Revolutionizing Cephalosporin Intermediate Production via Continuous Microreactor Technology
Introduction to Advanced Continuous Manufacturing for Beta-Lactam Intermediates
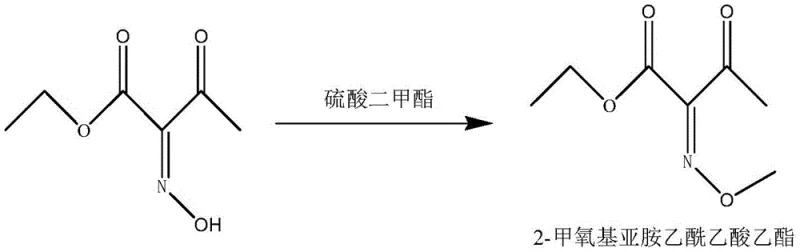
The pharmaceutical industry is currently witnessing a paradigm shift towards continuous manufacturing, particularly for critical intermediates used in the synthesis of life-saving antibiotics. Patent CN111072515B discloses a groundbreaking method for the continuous synthesis of an aminothiaxamic acid intermediate, specifically 2-methoxyimine ethyl acetoacetate, which serves as a pivotal building block for third-generation cephalosporins. This technology addresses long-standing inefficiencies in traditional batch processing by leveraging microreactor engineering to enhance mass transfer and thermal control. For R&D directors and process chemists, this represents a significant opportunity to optimize impurity profiles and increase overall process robustness. The transition from intermittent batch operations to a seamless continuous flow regime not only improves product quality consistency but also aligns with modern green chemistry principles by minimizing solvent usage and waste generation. As a leading entity in fine chemical synthesis, understanding the nuances of this patent is essential for maintaining competitiveness in the global supply chain of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2-methoxyimine ethyl acetoacetate has relied heavily on batch synthesis methods that suffer from inherent thermodynamic and kinetic limitations. In traditional setups, reagents such as dimethyl sulfate are added dropwise to a mixture containing the substrate and base, often requiring methanol or phase transfer catalysts to facilitate mass transfer between immiscible phases. This approach frequently results in localized zones of high alkalinity at the beginning of the reaction, which can lead to the degradation of the sensitive 2-hydroxyimine-ethyl acetoacetate substrate and the methylating agent. Furthermore, the use of auxiliary agents like phase transfer catalysts complicates downstream processing, making solvent recovery difficult and significantly increasing the Chemical Oxygen Demand (COD) of the resulting wastewater. The intermittent nature of batch reactors also introduces variability in product quality, as mixing efficiency and temperature control fluctuate throughout the reaction cycle, posing challenges for maintaining strict purity specifications required for antibiotic synthesis.
The Novel Approach
The innovative methodology outlined in the patent overcomes these deficiencies by implementing a two-stage continuous flow system comprising a microreactor followed by a pipeline reactor. By pumping the 2-hydroxyimine-ethyl acetoacetate solution, dimethyl sulfate, and sodium carbonate solution simultaneously into a micropore array microchannel reactor, the process ensures instantaneous and homogeneous mixing at the molecular level. This precise control eliminates the dangerous local high-alkalinity environments that plague batch processes, thereby preserving the integrity of the reactants and maximizing yield. Crucially, the enhanced mass transfer capabilities of the microreactor render the addition of methanol or phase transfer catalysts entirely unnecessary, simplifying the reaction matrix and facilitating easier solvent recovery. The effluent then flows into a pipeline reactor to complete the conversion, ensuring a consistent residence time that guarantees high production quality stability and operational safety while drastically reducing VOC emissions through a sealed, single-outlet system design.
Mechanistic Insights into Microreactor-Assisted O-Methylation
The core chemical transformation involves the nucleophilic attack of the oxime oxygen on the methyl group of dimethyl sulfate under basic conditions, a reaction that is highly sensitive to mixing efficiency and pH gradients. In the microreactor environment, the high surface-to-volume ratio facilitates rapid heat dissipation, preventing thermal runaways that could otherwise promote side reactions or decomposition of the thermally unstable diazo-like intermediates. The simultaneous introduction of the aqueous base and organic substrate into the microchannels creates a highly dispersed emulsion where the interfacial area for reaction is maximized without the need for surfactants. This intense mixing regime ensures that the concentration of hydroxide ions remains uniform throughout the reaction zone, preventing the localized excess base that typically hydrolyzes the ester moiety or degrades the oxime functionality in batch vessels. Consequently, the reaction proceeds with high selectivity towards the desired O-methylated product, minimizing the formation of by-products that are difficult to separate during purification.
From an impurity control perspective, the continuous flow architecture provides a deterministic environment where reaction parameters such as temperature and residence time are strictly regulated. Unlike batch reactors where concentration gradients evolve over time, the plug-flow characteristics of the pipeline reactor ensure that every molecule of substrate experiences identical reaction conditions. This uniformity is critical for suppressing the formation of dimerization products or over-methylated species that can arise from prolonged exposure to reactive conditions. Furthermore, the absence of phase transfer catalysts removes a significant source of potential metallic or organic contamination, resulting in a cleaner crude product profile. This mechanistic advantage translates directly to reduced burden on downstream purification units, allowing for more efficient distillation or crystallization steps to achieve the stringent purity levels demanded by regulatory bodies for cephalosporin intermediates.
