Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Pharmaceutical Applications
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Pharmaceutical Applications
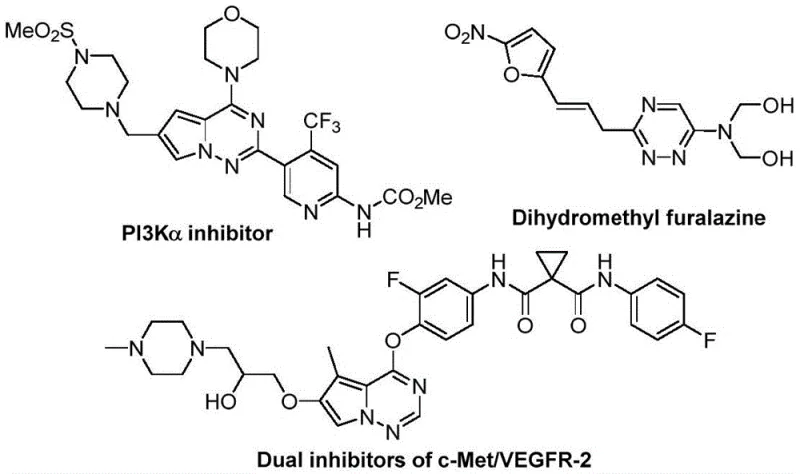
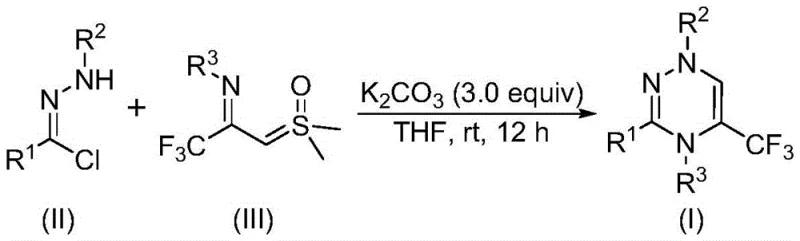
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective methods to construct nitrogen-containing heterocycles, particularly those incorporating fluorine atoms which often enhance metabolic stability and bioavailability. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds, addressing critical bottlenecks in current synthetic methodologies. This innovation utilizes a synergistic [3+3] cycloaddition strategy between chlorohydrazones and trifluoroacetyl sulfur ylides, promoted by inexpensive potassium carbonate. Unlike traditional routes that often rely on harsh conditions or toxic transition metals, this novel approach operates efficiently at room temperature under an air atmosphere. For R&D directors and procurement managers alike, this represents a significant leap forward in accessing high-value scaffolds found in numerous bioactive molecules, ranging from anticancer agents to antifungal drugs, thereby streamlining the supply chain for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has relied heavily on the condensation reactions of amidrazones with 1,2-diketones or alkynes, as well as multicomponent reactions involving hydrazides and dicarbonyl compounds. While these classical methods have served the community for decades, they suffer from inherent inefficiencies that hinder modern high-throughput drug discovery and large-scale manufacturing. Conventional protocols frequently require elevated temperatures, strictly anhydrous conditions, or the use of stoichiometric amounts of activating agents that generate substantial waste. Furthermore, the structural diversity achievable through these older pathways is often limited by the availability and stability of the requisite 1,2-dicarbonyl precursors. For supply chain heads, these limitations translate into higher raw material costs, complex waste treatment protocols, and potential delays due to the sensitivity of the reaction conditions to environmental factors like moisture and oxygen.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN116253692A offers a streamlined, metal-free alternative that fundamentally reshapes the synthetic landscape for these heterocycles. By employing chlorohydrazones and trifluoroacetyl sulfur ylides as key building blocks, the reaction proceeds via a unique mechanism that avoids the need for precious metal catalysts entirely. The use of potassium carbonate as a mild base promoter not only reduces reagent costs drastically but also simplifies the post-reaction workup, as inorganic salts are easily removed by filtration. This approach allows for the direct introduction of the trifluoromethyl group, a critical pharmacophore, without requiring separate fluorination steps that can be hazardous and low-yielding. The ability to run the reaction in common organic solvents like tetrahydrofuran at ambient temperatures significantly lowers the energy footprint of the process, aligning perfectly with green chemistry principles and reducing the total cost of ownership for manufacturing facilities.
Mechanistic Insights into Potassium Carbonate-Promoted [3+3] Cycloaddition
The mechanistic elegance of this transformation lies in the generation of a reactive nitrile imine intermediate in situ. Under the promotion of potassium carbonate, the chlorohydrazone undergoes dehydrohalogenation to release hydrogen chloride, forming the highly reactive nitrile imine species. This intermediate then engages in a concerted [3+3] cycloaddition with the trifluoroacetyl sulfur ylide. This step is crucial as it constructs the six-membered triazine ring while simultaneously eliminating a molecule of dimethyl sulfoxide. The reaction can also be conceptualized as proceeding through intermolecular nucleophilic addition followed by intramolecular nucleophilic substitution, ultimately yielding the stable aromatic triazine system. This pathway is exceptionally clean, minimizing the formation of polymeric byproducts or regioisomers that often plague condensation reactions, thus ensuring a high-purity crude product that requires minimal purification effort.
