Advanced Selenium-Catalyzed Synthesis of Propargyl Alpha-Amino Boronate Compounds for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex molecular architectures, particularly those involving boron-carbon-nitrogen frameworks which are pivotal in modern drug discovery. Patent CN114805416A introduces a groundbreaking preparation method for propargyl α-amino boronate compounds that addresses many limitations of prior art. This innovation utilizes a selenium-catalyzed system in the presence of a hypervalent iodine oxidant to facilitate the direct amination of propargyl boronate precursors. The significance of this technology lies in its ability to construct the critical C-N bond at the alpha position of the boronate ester with high efficiency and selectivity. For research and development teams focusing on proteasome inhibitors or other boron-containing therapeutics, this protocol offers a reliable pathway to access diverse chemical space that was previously difficult to navigate. The method eliminates the need for pre-functionalized substrates, allowing for a more convergent and atom-economical synthetic strategy that aligns with green chemistry principles.
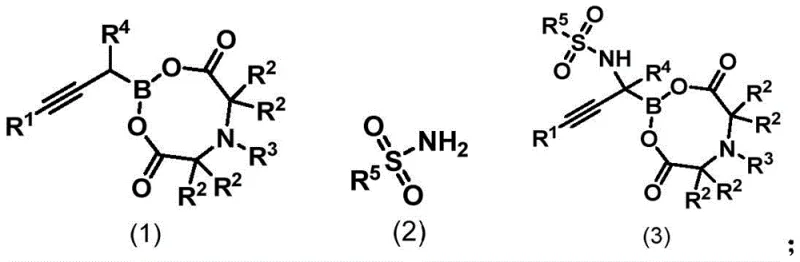
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α-aminoboronic acid derivatives has relied heavily on nucleophilic addition of boron species to imines or metal-catalyzed borylation of amines, both of which present substantial challenges for industrial application. Traditional routes often necessitate harsh reaction conditions, including elevated temperatures and pressures, which can compromise the integrity of sensitive functional groups commonly found in pharmaceutical intermediates. Furthermore, many existing protocols require multi-step protection and deprotection sequences to manage the reactivity of the amine and boron moieties, leading to reduced overall yields and increased waste generation. Comparative studies highlighted in the patent data indicate that conventional transition metal catalysts, such as organorhodium or organocobalt complexes, frequently fail to promote the desired transformation on propargyl boronate substrates, resulting in no reaction or poor conversion rates. These inefficiencies create bottlenecks in supply chains, driving up costs and extending lead times for critical active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the novel selenium-catalyzed methodology described in the patent provides a streamlined solution that bypasses these historical hurdles. By employing a specific combination of a selenium catalyst, such as IMeSe, and a hypervalent iodine oxidant like iodobenzene phenylacetate, the reaction proceeds smoothly under mild conditions ranging from 10°C to 60°C. This approach does not require any acidic or basic additives, thereby simplifying the reaction matrix and reducing the burden on downstream purification processes. The direct reaction between the propargyl boronate compound and the sulfamide reagent allows for the construction of the target α-amino boronate structure in a single operational step. This simplicity translates directly into operational excellence, as it minimizes the number of unit operations required and enhances the safety profile of the manufacturing process by avoiding extreme thermal or pressure conditions. The broad substrate scope further ensures that this method is versatile enough to handle a wide array of chemical diversity required for modern drug development pipelines.
Mechanistic Insights into Selenium-Catalyzed Amination
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific targets. The reaction initiates with the interaction between the sulfamide and the selenium catalyst in the presence of the hypervalent iodine oxidant, generating a reactive selenoimide species. This electrophilic intermediate then engages with the propargyl boronate substrate through an ene-type reaction mechanism. During this critical step, the imine moiety abstracts a proton from the alpha-position of the boronate ester while the selenium atom inserts into the carbon-carbon triple bond, forming a transient allene intermediate. This unique rearrangement is facilitated by the coordinating ability of the boron atom, which helps stabilize the transition state. Subsequently, the electrons from the imine double bond attack the carbon at the alpha-position of the boronate, triggering a 2,3-sigmatropic shift. This migration effectively transfers the nitrogen functionality to the desired alpha-carbon center. Finally, an oxidative turnover step regenerates the active selenium catalyst and releases the final propargyl α-amino boronate product. This detailed mechanistic pathway explains the high regioselectivity and the tolerance for various functional groups observed in the experimental data.
