Advanced Silane-Protection Strategy for High-Purity Tert-Butylsulfinamide Manufacturing
The pharmaceutical and fine chemical industries continuously seek robust methodologies for synthesizing chiral building blocks, particularly those serving as critical auxiliaries in asymmetric synthesis. Patent CN101735118B introduces a transformative process for synthesizing tert-butylsulfinamide, a cornerstone reagent often referred to as Ellman's auxiliary, by utilizing silane as a protective reagent. This innovation addresses longstanding challenges associated with the conventional production of this vital intermediate, specifically targeting the harsh reaction conditions and environmental burdens of previous methods. By shifting from a cryogenic liquid ammonia system to a silane-protected pathway, the technology offers a more sustainable and operationally feasible route for manufacturing high-purity chiral amines. For R&D directors and procurement specialists, understanding this shift is crucial for optimizing supply chains and reducing the total cost of ownership for complex chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tert-butylsulfinamide has relied heavily on the direct reaction of lithium amide with tert-butylsulfinic acid thio-tert-butyl ester in liquid ammonia, a process that demands extreme cryogenic conditions. As illustrated in the traditional pathway, this method requires maintaining temperatures between -78°C and -72°C, necessitating specialized refrigeration equipment and significant energy consumption to sustain such ultra-low thermal environments. Furthermore, the use of liquid ammonia as a bulk solvent introduces substantial safety risks and logistical complexities, including the need for pressure-rated vessels and rigorous containment protocols to prevent toxic leaks. The stoichiometric requirement for a massive excess of ammonia relative to lithium further complicates the post-reaction workup, as the volatilization of ammonia must be carefully managed to avoid product loss or degradation. These factors collectively create a high barrier to entry for manufacturers, driving up capital expenditure and limiting the scalability of the process for commercial applications.
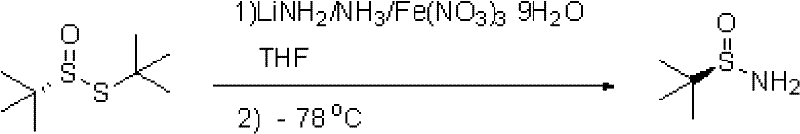
The Novel Approach
In stark contrast to the legacy methods, the patented silane-protection strategy fundamentally reengineers the synthetic logic by introducing a tert-butyldimethylsilyl (TBDMS) group to mask the reactive amine functionality during the critical coupling step. This approach allows the reaction to proceed at significantly milder temperatures, ranging from -40°C to -15°C, which are easily achievable with standard industrial cooling systems rather than exotic cryogenic setups. The introduction of the silyl group not only stabilizes the nitrogen center against unwanted side reactions but also enhances the solubility of the intermediate in common organic solvents like tetrahydrofuran (THF) and pentane. By avoiding the use of bulk liquid ammonia, the process drastically simplifies the reactor design and eliminates the environmental hazards associated with ammonia emissions. This technological leap represents a paradigm shift in how chiral sulfinamides are manufactured, offering a reliable pharmaceutical intermediate supplier pathway that balances efficiency with safety.
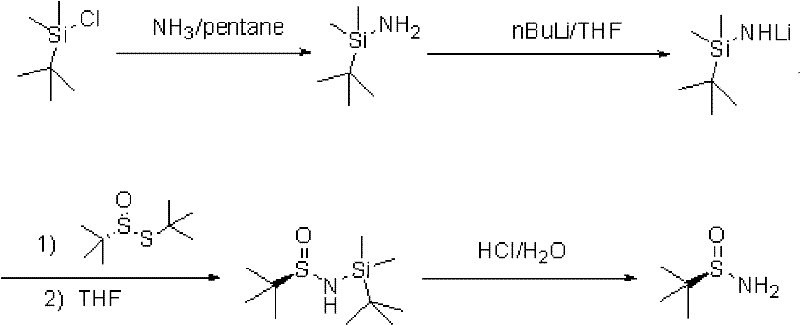
Mechanistic Insights into Silane-Protected Amine Synthesis
The core mechanistic advantage of this process lies in the strategic use of the silyl group to modulate the nucleophilicity and steric environment of the nitrogen atom during the formation of the sulfur-nitrogen bond. Initially, tert-butyldimethylsilyl chloride reacts with ammonia to form tert-butyldimethylaminosilane, which is subsequently lithiated using n-butyllithium to generate the key nucleophile, N-tert-butyldimethylsilyl-lithium amide. This lithiated species is sufficiently reactive to attack the sulfur center of the sulfinic thioester but is sterically shielded by the bulky silyl group, preventing over-alkylation or polymerization that often plagues unprotected amine syntheses. The reaction proceeds smoothly in THF, where the solvation of the lithium cation facilitates the nucleophilic substitution without the need for the extreme solvation power of liquid ammonia. Following the coupling, the silyl group acts as a temporary mask that can be cleanly removed under mild acidic conditions, revealing the free amine without racemization or decomposition of the sensitive sulfinyl moiety.
Impurity control is another critical aspect where this mechanism excels, as the stepwise nature of the silylation and deprotection allows for intermediate purification that is impossible in the one-pot liquid ammonia method. The patent data indicates that the final product achieves a gas chromatography (GC) purity of greater than 99%, a testament to the effectiveness of the silyl group in suppressing side reactions. The deprotection step utilizes dilute hydrochloric acid at room temperature, a gentle condition that ensures the stereochemical integrity of the chiral sulfur center is maintained throughout the process. For quality assurance teams, this means a much cleaner impurity profile, reducing the burden on downstream purification steps and ensuring that the final chiral auxiliary meets the stringent specifications required for API synthesis. The ability to isolate the protected intermediate also provides a valuable checkpoint for quality control before the final deprotection.
