Optimizing Pranlukast Intermediate Production via Advanced Fries Rearrangement and Hydrogenation
Optimizing Pranlukast Intermediate Production via Advanced Fries Rearrangement and Hydrogenation
The pharmaceutical industry continuously seeks robust synthetic pathways for critical asthma therapeutics, and the production of 2-acetamido-6-acetylphenol stands as a pivotal challenge in the supply chain for Pranlukast. Patent CN102060731B, filed in 2013, introduces a transformative methodology that redefines the efficiency of producing this essential pharmaceutical intermediate. By leveraging a streamlined three-step sequence starting from readily available 2-amino-4-chlorophenol, this technology bypasses the cumbersome and environmentally taxing procedures associated with legacy synthesis routes. The innovation lies not merely in the chemical transformations but in the holistic optimization of reaction conditions, solvent systems, and purification protocols that collectively enhance yield and purity profiles. For R&D directors and procurement strategists, understanding the nuances of this patent is crucial for securing a competitive edge in the manufacturing of leukotriene receptor antagonists. The following analysis dissects the technical merits and commercial viability of this approach, highlighting its potential to serve as a benchmark for modern intermediate production.
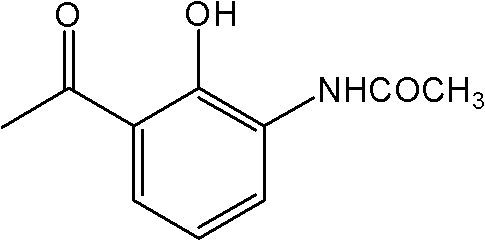
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-acetamido-6-acetylphenol relied on convoluted pathways originating from p-hydroxyphenol, which necessitated a series of chemically aggressive and operationally complex steps. Traditional routes typically involved initial acetylation followed by a Fries rearrangement, but critically, they required subsequent nitration and debromination reactions to install the necessary functional groups at the correct positions. These nitration steps are notorious for generating substantial quantities of hazardous acidic waste and pose significant safety risks due to the exothermic nature of the reactions. Furthermore, the debromination processes often require stoichiometric amounts of reducing agents or expensive catalysts, driving up the cost of goods sold (COGS) and complicating waste treatment protocols. The cumulative effect of these additional steps is a prolonged production cycle, increased exposure to potential impurities, and a larger environmental footprint, making such methods increasingly untenable in the face of modern green chemistry regulations and cost pressures.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN102060731B utilizes 2-amino-4-chlorophenol as a strategic starting material, effectively pre-installing the nitrogen functionality and simplifying the carbon skeleton construction. This novel approach condenses the synthesis into three distinct, high-yielding operations: acylation, rearrangement, and hydrogenation. By selecting a starting material that already possesses the amino group, the need for nitration is entirely eliminated, thereby removing a major source of process hazard and waste. The subsequent rearrangement step is optimized using specific fluxing agents to ensure homogeneous reaction conditions, while the final hydrogenation step employs standard palladium catalysis to cleanly remove the chloro substituent. This strategic redesign of the synthetic tree not only shortens the overall timeline but also aligns perfectly with the principles of atom economy and process intensification, offering a clear pathway to cost reduction in pharmaceutical intermediate manufacturing without compromising on quality or safety standards.
Mechanistic Insights into Fries Rearrangement and Catalytic Dechlorination
The core of this synthetic strategy relies on a meticulously controlled Fries rearrangement, where the phenolic ester intermediate undergoes migration of the acyl group to the ortho-position under Lewis acid catalysis. In this specific embodiment, aluminum chloride (AlCl3) serves as the primary Lewis acid, coordinating with the carbonyl oxygen to facilitate the cleavage of the ester bond and the subsequent electrophilic aromatic substitution. A critical innovation in this patent is the inclusion of fluxing agents such as ammonium chloride or sodium chloride, which act to lower the melting point of the reaction mixture and improve mass transfer at the elevated temperatures required (110-140°C). This ensures that the reaction proceeds homogeneously, minimizing the formation of poly-acylated byproducts and ensuring high regioselectivity for the desired 6-acetyl isomer. The careful management of thermal energy and stoichiometry during this step is paramount, as it dictates the purity profile of the intermediate entering the final stage, directly impacting the ease of downstream purification.
Following the rearrangement, the process employs a catalytic hydrogenation step to achieve the final dechlorination, transforming the chloro-substituted intermediate into the target 2-acetamido-6-acetylphenol. This transformation utilizes palladium on carbon (Pd/C) as a heterogeneous catalyst in the presence of hydrogen gas and an acid-binding agent. The mechanism involves the oxidative addition of the aryl-chloride bond to the palladium surface, followed by reductive elimination with hydrogen to release the dechlorinated product and hydrochloric acid, which is immediately neutralized by the added base. This step is particularly elegant because it avoids the use of harsh chemical reducing agents, relying instead on clean hydrogen gas which produces minimal waste. The choice of solvent, typically ethanol or methanol, further enhances the solubility of the intermediate while maintaining catalyst stability, ensuring that the reaction reaches completion with high conversion rates and minimal formation of over-reduced side products.
