Optimized Synthesis of Baloxavir Intermediate for Commercial Scale-Up
The pharmaceutical landscape for antiviral therapeutics has been significantly reshaped by the introduction of cap-dependent endonuclease inhibitors, most notably Baloxavir marboxil. As detailed in patent CN113527119A, the efficient production of key precursors is paramount to meeting global demand for influenza treatments. This technical insight focuses on a novel preparation method for 2-(2-aminoethoxy)-1,1-dimethoxyethane, a critical building block in the synthesis of this next-generation antiviral agent. The disclosed methodology addresses longstanding challenges in intermediate isolation, offering a pathway that combines operational simplicity with the rigorous purity standards required for GMP manufacturing. By optimizing the nucleophilic substitution parameters, this process eliminates the formation of stubborn byproducts that have historically plagued the supply chain.
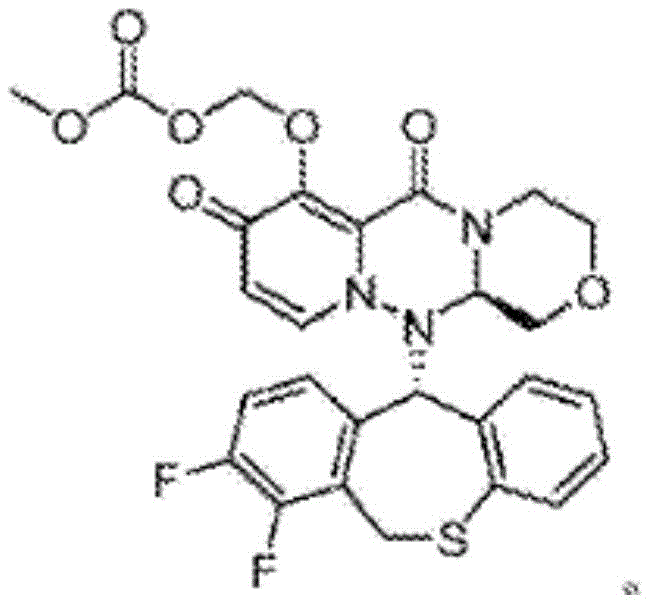
The strategic value of this synthesis lies in its ability to bypass the purification bottlenecks associated with earlier iterations of the route. In the context of large-scale pharmaceutical production, the ability to secure a reliable pharmaceutical intermediates supplier who can deliver consistent quality is non-negotiable. The technology described herein leverages common commodity chemicals—ethanolamine and chloroacetaldehyde derivatives—yet arranges them in a specific stoichiometric and thermal environment that maximizes yield while minimizing downstream processing burdens. This represents a significant step forward in cost reduction in pharmaceutical intermediates manufacturing, as it reduces the reliance on expensive chromatographic separations often necessitated by lower-purity crude outputs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic routes for generating the essential amino-ether linkage in Baloxavir precursors suffered from severe physicochemical limitations that impacted overall process efficiency. Specifically, the intermediate compound exhibited high water solubility, which created a paradoxical situation where standard aqueous workups failed to effectively partition the product from the reaction matrix. When tetrahydrofuran was employed as the primary solvent in these legacy systems, the resulting solution made it exceptionally difficult to precipitate or extract the pure product, leading to significant material loss. Furthermore, the inability to isolate Compound C with high fidelity meant that subsequent reactions were forced to proceed with contaminated feedstock. This carryover of impurities resulted in low conversion rates during the critical cyclization steps, often manifesting as the formation of intractable "black mud" states that resisted crystallization and complicated final purification.