How to Synthesize Tert-Butylsulfinamide Efficiently
The implementation of this silane-protected route involves a logical sequence of three distinct chemical transformations that can be seamlessly integrated into existing multipurpose reactor trains. The process begins with the formation of the silyl amine, followed by lithiation and coupling with the sulfinyl precursor, and concludes with an aqueous acidic workup to liberate the final product. Each step is designed to maximize yield while minimizing the generation of hazardous waste, aligning with modern green chemistry principles. The detailed standardized synthesis steps below outline the precise conditions and stoichiometry required to replicate the high yields reported in the patent documentation.
- Synthesize N-tert-butyldimethylsilyl-lithium amide by reacting tert-butyldimethylsilyl chloride with liquid ammonia followed by lithiation with n-butyllithium in THF.
- React the lithiated silyl amine with (S or R)-tert-butylsulfinic acid thio-tert-butyl ester at controlled low temperatures (-40°C to -15°C) to form the protected sulfinamide intermediate.
- Hydrolyze the silyl protecting group using dilute hydrochloric acid at room temperature to yield the final tert-butylsulfinamide product with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this silane-based methodology offers profound economic and logistical benefits that extend far beyond simple yield improvements. By eliminating the dependency on cryogenic infrastructure capable of reaching -78°C, manufacturers can significantly reduce capital investment in specialized refrigeration units and lower the ongoing energy costs associated with maintaining such extreme temperatures. The substitution of liquid ammonia with standard organic solvents like pentane and THF simplifies raw material sourcing and storage, as these chemicals are widely available and do not require the same level of regulatory oversight as anhydrous ammonia. This shift effectively de-risks the supply chain, ensuring continuity of supply even in regions with strict environmental regulations regarding ammonia usage. Furthermore, the milder reaction conditions enhance operator safety, potentially lowering insurance premiums and reducing the likelihood of production stoppages due to safety incidents.
- Cost Reduction in Manufacturing: The elimination of ultra-low temperature requirements translates directly into substantial operational expenditure savings, as the energy load for cooling is drastically reduced compared to cryogenic processes. Additionally, the removal of liquid ammonia from the solvent system avoids the costs associated with its recovery, neutralization, and disposal, leading to a leaner and more cost-effective manufacturing workflow. The high yields reported in the patent examples suggest that raw material utilization is optimized, further driving down the cost per kilogram of the final chiral auxiliary. These cumulative efficiencies allow for a more competitive pricing structure for high-purity pharmaceutical intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing common organic solvents and reagents like tert-butyldimethylsilyl chloride ensures that the supply chain is resilient to disruptions that might affect specialized chemical markets. The process does not rely on single-source suppliers for exotic catalysts or hard-to-source cryogens, making it easier to qualify multiple vendors for raw materials. This diversification of the supply base is critical for maintaining consistent production schedules and meeting the just-in-time delivery expectations of global pharmaceutical clients. The robustness of the chemistry also means that batch-to-batch variability is minimized, providing customers with a reliable source of material that consistently meets specification.
- Scalability and Environmental Compliance: The mild operating conditions and absence of toxic ammonia emissions make this process inherently easier to scale from pilot plant to commercial production volumes. Regulatory compliance is simplified as the process generates less hazardous waste and avoids the strict permitting requirements associated with large-scale ammonia handling. This environmental friendliness aligns with the increasing corporate sustainability goals of major pharmaceutical companies, making the manufacturer a preferred partner for green chemistry initiatives. The ability to scale safely and sustainably ensures that supply can grow in tandem with market demand for chiral drugs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silane-protected synthesis route for tert-butylsulfinamide. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and quality benchmarks associated with this technology. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement professionals assessing supplier capabilities.
Q: How does the silane protection method improve safety compared to traditional liquid ammonia processes?
A: The silane protection strategy eliminates the need for maintaining ultra-low cryogenic temperatures of -78°C and reduces the reliance on large volumes of liquid ammonia as a solvent, significantly lowering operational hazards and equipment costs.
Q: What is the expected purity profile of tert-butylsulfinamide produced via this route?
A: According to the patent data, the final product achieves a GC purity greater than 99% after simple slurry purification, indicating excellent impurity control suitable for sensitive asymmetric synthesis applications.
Q: Is this synthesis route scalable for industrial production of chiral intermediates?
A: Yes, the process operates at milder temperatures (-40°C to -15°C) and uses standard organic solvents like THF and pentane, making it highly adaptable for commercial scale-up without requiring specialized cryogenic infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tert-Butylsulfinamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral auxiliaries play in the development of next-generation therapeutics, and we are committed to delivering excellence in every batch. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that our tert-butylsulfinamide meets the highest industry standards for asymmetric synthesis. Our commitment to process innovation allows us to offer products that are not only chemically superior but also economically advantageous for your manufacturing operations.
We invite you to engage with our technical procurement team to discuss how our advanced silane-protection technology can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to our process. We encourage potential partners to contact us for specific COA data and route feasibility assessments to verify our capability to support your project from clinical trials through to commercial launch. Let us be your trusted partner in navigating the complexities of chiral intermediate manufacturing.
