to collaborate with us to leverage this cutting-edge synthesis for their pipeline projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green chemistry approach can optimize your supply chain economics. We encourage you to contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects, ensuring a secure and cost-effective supply of this critical intermediate.
