Advanced Enantioselective Synthesis of Chiral 3-Hydroxypyrrolones for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral heterocycles, which serve as critical scaffolds for bioactive molecules. Patent CN101735134A introduces a groundbreaking approach for the synthesis of chiral 3-hydroxypyrrolone compounds, specifically targeting the efficient production of optically pure 3-hydroxy-3-aryl-tetrahydropyrrole derivatives. This technology leverages an intramolecular enantioselective addition reaction of N-substituted enamides, catalyzed by chiral Lewis acids, to forge the pyrrolidone ring with exceptional stereocontrol. Unlike conventional methods that often struggle with racemization or require cumbersome resolution steps, this patented process delivers high enantiomeric excess (ee) values, frequently exceeding 95%, under relatively mild conditions. The versatility of this chemistry allows for the incorporation of diverse substituents at the nitrogen and carbon positions, making it a powerful tool for generating libraries of potential drug candidates. For R&D teams focused on pipeline acceleration, this method offers a direct route to high-purity intermediates that are otherwise difficult to access.
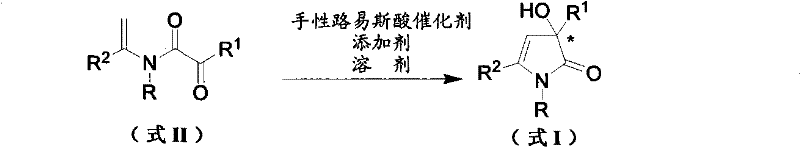
As a reliable pharmaceutical intermediate supplier, understanding the nuances of such catalytic systems is paramount for ensuring supply chain continuity. The core innovation lies in the transformation of readily available enamide precursors into complex chiral architectures in a single step. This not only reduces the number of synthetic operations but also minimizes waste generation, aligning with modern green chemistry principles. The structural diversity supported by this method, where R, R1, and R2 groups can vary from simple alkyls to complex substituted aryls, ensures that manufacturers can adapt the process for a wide array of therapeutic targets. The ability to tune the stereochemistry through the selection of specific catalyst enantiomers further enhances the strategic value of this technology for custom synthesis projects.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-hydroxy-3-aryl-tetrahydropyrroles has relied heavily on the reaction of pyrrolidin-3-ones with Grignard reagents, a methodology documented as early as the 1960s. While chemically straightforward, this classical approach suffers from a fundamental flaw: it inherently produces racemic mixtures. In the context of modern drug development, where regulatory agencies demand high optical purity to ensure safety and efficacy, obtaining a single enantiomer from a racemate necessitates additional resolution steps. These resolution processes, whether via crystallization or chiral chromatography, are notoriously inefficient, often resulting in a maximum theoretical yield of 50% for the desired isomer and generating substantial amounts of unwanted waste. Furthermore, Grignard reagents are highly sensitive to moisture and air, requiring stringent anhydrous conditions and specialized handling equipment, which complicates scale-up and increases operational costs. The harsh basicity of these reagents can also lead to compatibility issues with other functional groups present in complex molecular scaffolds, limiting the scope of substrates that can be effectively utilized.
The Novel Approach
In stark contrast, the methodology described in CN101735134A utilizes a chiral Lewis acid-catalyzed intramolecular cyclization of enamides to achieve direct asymmetric synthesis. This novel approach bypasses the formation of racemates entirely by inducing chirality during the bond-forming event itself. The reaction proceeds through the activation of the carbonyl group within the enamide substrate by a chiral metal complex, facilitating a highly selective nucleophilic attack by the alkene moiety. This mechanism allows for the construction of the quaternary stereocenter at the 3-position of the pyrrolidone ring with precision. The process operates under much milder conditions compared to Grignard chemistry, with reaction temperatures ranging from -78°C to 150°C depending on the specific substrate and catalyst system employed. Moreover, the catalysts used, such as salen-chromium or salen-copper complexes, are stable in air and inexpensive to procure, removing the need for glovebox operations and reducing raw material costs. The result is a streamlined process that delivers high yields, often above 90%, and exceptional enantioselectivity, fundamentally changing the economic and technical landscape for producing these valuable intermediates.
