Advanced Catalytic Hydroboration for Scalable Boronate Compound Manufacturing
The landscape of organoboron chemistry is undergoing a significant transformation, driven by the urgent need for safer, more stable, and highly efficient synthetic methodologies in the pharmaceutical and fine chemical sectors. Patent CN111718363B introduces a groundbreaking preparation method for boronate compounds that addresses the longstanding limitations of traditional borane reagents. By leveraging a novel catalytic system involving azacarbene borane and a Lewis acid catalyst, this technology enables the hydroboration of olefins under remarkably mild and controlled conditions. This innovation is particularly critical for the production of high-purity pharmaceutical intermediates, where impurity profiles and process safety are paramount concerns for R&D directors and supply chain managers alike. The disclosed method utilizes stable Lewis base-borane adducts, which offer superior stability and lower toxicity compared to conventional boranes, thereby streamlining the manufacturing workflow and reducing operational hazards associated with pyrophoric materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organoboron compounds via hydroboration has been plagued by significant safety and handling challenges. Traditional borane reagents, such as borane-tetrahydrofuran complex or diborane gas, are notoriously unstable, moisture-sensitive, and often pyrophoric, requiring stringent inert atmosphere techniques and specialized equipment for safe handling. These characteristics not only increase the capital expenditure for manufacturing facilities but also introduce substantial risks regarding worker safety and environmental compliance. Furthermore, conventional methods often suffer from poor selectivity when dealing with complex olefin substrates, leading to difficult purification processes and reduced overall yields. The instability of these reagents can also result in inconsistent batch-to-batch quality, which is unacceptable for the rigorous standards required in active pharmaceutical ingredient (API) manufacturing. Consequently, there has been a persistent demand for a more robust reagent system that maintains high reactivity without compromising on safety or stability.
The Novel Approach
The methodology described in patent CN111718363B represents a paradigm shift by employing azacarbene borane as a stable hydroborating agent. This approach utilizes a catalytic amount of tris(pentafluorophenyl)borane, B(C6F5)3, to activate the reaction between the stable borane adduct and 1,2-diphenylethylene derivatives. As illustrated in the general reaction scheme below, this system allows for the efficient conversion of various substituted stilbenes into valuable boronate esters.
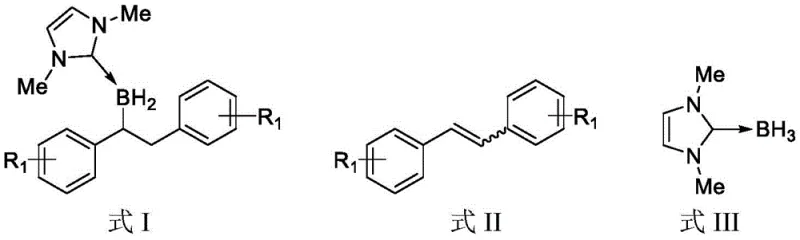
The use of azacarbene borane eliminates the need for handling hazardous free boranes, as the reagent exists as a stable solid or solution that is easy to weigh and transfer. The reaction proceeds smoothly in common organic solvents like toluene at moderate temperatures ranging from 60°C to 80°C. This mild thermal profile is energy-efficient and compatible with a wide range of functional groups, including halogens and ethers, which might be sensitive to harsher conditions. The result is a streamlined process that delivers high-purity boronate compounds with excellent yields, typically exceeding 90% for many substrates, thus providing a reliable pathway for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into B(C6F5)3-Catalyzed Hydroboration
The efficacy of this synthetic route lies in the unique mechanistic interplay between the Lewis acidic catalyst and the stable borane adduct. Tris(pentafluorophenyl)borane acts as a potent Lewis acid that coordinates with the nitrogen atom of the azacarbene moiety or interacts with the boron center to weaken the B-N bond temporarily. This activation lowers the energy barrier for the hydroboration step, allowing the boron species to add across the carbon-carbon double bond of the 1,2-diphenylethylene derivative. Unlike uncatalyzed reactions which might require excessive heat or pressure, this catalytic cycle operates efficiently at 80°C, ensuring that the reaction kinetics are favorable without promoting thermal decomposition of sensitive functional groups. The mechanism ensures that the boron adds regioselectively, preserving the structural integrity of the complex olefin backbone which is essential for downstream cross-coupling reactions commonly used in drug discovery.
Furthermore, the stability of the azacarbene borane adduct plays a crucial role in impurity control. Because the borane is sequestered by the nitrogen heterocycle until activated by the catalyst, side reactions such as polymerization of the olefin or over-reduction are minimized. This inherent selectivity results in a cleaner crude reaction profile, significantly reducing the burden on downstream purification units like column chromatography or crystallization. For process chemists, this means fewer impurities to track and validate, accelerating the timeline from laboratory scale to commercial production. The ability to tolerate both electron-withdrawing groups like trifluoromethyl and halogens, as well as electron-donating groups like methoxy and alkyl chains, demonstrates the robustness of this catalytic system across a diverse chemical space, making it a versatile tool for synthesizing complex building blocks.
