Advanced Synthesis of Dronedarone Hydrochloride for Commercial Scale-up and High Purity API Production
Advanced Synthesis of Dronedarone Hydrochloride for Commercial Scale-up and High Purity API Production
The pharmaceutical landscape for antiarrhythmic agents has evolved significantly with the introduction of Dronedarone Hydrochloride, a milestone innovation in treating atrial fibrillation and atrial flutter. As detailed in patent CN102321058B, a novel synthetic methodology has been established that addresses critical limitations in previous manufacturing protocols, offering a pathway to higher purity and improved process safety. This technical insight report analyzes the strategic advantages of this specific chemical route, which utilizes a Friedel-Crafts acylation strategy starting from 2-butyl-5-nitrobenzofuran. By bypassing traditional demethylation hurdles, this approach provides a robust framework for reliable pharmaceutical intermediates supplier networks seeking to optimize their supply chains. The structural integrity and pharmacological efficacy of the final compound depend heavily on the precision of these synthetic steps, making the choice of manufacturing partner crucial for global health outcomes.
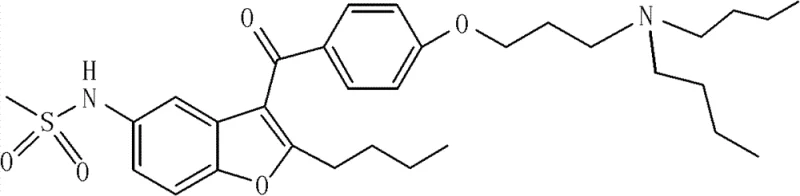
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Dronedarone have often been plagued by significant operational inefficiencies and environmental concerns that hinder cost reduction in API manufacturing. Prior art routes, such as those disclosed in earlier patents, frequently rely on the demethylation of methoxy intermediates using excessive amounts of anhydrous aluminum trichloride or harsh pyridine hydrochloride conditions at elevated temperatures. These aggressive reagents not only generate substantial hazardous waste but also complicate the purification process, leading to lower overall yields and potential impurity profiles that are difficult to manage. Furthermore, the use of acyl chlorides containing semicarbazide hydrochloride in some legacy methods results in poor product solubility and increased side reactions, making crystallization and final polishing extremely challenging for production teams. These factors collectively increase the cost of goods sold and introduce supply chain vulnerabilities due to the handling of dangerous chemicals.
The Novel Approach
In contrast, the methodology outlined in CN102321058B introduces a paradigm shift by utilizing p-chloropropoxybenzoyl chloride directly in the initial acylation step, effectively eliminating the need for subsequent demethylation. This strategic modification allows the reaction to proceed under much milder Lewis acid catalysis, significantly reducing the environmental footprint and operational complexity associated with traditional routes. The new process facilitates easier product isolation and purification, as the intermediates formed are more amenable to standard workup procedures without the formation of intractable complexes. By streamlining the synthetic sequence, this approach enhances the commercial scale-up of complex pharmaceutical intermediates, ensuring that production can be ramped up safely without compromising on quality or regulatory compliance. This represents a tangible advancement in green chemistry principles applied to cardiovascular drug synthesis.
