Scalable Liquid-Phase Synthesis of NRX-1074 Antidepressant Intermediate for Commercial Production
Introduction to Advanced NRX-1074 Manufacturing Technology
The pharmaceutical landscape for rapid-acting antidepressants has been significantly advanced by the development of NMDA receptor modulators, specifically the tetrapeptide NRX-1074. As detailed in the groundbreaking patent CN108864252B, published on July 28, 2020, a novel and highly efficient preparation method has been established to address the complex synthetic challenges associated with this molecule. Unlike its predecessor GLYX-13, NRX-1074 incorporates a non-natural benzyl-substituted proline residue, which confers superior oral bioavailability but introduces substantial steric hindrance during chemical synthesis. This technical disclosure represents a pivotal shift from labor-intensive racemic separations to a streamlined, chiral pool-based liquid-phase condensation strategy. For global procurement and R&D teams, understanding this proprietary pathway is essential for securing a reliable supply of high-purity pharmaceutical intermediates. The method described ensures that the critical steric barriers are managed through precise temperature control and optimized coupling reagents, resulting in a robust process suitable for industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex tetrapeptides containing unnatural amino acids like benzyl-proline has been plagued by inefficiencies inherent to traditional methodologies. Prior art methods often relied on the initial formation of racemic intermediates, necessitating multiple rounds of preparative high-performance liquid chromatography (HPLC) to isolate the desired optical isomer. This approach is not only economically prohibitive due to the high cost of chiral columns and solvents but also technically limiting for large-scale production. Furthermore, conventional solid-phase peptide synthesis (SPPS) strategies frequently encounter severe difficulties when coupling sterically bulky residues, leading to incomplete reactions, deletion sequences, and notoriously low overall yields. The reliance on FMOC protection strategies in solid-phase contexts often exacerbates these issues, making the purification process arduous and the final cost of goods unsustainably high for commercial antidepressant manufacturing.
The Novel Approach
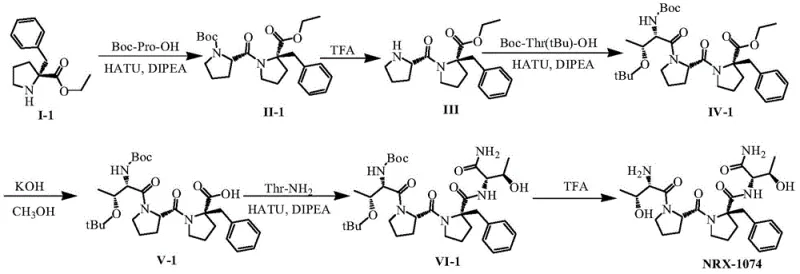
In stark contrast to these legacy techniques, the methodology outlined in patent CN108864252B employs a sophisticated liquid-phase condensation route that strategically circumvents steric hindrance issues. By utilizing optically pure (R)-2-benzyl proline ester as a defined starting material, the process eliminates the need for late-stage racemic resolution. The core innovation lies in the sequential elongation of the peptide chain using highly efficient coupling agents like HATU in conjunction with activated bases such as DIPEA. This specific combination facilitates amide bond formation even in the presence of the bulky benzyl group, maintaining high reaction kinetics. As illustrated in the comprehensive reaction scheme below, the process flows logically from the protected dipeptide intermediate through to the final tetrapeptide, achieving a remarkable final purity of 98.8% without requiring preparative HPLC. This represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, transforming a previously lab-scale curiosity into a commercially viable asset.
