Advanced Synthesis of N-Heteroatom Polycyclic Indolizines for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex nitrogen-containing heterocycles, which serve as the backbone for numerous bioactive molecules. Patent CN114773340A introduces a groundbreaking advancement in this domain by disclosing a novel synthesis process for N-heteroatom polycyclic indolizines and their derivatives. These structures are pivotal in the development of active pharmaceutical ingredients (APIs), agrochemicals, and optoelectronic materials due to their unique biological activities and electronic properties. The disclosed technology addresses critical bottlenecks in existing synthetic routes by offering a pathway that is not only chemically efficient but also economically viable for large-scale production. By leveraging a titanium tetrachloride-mediated cyclization strategy, this invention enables the construction of intricate polycyclic frameworks with high precision and selectivity. For R&D directors and procurement specialists, this represents a significant opportunity to streamline supply chains for high-value intermediates while adhering to stricter environmental and cost-efficiency standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indolizine derivatives has been plagued by several inherent inefficiencies that hinder commercial scalability. Traditional methods often rely on the use of expensive precious metal catalysts, such as palladium or rhodium complexes, which drastically inflate the raw material costs and necessitate rigorous metal removal steps to meet pharmaceutical purity specifications. Furthermore, many conventional protocols require harsh reaction conditions, including extreme temperatures or pressures, which pose safety risks and increase energy consumption. Another significant drawback is the poor functional group compatibility observed in older methodologies; sensitive moieties often degrade under the required reaction conditions, leading to low overall yields and complex impurity profiles that are difficult to purify. These factors collectively contribute to extended lead times and reduced process reliability, making it challenging for supply chain heads to guarantee consistent delivery of high-quality intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN114773340A employs a cost-effective and operationally simple strategy centered around the use of titanium tetrachloride as a Lewis acid catalyst. This approach eliminates the dependency on precious metals, thereby offering substantial cost reduction in pharmaceutical intermediate manufacturing. The reaction proceeds under mild conditions, typically at or below 10°C during reagent addition, which preserves the integrity of sensitive functional groups and minimizes the formation of unwanted by-products. The process demonstrates exceptional versatility, accommodating a wide array of substituents including halogens, nitro groups, and various aromatic systems without compromising yield. This high level of selectivity simplifies the downstream purification process, often requiring only standard extraction and chromatography techniques. Consequently, this novel route provides a reliable pharmaceutical intermediate supplier with the ability to deliver complex molecules with greater speed and consistency.
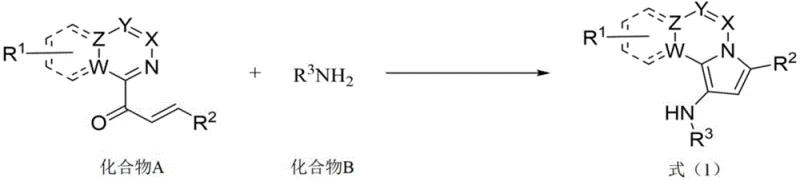
Mechanistic Insights into TiCl4-Catalyzed Cyclization
The core of this innovative synthesis lies in the mechanistic role of titanium tetrachloride (TiCl4) acting in concert with triethylamine (Et3N). In this catalytic system, TiCl4 functions as a potent Lewis acid that activates the carbonyl group of the alpha,beta-unsaturated ketone precursor, rendering it more susceptible to nucleophilic attack. Simultaneously, it facilitates the activation of the imine intermediate formed in situ, promoting the crucial cyclization step that constructs the indolizine core. Triethylamine plays a dual role in this ecosystem; it acts as a base to neutralize the hydrochloric acid generated during the reaction, maintaining a weakly alkaline environment that stabilizes the titanium species and prevents catalyst deactivation. This synergistic interaction ensures a smooth reaction trajectory with high atom economy. The mechanism avoids the formation of radical intermediates often seen in transition metal catalysis, which contributes to the cleaner reaction profile and reduced impurity burden observed in the final product.
From an impurity control perspective, the specificity of this Lewis acid-mediated pathway is paramount. The reaction conditions are finely tuned to favor the desired intramolecular cyclization over competing intermolecular polymerization or side reactions. The use of an inert atmosphere, such as nitrogen, further protects the reactive intermediates from oxidation, ensuring that the final product maintains stringent purity specifications. The patent data indicates that even with sterically demanding substrates, such as fused ring systems, the reaction proceeds with commendable efficiency, although yields may vary slightly due to steric hindrance. This mechanistic robustness allows for the synthesis of a diverse library of derivatives, providing medicinal chemists with a powerful tool for structure-activity relationship (SAR) studies without being constrained by synthetic feasibility.