Mechanistic Insights into Salen-Catalyzed Enantioselective Cyclization
The heart of this technological advancement is the chiral Lewis acid catalyst, typically based on a salen ligand framework complexed with a transition metal such as Chromium (III) or Copper (II). The salen ligand provides a rigid, chiral environment around the metal center, which is crucial for differentiating between the prochiral faces of the substrate during the reaction. In the proposed catalytic cycle, the metal center coordinates with the carbonyl oxygen of the keto-amide moiety in the enamide substrate, increasing its electrophilicity. Simultaneously, the chiral pocket created by the ligand directs the approach of the nucleophilic enamide double bond, ensuring that the cyclization occurs with a specific stereochemical outcome. This precise spatial arrangement is what enables the high enantiomeric excess values observed, frequently reaching 99% ee in optimized examples. The robustness of the salen framework allows it to withstand various reaction conditions, and the metal center can be tuned by changing counterions (e.g., Cl, OTf) to modulate Lewis acidity and reaction rates.
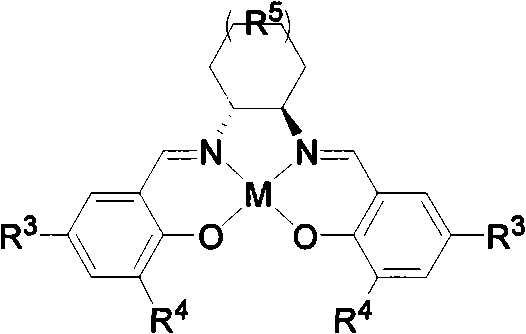
Furthermore, the patent highlights the importance of additives in controlling impurity profiles and maximizing yield. During the cyclization, there is a potential risk of carbocation rearrangement, which could lead to structural isomers and reduce the purity of the final product. To mitigate this, the process incorporates basic additives such as sodium carbonate, potassium carbonate, or organic bases like triethylamine. These additives function to neutralize any acidic byproducts or stabilize intermediate species, thereby suppressing undesired rearrangement pathways. This level of process control is critical for manufacturing high-purity pharmaceutical intermediates where impurity limits are strictly regulated. The ability to fine-tune the reaction environment through the combination of catalyst structure, solvent choice (ranging from non-polar benzene to polar dichloromethane), and additive loading demonstrates a sophisticated understanding of reaction engineering that translates directly to reproducible commercial scale-up.
How to Synthesize Chiral 3-Hydroxypyrrolones Efficiently
The practical implementation of this synthesis is designed for scalability and ease of operation, making it suitable for both laboratory discovery and industrial production. The general procedure involves charging a reaction vessel with the enamide substrate and the chiral catalyst under an inert atmosphere, followed by the addition of a dry solvent. The reaction is then allowed to proceed with stirring until the starting material is consumed, as monitored by TLC or HPLC. Workup typically involves quenching with a base like triethylamine, followed by standard purification techniques such as silica gel column chromatography. The versatility of the method is evidenced by its success with a wide variety of substrates, including those with electron-withdrawing and electron-donating groups on the aromatic rings. For detailed operational parameters and specific optimization strategies tailored to your target molecule, please refer to the standardized synthesis guide below.
- Prepare the reaction vessel under inert atmosphere and charge with the enamide substrate (Formula II) and the chiral salen-metal catalyst (e.g., Salen-Cr or Salen-Cu).
- Add the appropriate dry solvent (such as benzene, toluene, or dichloromethane) and optional additives like sodium carbonate to suppress side reactions.