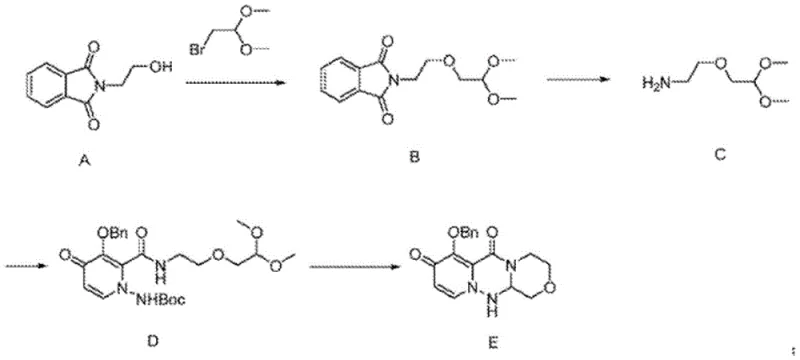
The Novel Approach
The methodology presented in CN113527119A fundamentally alters the reaction landscape by introducing a robust base-mediated protocol that facilitates clean phase separation. By utilizing sodium tert-butoxide in a controlled molar excess relative to the amine and electrophile, the reaction drives the equilibrium toward the desired ether product while suppressing side reactions. The switch to a dichloromethane-based extraction system during the workup phase is a critical innovation; unlike the problematic tetrahydrofuran systems of the past, this solvent pair allows for sharp interface formation between the organic and aqueous layers. This physical separation capability ensures that the hydrophilic impurities remain in the aqueous wash, while the lipophilic product is retained in the organic phase. Consequently, the final rectification step yields a product of exceptional purity, effectively breaking the cycle of impurity propagation that characterized previous manufacturing attempts.
Mechanistic Insights into Base-Mediated Nucleophilic Substitution
The core chemical transformation relies on a classic yet finely tuned nucleophilic substitution mechanism where the primary amine of ethanolamine attacks the electrophilic carbon of the chloroacetaldehyde dimethyl acetal. The presence of sodium tert-butoxide serves a dual purpose: it acts as a proton scavenger to neutralize the hydrochloric acid generated during the displacement, and it likely assists in deprotonating the ammonium species to regenerate the free amine nucleophile, thereby sustaining the reaction kinetics. Maintaining the temperature window between 80-90°C is essential; this thermal energy provides the activation barrier crossing required for the substitution without inducing thermal degradation of the sensitive acetal functionality. The stoichiometry, defined by a molar ratio of approximately 1:3.5-4.5:5-7 (Acetal:Base:Amine), ensures that the amine is present in sufficient excess to act as both reactant and solvent medium, while the base is available in quantities that guarantee complete acid neutralization throughout the reaction duration.
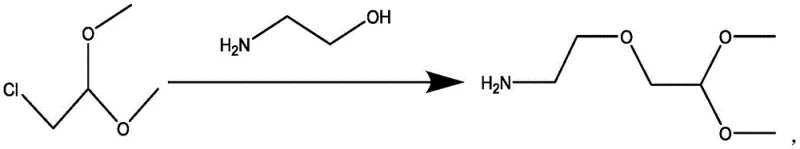
From an impurity control perspective, the mechanism is designed to minimize the formation of bis-alkylated byproducts or polymerization of the aldehyde equivalent. The use of the dimethyl acetal protects the aldehyde group from self-condensation or oxidation during the harsh basic conditions. Furthermore, the specific choice of base prevents the elimination reactions that could lead to vinyl ether byproducts. By strictly controlling the addition rate of the chloroacetal to the pre-heated amine/base mixture, the local concentration of the electrophile is kept low, favoring the intermolecular attack by the amine over any potential intermolecular side reactions. This kinetic control, combined with the thermodynamic stability of the acetal group, results in a clean reaction profile that simplifies the downstream purification burden significantly.
How to Synthesize 2-(2-aminoethoxy)-1,1-dimethoxyethane Efficiently
The execution of this synthesis requires precise adherence to the thermal and stoichiometric parameters outlined in the patent to ensure reproducibility at scale. The process begins with the activation of the nucleophile, followed by the controlled introduction of the electrophile, and concludes with a phase-separation driven workup. Operators must ensure that the heating mantle or jacket maintains the 80-90°C range consistently, as fluctuations can impact the reaction rate and selectivity. The following guide summarizes the standardized operational procedure derived from the experimental examples provided in the intellectual property documentation.
- Prepare a mixed solution by combining an alkaline reagent, specifically sodium tert-butoxide, with ethanolamine and heating the mixture to initiate activation.
