Advanced Zirconium-Catalyzed C7 Alkylation for Scalable Pharmaceutical Intermediate Manufacturing
Advanced Zirconium-Catalyzed C7 Alkylation for Scalable Pharmaceutical Intermediate Manufacturing
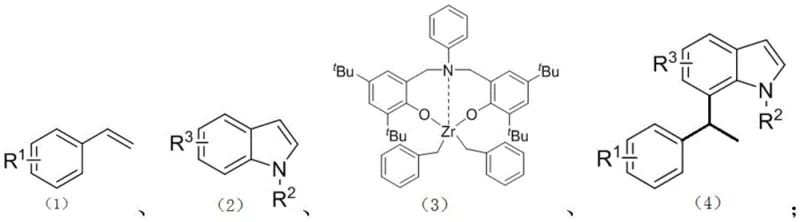
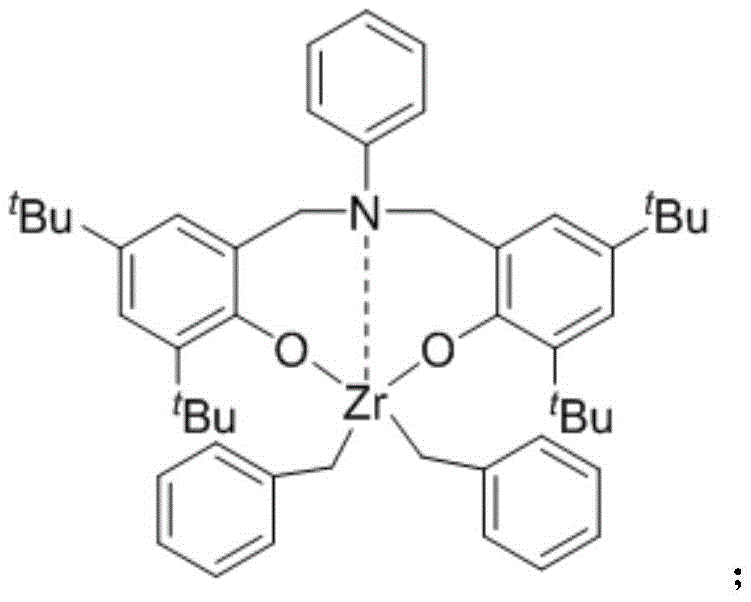
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the relentless pursuit of more efficient, cost-effective, and regioselective methodologies. A significant breakthrough in this domain is documented in patent CN109734644B, which details a novel method for the catalytic C7-position alkylation of N-alkylindoles. This technology addresses a long-standing challenge in organic synthesis: the direct functionalization of the sterically hindered and electronically less favorable C7 position of the indole ring without relying on pre-installed directing groups. By leveraging a cooperative catalytic system involving triphenylcarbotetrakis(pentafluorophenyl)borate and a specialized aniline-bridged bisaryloxyzirconium complex, this invention offers a robust pathway to access valuable indole derivatives that are critical scaffolds in drug discovery and agrochemical development.
For R&D directors and process chemists, the implications of this patent are profound. Traditional methods often struggle with selectivity, frequently yielding mixtures of C2 and C3 alkylated products, or requiring harsh conditions that compromise sensitive functional groups. The approach outlined in CN109734644B operates under relatively mild thermal conditions, typically between 110°C and 130°C, in standard aprotic solvents. This level of control not only simplifies the purification workflow but also enhances the overall safety profile of the manufacturing process. As we delve deeper into the technical specifics, it becomes clear that this zirconium-based strategy represents a viable, scalable alternative to noble metal catalysis, aligning perfectly with the industry's shift towards sustainable and economically feasible chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct alkylation of indoles has been dominated by reactions occurring at the C2 and C3 positions due to their inherent nucleophilicity. Achieving selectivity at the C7 position has traditionally required the installation of bulky or coordinating directing groups, which adds synthetic steps and generates additional waste. Furthermore, many existing protocols rely heavily on precious metal catalysts, particularly Rhodium (Rh), which are not only exorbitantly expensive but also subject to volatile market pricing and supply chain constraints. The removal of trace heavy metals from the final active pharmaceutical ingredient (API) is another critical bottleneck, often necessitating complex scavenging procedures that drive up production costs and extend lead times. These factors collectively render conventional C7 functionalization strategies less attractive for large-scale commercial applications where cost margins and supply reliability are paramount.