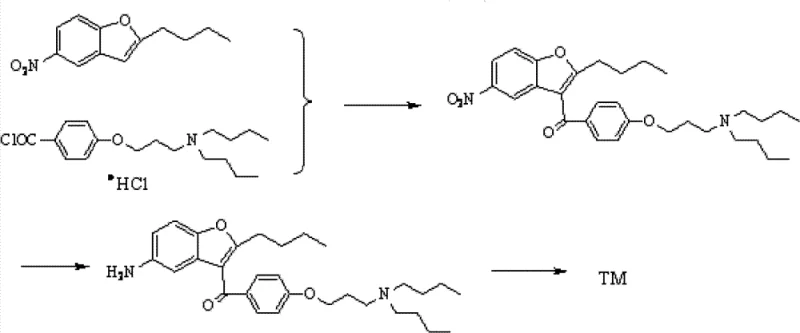
Mechanistic Insights into FeCl3-Catalyzed Friedel-Crafts Acylation
The core of this innovative synthesis lies in the precise execution of the Friedel-Crafts acylation reaction, where 2-butyl-5-nitrobenzofuran reacts with p-chloropropoxybenzoyl chloride in the presence of a Lewis acid catalyst such as FeCl3, AlCl3, or ZnCl2. The mechanism involves the generation of a highly reactive acylium ion intermediate, which selectively attacks the electron-rich position on the benzofuran ring to form the key ketone intermediate with high regioselectivity. Controlling the stoichiometry of the Lewis acid, typically maintained between 1.5 to 2.5 molar equivalents relative to the substrate, is critical to minimizing polyacylation side products while ensuring complete conversion of the starting material. The reaction is conducted in a halohydrocarbon solvent like dichloromethane at controlled temperatures, often below 25°C during addition, to prevent thermal degradation and maintain the stability of the nitro group. This careful management of reaction kinetics ensures that the impurity profile remains within tight specifications, which is essential for downstream processing and final drug safety.
Following the acylation, the process incorporates a nucleophilic substitution where the chloro-propoxy group is displaced by dibutylamine, forming the tertiary amine side chain essential for the drug's pharmacological activity. This step is performed in a non-protonic polar solvent such as acetonitrile or butanone, using an acid binding agent like potassium carbonate to neutralize the generated hydrochloric acid. The subsequent catalytic hydrogenation using palladium carbon at room temperature and normal pressure reduces the nitro group to an amine without affecting other sensitive functional groups, demonstrating excellent chemoselectivity. Finally, mesylation and salt formation with hydrogen chloride gas yield the target Dronedarone Hydrochloride with exceptional purity, often exceeding 99% HPLC area normalization after recrystallization. This sequence exemplifies a high-purity OLED material grade of chemical control adapted for pharmaceutical applications.
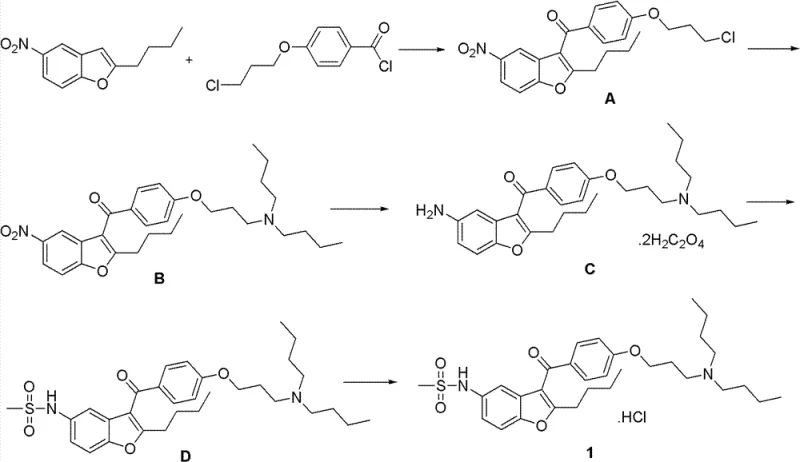
How to Synthesize Dronedarone Hydrochloride Efficiently
The synthesis of this critical cardiovascular API requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and quality consistency across batches. The process begins with the preparation of the acylated intermediate, followed by amination, reduction, and final functionalization, each step building upon the purity of the previous one. Detailed operational guidelines regarding temperature control, reagent addition rates, and workup procedures are essential for maintaining the integrity of the molecular structure throughout the transformation. For technical teams looking to implement this route, understanding the nuances of solvent selection and catalyst loading is paramount to achieving the reported yields and purity levels. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational protocol.
- Perform Friedel-Crafts acylation on 2-butyl-5-nitrobenzofuran with p-chloropropoxybenzoyl chloride using a Lewis acid catalyst.
- React the resulting intermediate with dibutylamine in a non-protonic polar solvent to form the amino-propoxy derivative.