- Stir the mixture at temperatures ranging from -78°C to 150°C until completion, then quench with triethylamine and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this catalytic technology offers transformative benefits in terms of cost structure and logistical reliability. The shift from stoichiometric reagents to catalytic processes inherently reduces the mass intensity of the synthesis, leading to significant cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for expensive chiral resolution steps and minimizing the loss of material associated with racemate separation, the overall process efficiency is drastically improved. The catalysts themselves are derived from abundant metals and ligands that are commercially available at competitive prices, and their air stability simplifies storage and handling requirements, reducing the need for specialized infrastructure. This accessibility ensures a stable supply of critical reagents, mitigating the risk of production delays caused by material shortages.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the high atom economy and the elimination of resolution losses. Traditional routes often discard half of the produced material during resolution, whereas this enantioselective pathway directs the synthesis towards the desired isomer from the outset. Additionally, the ability to lower catalyst loading to extremely low levels, as demonstrated in the patent data where loadings as low as 0.01 mol% were effective under certain conditions, further drives down the cost of goods sold. The use of common solvents and the avoidance of cryogenic conditions in many examples also contribute to lower energy consumption and utility costs, creating a leaner and more profitable manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the starting materials and the simplicity of the reaction setup. The enamide precursors are easily prepared in large quantities from readily available bulk chemicals, ensuring a consistent feedstock supply. The reaction conditions are forgiving, tolerating a range of temperatures and solvents, which provides flexibility in sourcing and production planning. This flexibility is crucial for maintaining continuity of supply in the face of market fluctuations or logistical disruptions. Furthermore, the high yields and purity achieved reduce the burden on downstream purification units, allowing for faster throughput and shorter lead times for high-purity pharmaceutical intermediates, enabling partners to meet tight project deadlines with confidence.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the homogeneous nature of the catalysis and the absence of hazardous reagents like pyrophoric Grignards. The process generates less waste compared to traditional methods, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The ability to operate at ambient or elevated temperatures without the need for extreme cooling reduces the energy footprint of the manufacturing facility. Moreover, the high selectivity of the reaction minimizes the formation of byproducts, simplifying waste treatment and disposal. This environmental compatibility not only reduces compliance costs but also enhances the brand reputation of the manufacturer as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this technology, we have compiled the following insights based on the patent specifications and experimental data. These questions cover critical aspects of the synthesis, including catalyst performance, substrate scope, and process optimization. Understanding these details is essential for evaluating the feasibility of integrating this method into your existing production workflows. The answers provided reflect the proven capabilities of the technology as demonstrated in the referenced intellectual property.
Q: What is the primary advantage of this salen-catalyzed method over traditional Grignard routes?
A: Unlike traditional Grignard additions which produce racemic mixtures requiring difficult resolution, this method utilizes chiral Lewis acid catalysis to achieve direct enantioselective cyclization with ee values up to 99%, significantly streamlining the production of optically pure intermediates.
Q: Can the catalyst loading be reduced for large-scale manufacturing?
A: Yes, the patent data indicates that catalyst loading can be drastically reduced to as low as 0.01 mol% while maintaining high yields and enantioselectivity, particularly when combined with additives and optimized temperature profiles, making it highly economically viable for ton-scale production.
Q: What types of substituents are tolerated on the enamide substrate?
A: The process exhibits excellent substrate scope, tolerating a wide range of groups including C1-C6 alkyls, allyl, benzyl, and various substituted phenyl rings (with fluoro, chloro, bromo, methoxy, or trifluoromethyl groups), allowing for the synthesis of diverse drug analogues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3-Hydroxypyrrolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced chiral synthesis technologies in accelerating drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, utilizing rigorous QC labs to verify every batch against the highest industry standards. Our capability to implement complex catalytic systems, such as the salen-mediated cyclization described here, positions us as a preferred partner for companies seeking to optimize their supply chains for chiral building blocks.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalytic route for your target molecules. We encourage you to reach out today to obtain specific COA data for our catalog items or to discuss route feasibility assessments for your proprietary compounds. Let us help you secure a reliable, cost-effective, and scalable supply of critical chiral intermediates to support your next breakthrough therapy.