How to Synthesize Boronate Compounds Efficiently
The practical implementation of this technology is straightforward and relies on standard laboratory and plant equipment. The process begins by charging a reactor with the specific 1,2-diphenylethylene derivative and the azacarbene borane reagent in a molar ratio of approximately 1:1.5, ensuring an excess of the borane source to drive the reaction to completion. The catalyst, B(C6F5)3, is added at a loading of 5 mol%, which is sufficient to initiate the catalytic cycle without requiring expensive stoichiometric amounts. The reaction is conducted in anhydrous toluene under a nitrogen atmosphere to prevent moisture interference, heated to 80°C, and maintained for 6 to 12 hours depending on the specific substrate reactivity. Detailed standardized synthesis steps follow below.
- Combine 1,2-diphenylethylene derivative, azacarbene borane (1.5 equivalents), and B(C6F5)3 catalyst (5 mol%) in anhydrous toluene under nitrogen protection.
- Heat the reaction mixture to 80°C and stir continuously for 6 to 12 hours to ensure complete conversion of the olefin substrate.
- Dilute the reaction with dichloromethane, concentrate the solution, and purify the crude product via column chromatography using n-hexane and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented hydroboration method offers tangible strategic advantages beyond mere chemical efficiency. The transition from hazardous traditional boranes to stable azacarbene borane adducts fundamentally alters the risk profile of the manufacturing process. By eliminating pyrophoric reagents, facilities can reduce insurance premiums and minimize the need for specialized containment infrastructure, leading to substantial cost savings in capital expenditure and operational overhead. Additionally, the high yields and selectivity reported in the patent examples translate directly into better material utilization, reducing the volume of raw materials required per kilogram of final product and minimizing waste disposal costs associated with failed batches or difficult purifications.
- Cost Reduction in Manufacturing: The use of a low-loading catalyst (5 mol%) combined with commercially available solvents like toluene creates a highly economical process. The elimination of expensive and dangerous borane handling protocols reduces labor costs and safety training requirements. Furthermore, the high conversion rates mean that less starting material is wasted, optimizing the cost of goods sold (COGS) for high-value pharmaceutical intermediates. The process avoids the need for cryogenic conditions or high-pressure reactors, further lowering energy consumption and equipment maintenance costs.
- Enhanced Supply Chain Reliability: The reagents involved, including the azacarbene borane and various substituted stilbenes, are either commercially available or easily synthesized via established routes, ensuring a stable supply chain. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in temperature or mixing, guaranteeing consistent output quality. This reliability is critical for maintaining continuous supply to downstream API manufacturers, reducing the risk of stockouts or delays that can disrupt the entire pharmaceutical production schedule.
- Scalability and Environmental Compliance: The reaction operates at atmospheric pressure and moderate temperatures, making it inherently scalable from gram-scale R&D to multi-ton commercial production without significant re-engineering. The use of stable reagents reduces the generation of hazardous waste streams, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. This green chemistry aspect not only mitigates regulatory risk but also aligns with the sustainability goals of modern chemical enterprises, enhancing the corporate reputation of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boronate synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process feasibility and product quality.
Q: What are the stability advantages of using azacarbene borane over traditional boranes?
A: Unlike traditional borane compounds which are often pyrophoric and unstable, azacarbene borane forms a stable Lewis base-borane adduct. This significantly enhances safety during storage and handling while maintaining high reactivity under catalytic conditions.
Q: What is the optimal catalyst loading for this hydroboration reaction?
A: The patent specifies that tris(pentafluorophenyl)borane (B(C6F5)3) is highly effective at low loadings. A molar amount of 5% relative to the diphenylethylene compound is preferred, though ranges up to 30% are permissible depending on substrate sterics.
Q: Does this method tolerate diverse functional groups on the styrene substrate?
A: Yes, the method exhibits excellent substrate scope. It successfully processes 1,2-diphenylethylene derivatives with various substituents including halogens (F, Cl, Br), alkyl groups (methyl, ethyl, tert-butyl), and electron-donating groups (methoxy), yielding products with high purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boronate Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced hydroboration technology in the synthesis of complex organic molecules. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the laboratory to the marketplace. Our state-of-the-art facilities are equipped to handle the specific requirements of organoboron chemistry, maintaining stringent purity specifications and utilizing rigorous QC labs to verify every batch against the highest industry standards. We are committed to delivering high-purity boronate compounds that meet the exacting demands of global pharmaceutical clients.
We invite you to collaborate with our technical team to explore how this efficient synthesis route can optimize your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your development timelines and reduce overall manufacturing costs.