Mechanistic Insights into Peptide Condensation and Chiral Resolution
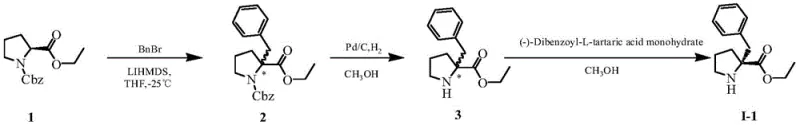
The success of this synthesis hinges on two critical mechanistic pillars: the efficient generation of the chiral building block and the management of steric bulk during coupling. The preparation of the key intermediate, (R)-2-benzyl proline ester, is achieved through a classical yet highly effective resolution process. As shown in the reaction diagram, the racemic ester is treated with L-dibenzoyltartaric acid in methanol, leveraging the differential solubility of diastereomeric salts to isolate the desired (R)-enantiomer with an ee value exceeding 99%. This ensures that the chirality is established early in the sequence, preventing the accumulation of difficult-to-remove impurities later in the synthesis. Following this, the peptide coupling steps are meticulously controlled at low temperatures, typically between 0°C and 5°C. This thermal regulation is crucial for suppressing side reactions such as racemization or epimerization at the alpha-carbon, which are common pitfalls in peptide chemistry. The use of HATU as the coupling reagent generates an active OAt ester intermediate that is sufficiently reactive to overcome the steric repulsion of the benzyl group without requiring excessive heat that could degrade the sensitive peptide backbone.
Furthermore, the impurity profile is tightly controlled through the strategic selection of protecting groups. The use of Boc (tert-butoxycarbonyl) and tBu (tert-butyl ether) protections provides orthogonality that allows for selective deprotection steps. For instance, the removal of the N-terminal Boc group using trifluoroacetic acid (TFA) proceeds cleanly without affecting the side-chain tBu ethers or the C-terminal ester until specifically targeted. This orthogonal stability is vital for maintaining the integrity of the growing peptide chain. The final saponification and amidation steps are similarly optimized; hydrolysis of the ethyl ester is performed under mild alkaline conditions (KOH in methanol) to avoid peptide bond cleavage, while the final coupling with threonine amide utilizes the same high-efficiency HATU/DIPEA system. This consistent mechanistic approach across different coupling nodes simplifies the operational protocol, reducing the risk of human error and ensuring batch-to-batch consistency, which is a primary concern for regulatory compliance in API production.
How to Synthesize NRX-1074 Efficiently
The synthesis of NRX-1074 described in the patent offers a clear, step-by-step protocol that balances chemical efficiency with operational simplicity. The process begins with the rigorous quality control of the chiral starting material, followed by a series of solution-phase couplings that can be monitored via standard analytical techniques like TLC or HPLC. The beauty of this route lies in its modularity; each peptide bond formation is treated as a discrete unit operation with defined workup procedures involving extraction and column chromatography. This modularity allows for easy troubleshooting and optimization at each stage. While the detailed stoichiometric ratios and specific solvent volumes are critical for reproducibility, the general workflow emphasizes the importance of maintaining anhydrous conditions during activation steps and precise pH control during aqueous workups. For process chemists looking to implement this technology, the key takeaway is the elimination of complex purification hardware in favor of robust chemical selectivity. Detailed standardized synthesis steps see the guide below.
- Prepare optically pure (R)-2-benzyl proline ester via chiral resolution of the racemate using L-dibenzoyltartaric acid.
- Perform sequential peptide condensation reactions using HATU and DIPEA at controlled low temperatures (0-5°C) to manage steric hindrance.
- Execute final deprotection steps using trifluoroacetic acid to yield the target tetrapeptide with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound advantages for supply chain stability and cost management. The most significant economic driver is the complete elimination of preparative HPLC purification. In traditional peptide manufacturing, preparative chromatography is often the bottleneck, consuming vast amounts of expensive solvents and stationary phases while limiting throughput. By achieving 98.8% purity through standard crystallization and flash chromatography, this method drastically reduces the cost of goods sold (COGS). This efficiency translates directly into more competitive pricing for downstream API manufacturers. Additionally, the use of readily available reagents like HATU, DIPEA, and common organic solvents (DMF, DCM, Methanol) ensures that the supply chain is not dependent on exotic or single-source catalysts. This accessibility enhances supply continuity, mitigating the risk of production delays caused by raw material shortages. The robustness of the liquid-phase chemistry also implies that the process is less sensitive to minor fluctuations in reaction parameters, further securing the reliability of supply.