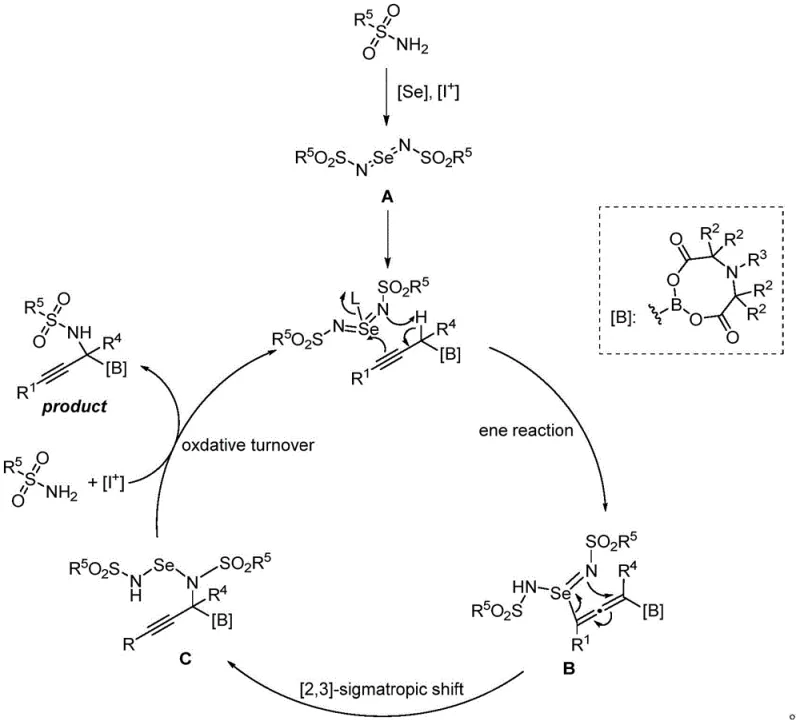
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or uncontrolled metal-catalyzed pathways. The concerted nature of the sigmatropic shift and the specific activation of the alpha-proton by the selenoimide species minimize the formation of random byproducts or polymerization of the alkyne moiety. The use of hypervalent iodine oxidants ensures a clean oxidation potential that is sufficient to drive the catalytic cycle without over-oxidizing sensitive groups like sulfides or amines elsewhere in the molecule. Furthermore, the absence of heavy metal residues, which are a major concern in pharmaceutical manufacturing due to strict regulatory limits, simplifies the purification strategy. The reaction byproducts are generally organic and easier to separate via standard chromatographic techniques compared to inorganic metal salts. This inherent cleanliness of the reaction profile contributes to higher crude purity, reducing the load on final purification steps and ensuring that the final API intermediate meets stringent quality specifications with minimal effort.
How to Synthesize Propargyl Alpha-Amino Boronate Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires attention to specific reagent grades and mixing protocols to ensure reproducibility. The process begins by charging a reaction vessel with the propargyl boronate starting material, the sulfamide amination reagent, and the selenium catalyst in a molar ratio that optimizes turnover, typically around 1:1-2:0.05-0.1. Dichloromethane is the preferred solvent due to its ability to dissolve all components effectively while maintaining a stable reaction environment. Once the initial mixture is homogenized, the hypervalent iodine oxidant is introduced, and the system is sealed to prevent moisture ingress which could degrade the oxidant. The reaction is then allowed to proceed with stirring at a controlled temperature, usually around 40°C, for a duration sufficient to reach full conversion, often within 12 to 24 hours. Monitoring the reaction progress via TLC or HPLC is recommended to determine the precise endpoint. Upon completion, the mixture undergoes a straightforward workup involving filtration to remove solid byproducts followed by concentration. The crude product is then purified using silica gel column chromatography to isolate the target propargyl α-amino boronate compound in high yield and purity.
- Combine the propargyl boronate starting material with sulfamide and a selenium catalyst (e.g., IMeSe) in a suitable solvent like dichloromethane.
- Add a hypervalent iodine oxidant (e.g., iodobenzene phenylacetate) and stir the mixture at mild temperatures between 10°C and 60°C until completion.