How to Synthesize 2-Methoxyimine Ethyl Acetoacetate Efficiently
The synthesis of this critical cephalosporin intermediate via continuous flow technology requires precise calibration of flow rates and thermal zones to replicate the high-yield conditions demonstrated in the patent literature. Operators must prepare three distinct feed streams: a chloroform solution of the hydroxyimine substrate, neat dimethyl sulfate, and an aqueous sodium carbonate solution, ensuring that the molar ratios are tightly controlled to prevent reagent accumulation. The detailed standardized synthesis steps involve pumping these streams into the microreactor assembly, managing the exothermic nature of the methylation, and executing the subsequent workup procedures to isolate the high-purity product. For a comprehensive breakdown of the specific equipment setup, flow rate calculations, and safety protocols required to implement this route, please refer to the technical guide below.
- Prepare feed solutions of 2-hydroxyimine-ethyl acetoacetate in chloroform, dimethyl sulfate, and aqueous sodium carbonate with precise molar ratios.
- Pump all streams simultaneously into a micropore array microchannel reactor maintained at 40-70°C for rapid initial mixing and reaction.
- Transfer the effluent into a pipeline reactor for completion (15-45 min residence), followed by chloroform extraction and distillation to isolate the crude product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this continuous microreactor technology offers profound strategic advantages beyond mere technical superiority. The elimination of expensive and environmentally burdensome phase transfer catalysts and co-solvents like methanol directly translates to a simplified bill of materials and reduced raw material costs. By removing these auxiliaries, the process significantly lowers the complexity of wastewater treatment, avoiding the high costs associated with reducing COD levels and disposing of hazardous chemical waste. This streamlining of the production workflow enhances the overall economic viability of manufacturing this key pharmaceutical intermediate, allowing suppliers to offer more competitive pricing structures without compromising on quality standards. Additionally, the continuous nature of the process reduces the dependency on large batches of varying quality, ensuring a steady and predictable output that stabilizes inventory management.
- Cost Reduction in Manufacturing: The removal of phase transfer catalysts and methanol from the reaction recipe eliminates the need for complex recovery systems and reduces the consumption of auxiliary chemicals. This simplification of the chemical process leads to substantial cost savings in raw material procurement and waste disposal fees. Furthermore, the higher yield and purity achieved through continuous flow reduce the loss of valuable starting materials and minimize the energy consumption required for downstream purification steps. These cumulative efficiencies result in a leaner manufacturing operation that is better positioned to absorb market fluctuations in raw material prices while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Continuous manufacturing systems are inherently more scalable and responsive to demand changes compared to rigid batch processes. The ability to run the reactor for extended periods without the downtime associated with charging, heating, cooling, and discharging batch vessels ensures a consistent supply of the intermediate. This reliability is crucial for downstream API manufacturers who require just-in-time delivery to maintain their own production schedules. Moreover, the improved safety profile of the microreactor system, which contains hazardous reagents like dimethyl sulfate within small channels, reduces the risk of unplanned shutdowns due to safety incidents, thereby guaranteeing uninterrupted supply continuity for global partners.
- Scalability and Environmental Compliance: Scaling continuous flow processes is often more straightforward than scaling batch reactions, as it primarily involves numbering up microreactor units rather than dealing with the non-linear heat and mass transfer issues of larger vessels. This modularity allows for rapid capacity expansion to meet surging market demand for cephalosporin antibiotics. From an environmental standpoint, the closed system design drastically reduces VOC emissions by limiting the number of open transfer points and exhaust ports. The reduction in wastewater COD and the elimination of difficult-to-remove catalysts make the process compliant with increasingly stringent environmental regulations, future-proofing the supply chain against regulatory risks and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous synthesis of aminothiazoyl oxime acid intermediates. These insights are derived directly from the experimental data and process descriptions found in the relevant patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. The answers highlight the specific operational benefits and quality improvements that distinguish this method from legacy batch processes.
Q: Why is continuous flow preferred over batch processing for this methylation reaction?
A: Continuous flow eliminates the localized high alkalinity found in batch reactors which degrades sensitive substrates. It also removes the need for phase transfer catalysts, significantly simplifying wastewater treatment and solvent recovery.
Q: What are the purity specifications achievable with this microreactor method?
A: The patented continuous process consistently achieves quantitative content of approximately 93% with yields reaching up to 95%, surpassing traditional batch methods which often struggle with impurity profiles due to poor mass transfer.
Q: How does this process impact environmental compliance and VOC emissions?
A: By utilizing a closed continuous system with a single gas outlet, volatile organic compound (VOC) emissions are drastically reduced compared to multiple batch reactors. Furthermore, eliminating methanol and phase transfer catalysts lowers the COD of wastewater.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methoxyimine Ethyl Acetoacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain, particularly for essential antibiotics like cephalosporins. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of multinational corporations with ease. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify identity and assay. By leveraging advanced continuous manufacturing technologies similar to those described in recent patents, we can offer superior process control and consistency that traditional manufacturers cannot match.
We invite you to collaborate with us to optimize your sourcing strategy for this vital intermediate. Our experts are ready to provide a Customized Cost-Saving Analysis that demonstrates how switching to our supply can reduce your total landed cost. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project needs. Let us be your partner in driving efficiency and quality in your antibiotic production lines.