- Introduce 2-chloroacetaldehyde dimethyl acetal into the heated mixture under stirring, maintaining a reaction temperature between 80-90°C to drive the substitution.
- Quench the reaction with ice water and dichloromethane, separate the organic phase, wash with sodium sulfate solution, and purify via rectification to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the unit operations required to achieve pharmaceutical-grade purity. By eliminating the need for complex chromatographic columns or multiple recrystallization cycles that were previously necessary to remove "black mud" impurities, the overall processing time is significantly compressed. This reduction in processing steps translates directly into lower utility consumption and reduced labor costs per kilogram of output. Additionally, the reagents employed—ethanolamine, sodium tert-butoxide, and chloroacetaldehyde dimethyl acetal—are widely available commodity chemicals, mitigating the risk of raw material shortages that often plague supply chains dependent on exotic or custom-synthesized starting materials.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the high atom economy of the substitution reaction and the avoidance of expensive purification media. Since the workup relies on liquid-liquid extraction rather than solid-phase adsorption, the consumption of silica gel or specialized resins is virtually eliminated. This shift not only lowers the direct material cost but also reduces the volume of hazardous solid waste generated, leading to substantial savings in waste disposal fees. Furthermore, the ability to recover and recycle the dichloromethane solvent through standard distillation trains adds another layer of cost efficiency, ensuring that the operating expenditure remains competitive even at fluctuating market prices for solvents.
- Enhanced Supply Chain Reliability: Robustness is a key metric for supply chain continuity, and this method excels by tolerating minor variations in reagent quality without compromising the final product specification. The use of a biphasic workup system provides a natural "filtering" effect, where water-soluble contaminants are washed away, ensuring that the crude material entering the final distillation is already of high quality. This reliability reduces the risk of batch failures and the associated delays in production schedules. For a reliable pharmaceutical intermediates supplier, this consistency means that delivery timelines can be met with greater confidence, shielding downstream API manufacturers from the volatility of production stoppages caused by off-spec intermediate batches.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale is facilitated by the mild reaction conditions and the absence of highly exothermic events that require specialized cooling infrastructure. The reaction temperature of 80-90°C is easily achievable with standard steam heating, making it compatible with existing glass-lined or stainless steel reactors in most multipurpose plants. From an environmental standpoint, the process generates less saline wastewater compared to acid-base neutralization heavy routes, and the containment of volatile organics within a closed distillation system aligns with stringent modern environmental regulations. This ease of commercial scale-up of complex pharmaceutical intermediates ensures that production capacity can be ramped up rapidly to meet surges in demand for antiviral medications.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and quality control of this specific intermediate synthesis. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the operational boundaries and quality expectations for this chemical entity.
Q: Why is the purity of 2-(2-aminoethoxy)-1,1-dimethoxyethane critical for Baloxavir synthesis?
A: Impurities in this intermediate, particularly due to its high water solubility making isolation difficult in prior art, lead to low conversion in subsequent cyclization steps and the formation of intractable black mud byproducts.
Q: What are the key reaction conditions for this novel preparation method?
A: The process utilizes sodium tert-butoxide as a base at a molar ratio of roughly 3.5-4.5 equivalents relative to the acetal, with a reaction temperature maintained strictly between 80-90°C to ensure optimal yield without degradation.
Q: How does this method improve upon previous synthetic routes?
A: Unlike conventional methods that struggle with tetrahydrofuran solubility issues, this approach employs a dichloromethane extraction system that allows for efficient phase separation and high-purity isolation via rectification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-aminoethoxy)-1,1-dimethoxyethane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of the final API is inextricably linked to the quality of its precursors. Our technical team has thoroughly analyzed the route described in CN113527119A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications to ensure that every batch of 2-(2-aminoethoxy)-1,1-dimethoxyethane meets the exacting standards required for antiviral drug synthesis. Our commitment to process optimization ensures that we can deliver this critical intermediate with the consistency and reliability that global pharmaceutical partners demand.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this superior manufacturing route. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments tailored to your production volumes, ensuring a seamless integration of this high-quality intermediate into your manufacturing pipeline.