- Purify the resulting propargyl alpha-amino boronate product using standard silica gel column chromatography to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this selenium-catalyzed technology represents a strategic opportunity to enhance supply security and reduce total cost of ownership. The elimination of expensive and scarce transition metal catalysts like rhodium or cobalt, which were shown to be ineffective for this specific transformation, removes a significant cost driver and supply risk from the manufacturing equation. Selenium catalysts are generally more abundant and cost-effective, and the loading levels required are low, contributing to substantial cost savings in raw material procurement. Additionally, the mild reaction conditions mean that standard glass-lined or stainless steel reactors can be used without the need for specialized high-pressure or high-temperature equipment, lowering capital expenditure requirements for scale-up. The simplified workup and purification process also reduce solvent consumption and waste disposal costs, aligning with sustainability goals and reducing the environmental footprint of the production facility.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of precious metal catalysts with more economical selenium-based systems and the reduction in processing steps. By avoiding multi-step protection strategies and harsh conditions that require expensive energy inputs, the overall manufacturing cost is significantly lowered. The high yields reported across a broad range of substrates mean that less starting material is wasted, improving the mass balance and further driving down the cost per kilogram of the final intermediate. This efficiency allows for more competitive pricing models when sourcing these critical building blocks for drug development programs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially available and stable reagents that do not rely on geopolitically constrained rare earth metals. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs or out-of-specification results. The ability to synthesize a wide variety of derivatives from a common set of starting materials allows for greater flexibility in responding to changing demand patterns for different API candidates. This versatility ensures that suppliers can maintain continuous production schedules even as project requirements evolve, providing a stable and reliable source of high-quality intermediates for downstream partners.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the absence of hazardous high-pressure steps and the use of common organic solvents. The mild thermal profile reduces the risk of thermal runaway incidents, enhancing operational safety in large-scale plants. From an environmental compliance standpoint, the reduction in heavy metal waste simplifies effluent treatment and lowers the regulatory burden associated with metal residue limits in pharmaceutical products. The atom economy of the direct amination approach minimizes waste generation, supporting corporate sustainability initiatives and helping manufacturers meet increasingly stringent environmental regulations without compromising on production efficiency or product quality.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the scope and practicality of this synthesis method. These insights are derived directly from the extensive experimental data provided in the patent documentation, covering aspects from catalyst selection to substrate compatibility. Understanding these details is essential for integrating this new methodology into existing synthetic routes and for assessing its potential impact on project timelines and budgets. We encourage stakeholders to review these technical clarifications to fully appreciate the advantages offered by this innovative approach to alpha-amino boronate synthesis.
Q: What are the advantages of this selenium-catalyzed method over traditional metal catalysts?
A: Unlike traditional rhodium or cobalt catalysts which may fail to react with specific propargyl boronate substrates, this selenium-catalyzed approach demonstrates superior reactivity and broader substrate tolerance under significantly milder conditions without requiring expensive transition metals.
Q: Does this method require harsh reaction conditions or additives?
A: No, the process operates efficiently at temperatures between 10°C and 60°C without the need for strong acid or base additives, simplifying the workup procedure and enhancing safety profiles for large-scale manufacturing.
Q: What types of functional groups are compatible with this synthesis route?
A: The method exhibits excellent functional group tolerance, successfully accommodating substrates containing halogens, cyano groups, azides, esters, ethers, and various heterocyclic aromatic systems without significant side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Propargyl Alpha-Amino Boronate Supplier
As the global demand for boron-containing therapeutics continues to rise, securing a supply partner with deep technical expertise and scalable manufacturing capabilities is paramount. NINGBO INNO PHARMCHEM stands ready to support your development needs by leveraging advanced synthetic methodologies like the selenium-catalyzed amination described herein. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in early-stage discovery or late-stage clinical supply. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of propargyl alpha-amino boronate intermediate meets the highest industry standards for quality and consistency. Our commitment to technical excellence allows us to troubleshoot complex synthetic challenges and deliver customized solutions tailored to your specific molecular targets.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can benefit your specific project. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic advantages of switching to this more efficient manufacturing process. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-purity propargyl alpha-amino boronate compounds reliably. Let us collaborate to accelerate your drug development timeline and optimize your supply chain for long-term success.
