Scalable Preparation of High-Purity Imaric Acid Hydrochloride for Oncology Drug Manufacturing
Scalable Preparation of High-Purity Imaric Acid Hydrochloride for Oncology Drug Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for critical oncology intermediates, and the preparation of Imaric Acid stands as a pivotal challenge in the supply chain for Imatinib Mesylate. Patent CN112321535B introduces a transformative methodology that addresses long-standing inefficiencies in producing 4-(4-methylpiperazine-1-methyl)benzoic acid and its hydrochloride salt. This innovation shifts the paradigm from problematic cyano or direct acid condensation routes to a highly efficient ketone-based strategy. By utilizing 4-halogenated methyl acetophenone as the starting scaffold, the process effectively circumvents the notorious salt-formation issues that have historically plagued yield and purity. For R&D directors and procurement specialists, this represents a significant leap forward in process reliability, offering a pathway that ensures high conversion rates while minimizing the generation of polymeric impurities. The structural integrity of the final product is paramount for downstream drug synthesis, and this patent outlines a protocol that delivers exceptional quality standards suitable for immediate use in GMP environments.
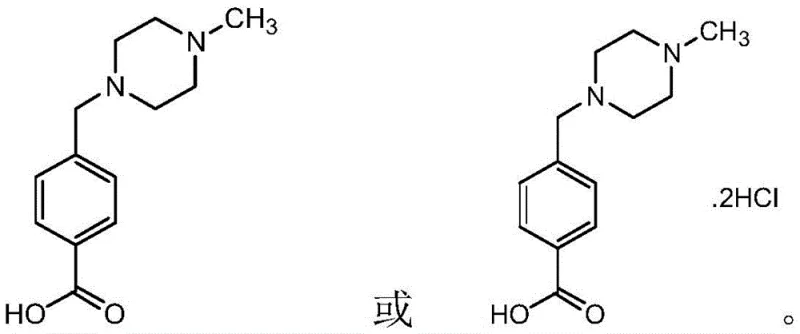
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Imaric Acid has been hindered by fundamental chemical incompatibilities in the starting materials used in prior art methods. One prevalent conventional route involves the condensation of N-methylpiperazine with p-cyanobenzyl bromide, followed by hydrolysis of the nitrile group. However, this approach is fraught with difficulties; during the initial condensation phase, the cyano group tends to interact with the amine, forming stable salts that render the reaction incomplete. Furthermore, the subsequent hydrolysis step often fails to proceed to completion, typically stalling at around 85 percent conversion, which necessitates complex and costly refining procedures to remove unreacted intermediates. Another common method attempts direct condensation using p-chloromethylbenzoic acid. This route is equally problematic because the carboxyl group readily forms salts with N-methylpiperazine, forcing manufacturers to use a vast excess of the amine reagent to drive the reaction. This excess not only inflates raw material costs but also promotes polymerization side reactions, generating a complex mixture of impurities that are difficult to separate. These legacy processes result in low overall yields and inconsistent product quality, creating bottlenecks for reliable commercial scale-up of complex pharmaceutical intermediates.
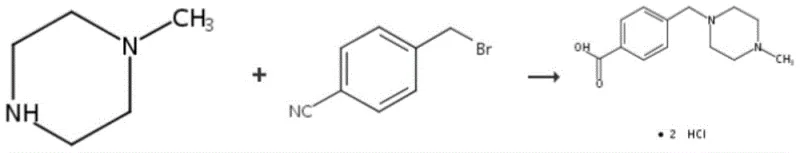
The Novel Approach
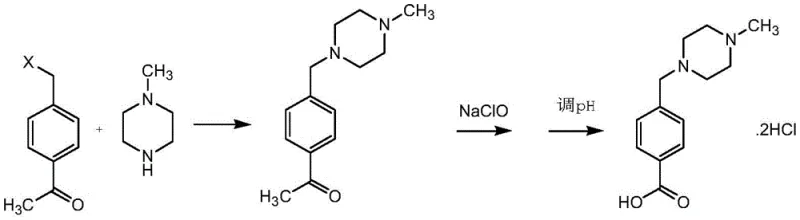
In stark contrast to these flawed methodologies, the novel approach detailed in the patent utilizes 4-halogenated methyl acetophenone as the foundational building block. This strategic selection of a ketone-containing precursor fundamentally alters the reaction landscape by eliminating the acidic or highly polar functional groups that cause salt formation with the piperazine ring. The process begins with a clean nucleophilic substitution where the halogenated methyl group reacts with N-methylpiperazine in the presence of a mild acid-binding agent. Because the para-position holds a ketone rather than a carboxylic acid or nitrile, the amine remains free to react efficiently without being sequestered into inactive salt complexes. This allows the reaction to proceed with theoretical molar equivalents, drastically reducing reagent waste. Following the formation of the intermediate ketone, a subsequent oxidation step using hypochlorite converts the acetyl group into the desired carboxylic acid functionality under mild conditions. This two-stage sequence ensures that the final product is obtained with high purity and minimal byproduct formation, effectively solving the yield and purification challenges associated with previous generations of synthesis technology.