- Execute palladium carbon catalytic hydrogenation at room temperature followed by mesylation and hydrochloride formation to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this novel synthetic route offers substantial cost savings and supply chain resilience compared to legacy manufacturing methods. By eliminating the need for expensive and hazardous demethylation reagents, the overall material cost is significantly reduced, while the simplified purification process lowers energy consumption and waste disposal fees. This efficiency translates directly into a more competitive pricing structure for the final API, allowing pharmaceutical companies to manage their budget allocations more effectively without sacrificing quality standards. Moreover, the use of common, readily available starting materials reduces the risk of raw material shortages, ensuring a stable and continuous supply flow for long-term production planning. These factors combined make this method highly attractive for reducing lead time for high-purity pharmaceutical intermediates in a volatile global market.
- Cost Reduction in Manufacturing: The elimination of harsh demethylation steps removes the requirement for large quantities of anhydrous aluminum trichloride and high-temperature processing equipment, leading to drastic simplification of the production line. This reduction in reagent complexity directly correlates with lower operational expenditures, as there is less need for specialized corrosion-resistant reactors and extensive waste neutralization systems. Additionally, the higher yields observed in the hydrogenation and mesylation steps mean that less starting material is wasted, further optimizing the cost per kilogram of the final active ingredient. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available reagents such as p-chloropropoxybenzoyl chloride and dibutylamine ensures that production is not held hostage by the scarcity of exotic chemicals. The mild reaction conditions, particularly the room temperature hydrogenation, reduce the dependency on specialized high-pressure infrastructure, allowing for more flexible manufacturing scheduling and faster turnaround times. This operational flexibility enhances the reliability of the supply chain, enabling manufacturers to respond more agilely to fluctuations in market demand for antiarrhythmic medications. Consequently, partners can expect consistent delivery schedules and reduced risk of production delays caused by equipment maintenance or safety incidents.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with each step demonstrating robustness when transitioning from laboratory bench scale to multi-ton commercial production. The avoidance of toxic byproducts and the use of recyclable solvents align with increasingly stringent environmental regulations, minimizing the regulatory burden on manufacturing facilities. This compliance advantage reduces the risk of shutdowns due to environmental violations and supports corporate sustainability goals, which are becoming critical criteria for vendor selection in the pharmaceutical industry. The ability to scale this green chemistry approach ensures long-term viability and social responsibility in the production of life-saving medicines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent's background and beneficial effects sections. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios. The answers provided reflect the specific advantages of avoiding harsh conditions and achieving high purity, which are central to the value proposition of this patent. Stakeholders are encouraged to review these points to gain a clearer picture of the operational benefits.
Q: How does this synthesis method improve upon conventional Dronedarone production routes?
A: This method avoids the harsh demethylation conditions requiring excessive aluminum trichloride or high-temperature pyridine hydrochloride found in prior art, significantly reducing environmental pollution and simplifying purification.
Q: What is the expected purity level achievable with this novel process?
A: The process is designed to achieve high purity specifications, with experimental data indicating HPLC purity levels reaching 99.4% after recrystallization, meeting stringent pharmaceutical standards.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the use of mild reaction conditions, room temperature hydrogenation, and easily removable byproducts makes this route highly scalable and safe for industrial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dronedarone Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing. Our commitment to stringent purity specifications and rigorous QC labs guarantees that every batch of Dronedarone Hydrochloride meets the highest international standards for safety and efficacy. We understand the critical nature of cardiovascular APIs and have built our infrastructure to support the complex requirements of modern drug synthesis with unwavering reliability. Partnering with us means gaining access to a team of experts dedicated to optimizing your supply chain and delivering consistent quality.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By engaging with us, you can obtain specific COA data and route feasibility assessments that will empower your decision-making process and secure your supply of this vital medication. Let us collaborate to bring this advanced synthesis technology to your production line, ensuring a stable and cost-effective source of high-quality Dronedarone Hydrochloride for the global market.