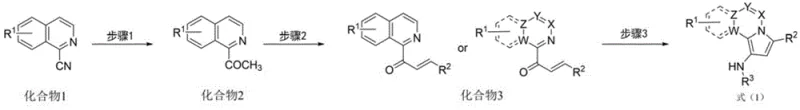
How to Synthesize N-Heteroatom Polycyclic Indolizines Efficiently
The synthesis protocol described in the patent is designed for practical implementation in both laboratory and pilot plant settings. It follows a logical three-step sequence that begins with readily available cyano-substituted heterocycles. The initial step involves the conversion of these precursors into acetyl derivatives using Grignard reagents, followed by an aldol condensation to install the necessary unsaturated side chain. The final and most critical step is the cyclization reaction, where the pre-functionalized ketone reacts with a sulfonamide in the presence of the titanium catalyst system. This modular approach allows for the late-stage diversification of the molecule, enabling the rapid generation of analogues. Detailed standardized synthesis steps see the guide below.
- Synthesize acetyl N-heteroaromatic compounds (Compound 2) from cyano precursors using Grignard reagents.
- Perform aldol condensation with various aldehydes to generate alpha,beta-unsaturated carbonyl compounds (Compound 3).
- Execute the key cyclization step using titanium tetrachloride and triethylamine with sulfonamides to yield the final indolizine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis technology translates into tangible strategic benefits that extend beyond mere chemical curiosity. The shift away from precious metal catalysts to abundant Lewis acids like titanium tetrachloride fundamentally alters the cost structure of the manufacturing process. This change eliminates the volatility associated with precious metal markets and removes the need for expensive scavenging resins or complex filtration units required to meet residual metal limits in APIs. Furthermore, the simplicity of the workup procedure, which relies on common solvents and standard separation techniques, reduces the operational burden on production facilities. This efficiency gain allows for faster batch turnover and improved asset utilization, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The replacement of costly noble metal catalysts with inexpensive titanium tetrachloride results in a drastic reduction in raw material expenses. Additionally, the high reaction selectivity minimizes waste generation and reduces the solvent volume required for purification, leading to substantial cost savings in waste disposal and solvent recovery. The overall process efficiency ensures that the cost per kilogram of the final intermediate is significantly lower compared to traditional methods, enhancing profit margins for downstream drug products.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including various substituted benzaldehydes and heterocyclic nitriles, are commodity chemicals with stable and robust global supply chains. This availability mitigates the risk of production delays caused by raw material shortages. Moreover, the mild reaction conditions reduce the likelihood of equipment failure or safety incidents, ensuring continuous and uninterrupted production schedules. This reliability is crucial for maintaining just-in-time inventory levels and meeting the demanding delivery timelines of multinational pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively across a range of substrate complexities without requiring specialized high-pressure reactors. The reduced use of hazardous reagents and the generation of less toxic by-products align with modern green chemistry principles and increasingly strict environmental regulations. This compliance simplifies the permitting process for new manufacturing lines and reduces the long-term liability associated with environmental footprint, making it a sustainable choice for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on its practical application and potential limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this new indolizine synthesis method over traditional routes?
A: The novel method utilizes inexpensive and commercially available catalysts like titanium tetrachloride instead of precious metals. It offers superior functional group compatibility, higher reaction selectivity, and a simplified post-treatment process, significantly reducing environmental impact and operational complexity.
Q: Can this synthesis protocol accommodate diverse functional groups for drug discovery?
A: Yes, the process demonstrates excellent functional group tolerance. It successfully accommodates electron-donating and electron-withdrawing groups, heterocycles, fused rings, and aliphatic chains, making it highly versatile for generating diverse libraries of pharmaceutical intermediates.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Absolutely. The reaction conditions are mild, utilizing standard solvents like dichloromethane and common reagents. The straightforward workup involving extraction and silica gel chromatography ensures that the process can be scaled up efficiently from laboratory to industrial production without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Heteroatom Polycyclic Indolizine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis methods described in CN114773340A for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N-heteroatom polycyclic indolizines meets the highest industry standards. We are committed to leveraging this advanced chemistry to deliver cost-effective solutions that accelerate your drug development timelines.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your target molecule. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to be your trusted partner in bringing next-generation therapeutics to market efficiently and reliably.