The Novel Approach
The methodology presented in the patent circumvents these hurdles by employing an earth-abundant zirconium catalyst system that inherently favors C7 activation. The core of this innovation lies in the specific design of the aniline-bridged bisaryloxyzirconium complex, which creates a steric and electronic environment conducive to C-H bond activation at the desired position. Unlike previous methods that might require stoichiometric amounts of oxidants or harsh Lewis acids, this co-catalytic system utilizes a boron activator to generate a highly reactive cationic zirconium species in situ. This allows for the direct coupling of readily available styrenes with N-alkylindoles in a single step. The operational simplicity is further enhanced by the use of common solvents like chlorobenzene or toluene, making the transition from bench-scale optimization to pilot plant production seamless and straightforward for any competent chemical manufacturing facility.
Mechanistic Insights into Zirconium-Catalyzed C-H Activation
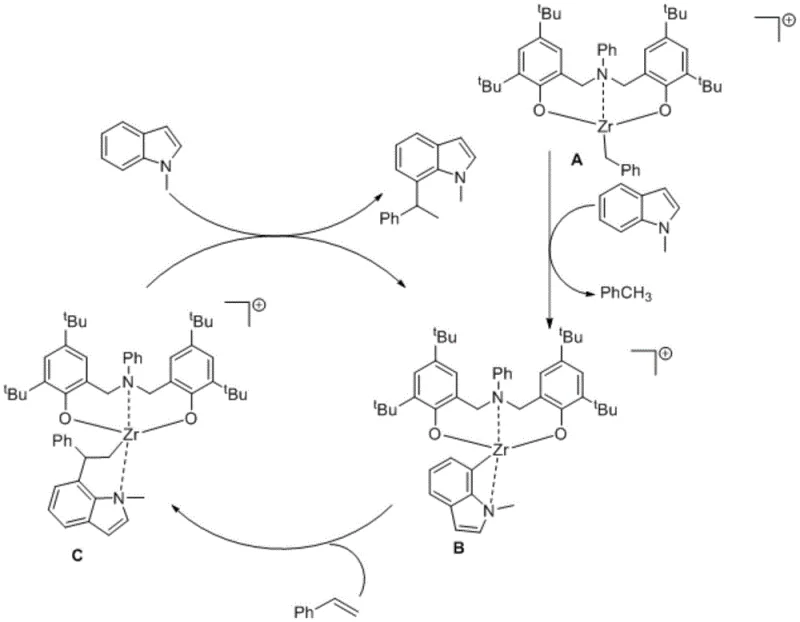
Understanding the mechanistic underpinnings of this transformation is crucial for process optimization and troubleshooting. The reaction initiates with the interaction between the neutral zirconium precursor and the boron activator, [Ph3C][B(C6F5)4], to form a cationic zirconium species. This electrophilic metal center is key to the subsequent C-H activation step. It coordinates with the nitrogen atom of the N-alkylindole, positioning the metal in close proximity to the C7 hydrogen. Through a sigma-bond metathesis or electrophilic substitution pathway, the Zr-C7 bond is formed, releasing a molecule of alkane or protonated species depending on the exact mechanistic nuance. This step is the turnover-limiting determinant of regioselectivity, effectively blocking the more reactive C2/C3 sites through steric shielding provided by the bulky tert-butyl groups on the ligand framework.
Following the formation of the zirconium-indole intermediate, the styrene substrate undergoes migratory insertion into the Zr-C bond. This step constructs the new carbon-carbon bond that defines the alkylated product. The resulting alkyl-zirconium species then undergoes protonolysis with a second molecule of the indole substrate, regenerating the active catalytic species and releasing the final C7-alkylated product. This catalytic cycle, depicted clearly in the patent literature, highlights the atom-economic nature of the process, as the only byproduct is typically a simple hydrocarbon. The robustness of this cycle allows for high turnover numbers, ensuring that even with low catalyst loadings (typically around 5 mol%), excellent yields ranging from 76% to 90% can be consistently achieved across a diverse range of substrates.