Mechanistic Insights into Ketone-Based Substitution and Oxidation
The core of this technological advancement lies in the precise control of reaction mechanisms to prevent side reactions. In the first stage, the nucleophilic attack of the secondary amine nitrogen in N-methylpiperazine on the benzylic carbon of the 4-halogenated methyl acetophenone is facilitated by the absence of competing electrostatic interactions. The use of an acid-binding agent, such as sodium hydroxide or potassium carbonate, neutralizes the hydrogen halide byproduct immediately, driving the equilibrium forward without degrading the sensitive amine. This results in an intermediate where the piperazine ring is cleanly attached to the benzene scaffold, with the ketone group remaining intact and inert to the substitution conditions. The second stage involves a haloform-type oxidation where hypochlorite species attack the methyl ketone. This transformation is highly selective, cleaving the carbon-carbon bond adjacent to the carbonyl to generate the carboxylate anion. The mild nature of hypochlorite oxidation prevents over-oxidation or degradation of the piperazine ring, which is a common risk with stronger oxidizing agents. By maintaining the reaction temperature between 40 to 60°C during oxidation, the process ensures complete conversion of the intermediate while keeping energy inputs low. This mechanistic elegance translates directly into a cleaner impurity profile, as the potential for polymerization or thermal degradation is significantly suppressed compared to high-temperature acid hydrolysis methods.
Impurity control is further enhanced by the ability to monitor and limit residual starting materials to extremely low levels. The patent data indicates that raw material residues can be effectively controlled within less than 0.2 percent, a metric that is critical for regulatory compliance in API manufacturing. The avoidance of salt formation byproducts means that the reaction mixture does not require aggressive washing or extraction steps to remove polymeric sludge, which often traps the desired product. Instead, the final acidification step simply precipitates the pure hydrochloride salt from the solution. This streamlined purification capability is a direct result of the chemical stability of the ketone intermediate and the specificity of the hypochlorite oxidation. For quality assurance teams, this means that the final product consistently meets stringent specifications, with any single impurity detected being less than 0.1 percent. Such high purity eliminates the need for additional recrystallization or chromatographic purification, thereby reducing processing time and solvent consumption. The robustness of this mechanism ensures that even on a large industrial scale, the reaction kinetics remain predictable and manageable, providing a reliable foundation for consistent batch-to-batch quality.
How to Synthesize Imaric Acid Efficiently
The synthesis of Imaric Acid via this patented route offers a straightforward operational protocol that balances efficiency with safety. The process is designed to be adaptable to various solvent systems, including alcohols like methanol and ethanol, as well as aprotic solvents like acetonitrile or THF, allowing manufacturers to optimize based on existing infrastructure. The initial substitution reaction is conducted at ambient temperatures ranging from 20 to 30°C, which minimizes thermal hazards and energy costs. Following the formation of the intermediate, the oxidation step can be performed in the same vessel (one-pot synthesis) by simply adjusting the temperature and adding the oxidant, which simplifies the workflow and reduces material transfer losses. The detailed standardized synthesis steps below outline the precise molar ratios, temperature controls, and workup procedures required to achieve the high yields and purity described in the patent documentation.
- React 4-halogenated methyl acetophenone with N-methylpiperazine in an alcoholic solvent with an acid-binding agent at 20-30°C.
- Oxidize the resulting intermediate using sodium hypochlorite at 40-60°C to convert the acetyl group to a carboxylic acid.