- Cost Reduction in Manufacturing: The economic benefits of this process are primarily derived from the simplification of the purification train. By avoiding the need for chiral preparative HPLC, manufacturers save substantially on consumables and equipment maintenance. The high overall yield of 26.1% from the chiral ester starting point is exceptionally favorable for a tetrapeptide of this complexity, minimizing waste and maximizing the output per kilogram of input material. Furthermore, the ability to perform reactions at near-ambient or mildly cooled temperatures (0-5°C) rather than cryogenic conditions reduces energy consumption for cooling infrastructure. These cumulative factors contribute to a leaner manufacturing model that supports substantial cost savings without compromising on the stringent quality requirements necessary for clinical-grade materials.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals and standard unit operations. The reagents specified, such as Boc-protected amino acids and carbodiimide coupling agents, are produced by multiple global suppliers, preventing vendor lock-in. The process does not rely on sensitive transition metal catalysts that might require specialized removal steps or pose contamination risks, simplifying the quality control workflow. This chemical robustness ensures that production schedules can be maintained consistently, reducing lead time for high-purity pharmaceutical intermediates. For procurement managers, this means a lower risk of batch failure and a more predictable delivery timeline, which is critical for aligning with clinical trial milestones or commercial launch dates.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is facilitated by the solution-phase nature of the synthesis. Unlike solid-phase synthesis, which faces mixing and diffusion limitations at large scales, liquid-phase reactions can be easily scaled in standard stirred-tank reactors. The workup procedures involve standard liquid-liquid extractions and filtrations, which are well-understood unit operations in chemical engineering. Moreover, the avoidance of heavy metal catalysts and the reduction in solvent usage associated with HPLC purification align with green chemistry principles. This reduces the environmental footprint of the manufacturing process, easing the burden on waste treatment facilities and ensuring compliance with increasingly strict environmental regulations. The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, capable of moving from 100 kgs to 100 MT annual production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of NRX-1074 based on the disclosed patent technology. These insights are derived directly from the experimental data and process descriptions provided in the intellectual property documentation. Understanding these nuances is critical for stakeholders evaluating the feasibility of this route for their specific supply chain needs. The answers reflect the balance between chemical rigor and practical manufacturability that defines this innovative approach.
Q: Why is the liquid-phase method preferred over solid-phase synthesis for NRX-1074?
A: Due to the significant steric hindrance introduced by the unnatural benzyl-substituted proline residue, solid-phase synthesis often results in difficult couplings and low yields. The patented liquid-phase method optimizes reaction conditions to overcome this spatial barrier.
Q: Does this synthesis route require preparative HPLC purification?
A: No, a key advantage of this method is that it achieves a final product purity of 98.8% through standard column chromatography and crystallization, eliminating the need for costly and time-consuming preparative HPLC separation.
Q: What is the overall yield of the NRX-1074 synthesis described in the patent?
A: The patent reports a total overall yield of 26.1% calculated from the (R)-2-benzylproline ethyl ester starting material, which is considered high for a tetrapeptide containing sterically hindered unnatural amino acids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NRX-1074 Supplier
As the demand for next-generation antidepressants grows, securing a partner with the technical capability to execute complex peptide syntheses is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in peptide chemistry to deliver high-quality intermediates. Our facility is equipped to handle the specific challenges of sterically hindered couplings and chiral resolutions described in patent CN108864252B. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that in the pharmaceutical industry, purity is not just a metric but a safety imperative, and our processes are designed to consistently exceed the 98% purity threshold required for clinical applications.
We invite you to collaborate with us to optimize your supply chain for NRX-1074 and related peptide therapeutics. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable pharmaceutical intermediate supplier dedicated to accelerating your drug development timeline through superior chemical manufacturing solutions. Let us help you navigate the complexities of peptide production with confidence and efficiency.