How to Synthesize C7-Alkylated N-Alkylindoles Efficiently
Implementing this synthesis route requires careful attention to reaction conditions, particularly regarding moisture and oxygen sensitivity, which are common characteristics of organozirconium chemistry. The patent provides a clear blueprint for execution, emphasizing the need for an inert atmosphere such as argon or nitrogen to prevent catalyst deactivation. The procedure involves dissolving the catalyst components in an aprotic solvent, activating the system, and then introducing the substrates. While the specific stoichiometric ratios can be tuned—for instance, using an excess of styrene to drive the reaction to completion—the core protocol remains consistent. For a comprehensive, step-by-step guide tailored to your specific equipment and scale requirements, please refer to the standardized synthesis protocol provided below.
- Prepare the catalytic system by mixing triphenylcarbotetrakis(pentafluorophenyl)borate and the aniline-bridged bisaryloxyzirconium complex in an aprotic solvent like chlorobenzene under inert atmosphere.
- Add the styrene derivative and N-alkylindole substrate to the activated catalyst solution, ensuring strict anhydrous and oxygen-free conditions are maintained throughout the process.
- Heat the reaction mixture to 110-130°C for approximately 24 hours, then purify the resulting C7-alkylated product via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this zirconium-catalyzed technology offers substantial strategic advantages over legacy methods. The most immediate impact is seen in raw material costs. By replacing expensive rhodium catalysts with zirconium complexes and commercially available boron salts, the direct material cost of the catalytic system is drastically reduced. Furthermore, the ligands used in the zirconium complex are synthesized from readily available precursors, ensuring a stable and reliable supply chain that is not subject to the geopolitical volatility often associated with platinum group metals. This stability translates directly into more predictable pricing models for long-term contracts, allowing procurement managers to budget with greater confidence and reduce the risk of cost overruns due to catalyst price spikes.
- Cost Reduction in Manufacturing: The elimination of noble metals removes the necessity for expensive metal scavenging resins and rigorous heavy metal testing, which are significant cost drivers in API manufacturing. Additionally, the high yields reported (up to 90%) minimize the loss of valuable starting materials, thereby improving the overall mass balance and reducing the cost per kilogram of the final product. The simplified workup procedure, often requiring only standard column chromatography or recrystallization, further reduces labor and solvent consumption costs associated with complex purification trains.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as styrenes and simple N-alkylindoles ensures that the supply chain is robust and resilient. These starting materials are produced on a massive scale globally, meaning that sourcing disruptions are highly unlikely. Moreover, the catalyst components themselves are either commercially available or easily synthesized from stable precursors, removing the dependency on specialized catalyst vendors who might have long lead times or limited production capacity. This decentralization of supply risk is critical for maintaining continuous manufacturing operations.
- Scalability and Environmental Compliance: The reaction conditions are compatible with standard stainless steel reactors and do not require exotic high-pressure equipment, facilitating easy scale-up from grams to tons. The use of common solvents like chlorobenzene or toluene allows for established solvent recovery and recycling protocols, significantly reducing hazardous waste generation. This alignment with green chemistry principles not only lowers waste disposal costs but also ensures compliance with increasingly stringent environmental regulations, safeguarding the facility against potential regulatory fines or shutdowns.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common questions regarding the reaction scope, limitations, and operational parameters. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a realistic view of what can be achieved in a production environment. Understanding these nuances is essential for successful technology transfer and process validation.
Q: What are the primary advantages of this Zirconium-catalyzed method over traditional Rhodium catalysis?
A: This method eliminates the need for expensive noble metal Rhodium catalysts and complex directing groups, significantly reducing raw material costs while achieving high regioselectivity at the C7 position.
Q: What is the substrate scope for this C7 alkylation reaction?
A: The process demonstrates broad applicability, accommodating various styrene derivatives including those with halogen substituents (fluoro, chloro) and different N-alkylindoles with methyl or ethyl groups.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of commercially available boron reagents, simple zirconium complexes, and standard aprotic solvents makes the process highly scalable and operationally simple for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkylindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN109734644B. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these laboratory innovations into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to market is smooth and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of C7-alkylated indole intermediates meets the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cost-effective synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this zirconium-catalyzed process for your specific molecule. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your supply chain.