From an impurity control perspective, the absence of transition metals is a paramount advantage for pharmaceutical applications. Heavy metal residues are a major regulatory concern, often necessitating expensive scavenging resins or complex recrystallization processes to meet ICH Q3D guidelines. By utilizing an organic base and sulfur ylide chemistry, this method inherently produces a metal-free product profile. Additionally, the mild reaction conditions (20-40°C) prevent thermal degradation of sensitive functional groups that might be present on the aromatic rings of the substrates. This tolerance for diverse functional groups, including halogens, alkoxy, and nitro substituents, allows medicinal chemists to explore a vast chemical space without worrying about compatibility issues. The result is a robust process capable of delivering high-purity intermediates suitable for direct progression into preclinical and clinical supply chains.
How to Synthesize Trifluoromethyl Substituted 1,2,4-Triazine Efficiently
The operational simplicity of this synthesis makes it accessible for both laboratory-scale optimization and pilot-plant production. The protocol involves simply mixing the three key components—chlorohydrazone, trifluoroacetyl sulfur ylide, and potassium carbonate—in a suitable organic solvent such as tetrahydrofuran. The mixture is then stirred at room temperature for a period of 10 to 14 hours, requiring no specialized equipment like gloveboxes or high-pressure reactors. Upon completion, the reaction mixture is filtered to remove the inorganic salts, and the filtrate is concentrated. The crude product can be further purified using standard silica gel column chromatography to afford the target triazine compound in high yields. This straightforward workflow minimizes operator training requirements and reduces the risk of batch-to-batch variability, which is essential for maintaining consistent quality in commercial manufacturing.
- Mix potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide in an organic solvent like THF.
- Stir the reaction mixture at room temperature (20-40°C) for 10-14 hours under an air atmosphere.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this synthetic route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The elimination of heavy metal catalysts removes a significant cost center associated with catalyst procurement, recovery, and the rigorous analytical testing required to certify low residual metal levels. Furthermore, the reliance on commodity chemicals like potassium carbonate and widely available organic solvents ensures a stable and resilient supply chain, insulating production schedules from the volatility often seen in the market for specialized reagents. The mild reaction conditions also imply lower energy consumption, as there is no need for heating mantles, cryogenic cooling, or inert gas blanketing, leading to substantial operational expenditure savings over the lifecycle of the product.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound. By removing the dependency on expensive palladium, copper, or rhodium catalysts, the direct material cost per kilogram of the intermediate is significantly reduced. Additionally, the simplified workup procedure, which primarily involves filtration and standard chromatography rather than complex metal scavenging or distillation, reduces labor hours and solvent usage. This leaner process flow translates directly into improved margins for contract manufacturing organizations and internal production units alike, making the final API more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of robust, shelf-stable starting materials. Chlorohydrazones and sulfur ylides used in this process are generally easier to source and store compared to air-sensitive organometallic reagents. The tolerance of the reaction to air and moisture further de-risks the manufacturing process, reducing the likelihood of batch failures due to environmental excursions. This reliability allows for more accurate forecasting and inventory management, ensuring that critical drug development timelines are met without unexpected delays caused by reagent shortages or failed batches.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and safety, but this methodology is inherently scalable due to its exothermic neutrality and ambient temperature operation. The absence of toxic heavy metals simplifies waste stream management, lowering the costs associated with hazardous waste disposal and environmental compliance reporting. As regulatory pressures on pharmaceutical manufacturing continue to intensify, adopting greener, metal-free syntheses positions companies favorably for audits and inspections, facilitating faster regulatory approvals and market entry for new drug candidates.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is vital for stakeholders evaluating its integration into existing pipelines. The following questions address common inquiries regarding the scope, scalability, and practical implementation of the technology described in patent CN116253692A. These insights are derived directly from the experimental data and mechanistic studies provided in the patent documentation, offering a clear picture of what partners can expect when leveraging this chemistry for their specific projects.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method eliminates the need for expensive and toxic heavy metal catalysts, operates under mild conditions (room temperature, air atmosphere), and uses cheap, readily available starting materials like potassium carbonate.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is highly scalable. It avoids complex inert gas protections and uses simple workup procedures (filtration and chromatography), making it ideal for commercial scale-up from grams to tons.
Q: What is the structural diversity achievable with this method?
A: The method supports a wide range of substituents (R1, R2, R3) including alkyl, phenyl, naphthyl, and furyl groups with various functional groups like halogens, alkoxy, and nitro groups, allowing for broad library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of trifluoromethyl-substituted 1,2,4-triazine meets the highest industry standards for potency and impurity profiles. We are committed to delivering not just a chemical product, but a comprehensive supply solution that enhances your operational efficiency.
We invite you to collaborate with our technical team to evaluate the feasibility of this route for your specific target molecules. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of how implementing this technology can optimize your budget and timeline. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your project's unique requirements. Contact our technical procurement team today to discuss how we can support your next breakthrough in drug development with reliable, high-quality intermediates produced via this cutting-edge methodology.