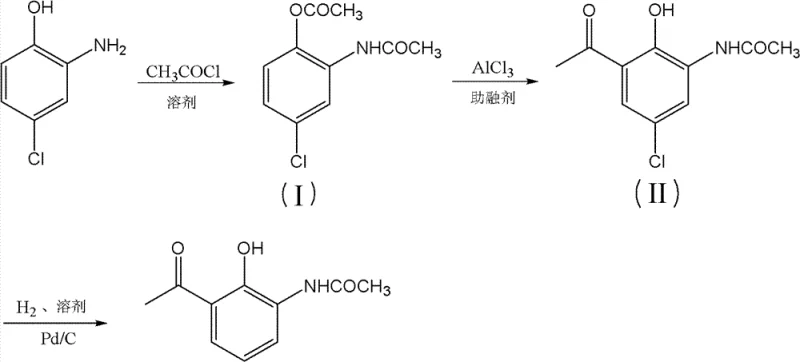
How to Synthesize 2-Acetamido-6-acetylphenol Efficiently
The practical execution of this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and high purity. The process begins with the acylation of 2-amino-4-chlorophenol using acetyl chloride in a suitable organic solvent, where temperature control is vital to prevent di-acylation. Following isolation of the first intermediate, the Fries rearrangement is conducted under anhydrous conditions with precise molar ratios of aluminum chloride and fluxing agents to drive the migration to completion. Finally, the hydrogenation step demands careful monitoring of hydrogen uptake and catalyst loading to ensure complete dechlorination without affecting the sensitive acetyl or amide functionalities. Detailed standardized operating procedures for each of these critical stages are outlined below to guide process engineers in implementing this technology.
- Perform acylation of 2-amino-4-chlorophenol with acetyl chloride in solvent with acid-binding agent to form Intermediate I.
- Execute Fries rearrangement of Intermediate I using AlCl3 and fluxing agent at elevated temperatures to generate Intermediate II.
- Conduct catalytic hydrogenation of Intermediate II using Pd/C to remove the chloro group and yield the final target product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthesis route offers profound strategic benefits that extend beyond simple chemical efficiency. The reliance on 2-amino-4-chlorophenol as a starting material leverages a commodity chemical with a stable and widespread global supply base, mitigating the risks associated with sourcing exotic or custom-synthesized precursors. This stability in raw material availability translates directly into enhanced supply chain reliability, ensuring that production schedules can be maintained without interruption due to upstream shortages. Furthermore, the elimination of nitration and debromination steps drastically simplifies the waste management infrastructure required, as the process generates significantly less hazardous effluent. This reduction in environmental compliance burden allows for faster regulatory approvals and lower operational overheads related to waste disposal, contributing to a more sustainable and cost-effective manufacturing model.
- Cost Reduction in Manufacturing: The streamlined three-step sequence inherently reduces operational costs by minimizing the number of unit operations, solvent exchanges, and isolation steps required to reach the final product. By avoiding expensive reagents associated with nitration and debromination, and by utilizing cost-effective catalysts like aluminum chloride and recoverable palladium on carbon, the overall variable cost of production is substantially lowered. Additionally, the high yields reported in the patent examples indicate efficient material utilization, which further drives down the cost per kilogram of the active intermediate, providing significant margin improvement for downstream API manufacturers.
- Enhanced Supply Chain Reliability: The use of common industrial solvents such as ethyl acetate, toluene, and ethanol ensures that the process is not dependent on specialized or restricted chemical supplies that could face logistical bottlenecks. The robustness of the reaction conditions, particularly the tolerance demonstrated in the pilot-scale examples, suggests that the process can be reliably transferred between different manufacturing sites without loss of performance. This flexibility empowers supply chain managers to diversify their manufacturing network, reducing dependency on single-source suppliers and enhancing the overall resilience of the pharmaceutical supply chain against global disruptions.
- Scalability and Environmental Compliance: The patent explicitly demonstrates successful scale-up from laboratory to pilot plant scales, confirming that the thermal and mixing requirements of the Fries rearrangement can be safely managed in larger reactors. The absence of highly toxic reagents and the generation of manageable waste streams align with increasingly stringent environmental regulations, facilitating smoother permitting processes for new production facilities. This scalability ensures that the technology can meet growing market demand for Pranlukast without requiring disproportionate increases in capital expenditure or environmental remediation costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and process data. These insights are intended to clarify the operational feasibility and quality implications for stakeholders evaluating this route for potential adoption or outsourcing. Understanding these details is essential for making informed decisions about process integration and supplier qualification.
Q: What are the key advantages of this synthesis route over traditional nitration methods?
A: This route eliminates hazardous nitration and debromination steps, significantly reducing waste generation and improving overall process safety while shortening the synthetic timeline.
Q: How is the purity of the final intermediate controlled during the rearrangement step?
A: Purity is managed through precise temperature control during the Fries rearrangement and optimized recrystallization using specific solvent mixtures like ethyl acetate and toluene.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly details pilot-scale examples demonstrating robust scalability, utilizing common industrial solvents and standard catalytic hydrogenation equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Acetamido-6-acetylphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of respiratory therapeutics like Pranlukast. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN102060731B can be executed with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-acetamido-6-acetylphenol meets the exacting standards required for GMP API synthesis. Our commitment to technical excellence ensures that our partners receive a product that facilitates smooth downstream processing and final drug approval.
We invite pharmaceutical companies and contract manufacturers to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make data-driven decisions that enhance your competitive position in the global asthma medication market.