- Adjust the pH to 1-2 using concentrated hydrochloric acid to crystallize the final Imaric Acid hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the raw material inventory and the reduction of waste disposal costs. By eliminating the need for excessive amounts of N-methylpiperazine and avoiding the use of harsh hydrolysis reagents like concentrated hydrochloric acid at high temperatures, the process significantly lowers the consumption of hazardous chemicals. This reduction in hazardous material usage not only decreases procurement costs but also simplifies environmental compliance and waste treatment protocols. Furthermore, the ability to achieve high conversion rates with near-stoichiometric amounts of reagents means that raw material utilization is maximized, leading to substantial cost savings in the bill of materials. The streamlined purification process also reduces the demand for organic solvents and filtration media, contributing to a leaner and more cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of complex refining steps and the reduction in reagent excess directly translate to lower production costs. Since the reaction avoids the formation of difficult-to-remove polymeric impurities, the need for extensive chromatographic purification or multiple recrystallizations is removed. This simplification of the downstream processing significantly reduces labor hours, utility consumption, and solvent recovery costs. Additionally, the use of inexpensive and readily available oxidants like sodium hypochlorite replaces more costly or specialized reagents required in alternative pathways. The overall effect is a manufacturing process that is inherently more economical, allowing for competitive pricing strategies in the global market for Imatinib intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 4-halogenated methyl acetophenone and sodium hypochlorite ensures a stable and resilient supply chain. Unlike specialized reagents that may be subject to geopolitical shortages or long lead times, the raw materials for this process are widely produced and easily sourced from multiple vendors. The mild reaction conditions also reduce the risk of production delays caused by equipment failures or safety incidents, as the process does not require high-pressure or extreme temperature reactors. This operational stability ensures consistent delivery schedules, which is critical for pharmaceutical customers who rely on just-in-time inventory models. By mitigating the risks associated with complex chemistry and scarce reagents, manufacturers can offer greater assurance of supply continuity to their downstream partners.
- Scalability and Environmental Compliance: The process is uniquely suited for large-scale industrial production due to its mild operating parameters and simplified workup. The ability to run the reaction at near-ambient temperatures reduces the load on heating and cooling systems, making it easier to scale from pilot plants to multi-ton commercial reactors without significant engineering modifications. Moreover, the reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations. The avoidance of heavy metal catalysts and the minimization of organic solvent waste contribute to a greener manufacturing footprint. This environmental compatibility not only reduces disposal fees but also enhances the corporate sustainability profile of the manufacturer, meeting the growing demand for eco-friendly pharmaceutical production practices among international clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines or for sourcing high-quality intermediates from external suppliers. The clarity provided here aims to bridge the gap between laboratory-scale innovation and commercial-scale application.
Q: Why is the ketone-based route superior to the cyano-based route for Imaric Acid?
A: The traditional cyano route suffers from salt formation between the cyano group and N-methylpiperazine, leading to incomplete reactions and difficult hydrolysis. The new ketone-based route avoids this salt formation entirely, ensuring higher conversion rates and easier purification.
Q: What are the critical quality parameters for Imaric Acid in Imatinib synthesis?
A: Critical parameters include residual starting material content (controlled to less than 0.2%) and single impurity levels (less than 0.1%). The patented method achieves these specifications without complex refining steps.
Q: How does this process impact production costs?
A: By eliminating the need for excessive amounts of N-methylpiperazine and avoiding complex hydrolysis conditions, the process significantly reduces raw material waste and energy consumption, leading to substantial cost savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imaric Acid Supplier
As the global demand for oncology therapeutics continues to rise, the need for high-quality, reliably sourced intermediates like Imaric Acid has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies to deliver products that meet the rigorous standards of the pharmaceutical industry. Our expertise extends beyond simple manufacturing; we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply regardless of market fluctuations. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest international benchmarks. By adopting innovative processes such as the ketone-based route described in recent patents, we ensure that our Imaric Acid is free from the impurities that often plague older synthesis methods, providing a superior foundation for your final drug product.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize their supply chains and reduce overall production costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Whether you are looking to secure a long-term supply agreement or explore the technical viability of this advanced synthesis route for your own facilities, NINGBO INNO PHARMCHEM is your trusted partner for delivering excellence in fine chemical manufacturing.
