Advanced Synthesis of Chiral Ferrocene Amines for Industrial Ligand Production
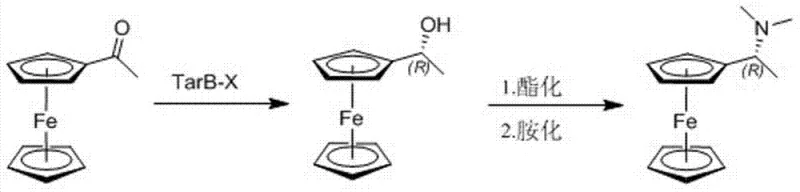
The landscape of asymmetric catalysis relies heavily on the availability of high-purity chiral ligands, with ferrocene-based diphosphine ligands standing out as critical components for industrial hydrogenation reactions. Patent CN103755748A introduces a groundbreaking preparation process for chiral (R)-1-ferrocenyl ethyl dimethylamine, a pivotal intermediate in the synthesis of these valuable ligands. Unlike traditional methods that struggle with low yields and complex purification, this invention utilizes acetylferrocene as a starting material, subjecting it to asymmetric reduction to obtain R-configured ferrocenyl ethanol. This is followed by a highly efficient one-pot esterification and amination sequence to yield the optically pure target amine. The significance of this technical disclosure lies in its ability to bypass resolution steps entirely, thereby maximizing atomic economy and ensuring consistent optical purity suitable for demanding pharmaceutical applications. For R&D directors and procurement specialists, this represents a shift towards more sustainable and economically viable manufacturing protocols for complex organometallic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral ferrocenyl ethyl dimethylamine has been plagued by inherent inefficiencies associated with chiral resolution techniques. Conventional approaches typically involve synthesizing the racemic mixture first and then attempting to separate the desired enantiomer using chiral resolving agents. This methodology is fundamentally flawed from a production standpoint because it inherently caps the maximum theoretical yield at 50%, meaning half of the produced material is often discarded or requires costly recycling processes. Furthermore, the resolution process itself adds multiple unit operations, including crystallization, filtration, and regeneration of the resolving agent, which significantly increases solvent consumption and energy requirements. The operational complexity is further compounded by the need for precise control over crystallization conditions to ensure high optical purity, often resulting in batch-to-batch variability. For supply chain managers, these factors translate into unpredictable lead times and inflated raw material costs, making the conventional route less attractive for large-scale commercial adoption in the competitive fine chemical market.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this synthesis by employing a direct asymmetric reduction strategy followed by a telescoped one-pot transformation. By utilizing a specific chiral ligand, such as TarB-X, in conjunction with a borohydride reductant, the process achieves high stereoselectivity directly from the prochiral ketone, acetylferrocene. This eliminates the need for resolution, effectively doubling the potential yield compared to racemic methods. The subsequent conversion of the chiral alcohol to the amine is achieved through a seamless one-pot procedure where esterification and amination occur sequentially without isolating the intermediate ester. This telescoping of reactions not only simplifies the workflow but also minimizes the exposure of sensitive intermediates to potential degradation. The result is a robust, high-yielding process that operates under mild conditions, offering a compelling alternative for manufacturers seeking to optimize their production of high-purity pharmaceutical intermediates while reducing overall process mass intensity.
Mechanistic Insights into TarB-X Catalyzed Asymmetric Reduction
The core of this technological advancement lies in the stereoselective reduction of the ketone group in acetylferrocene, mediated by the chiral environment provided by the TarB-X ligand. The mechanism involves the formation of a chiral borate complex where the TarB-X ligand coordinates with the borohydride species, creating a sterically defined pocket for hydride delivery. This chiral pocket discriminates between the two faces of the planar carbonyl group, preferentially delivering the hydride ion to one face to generate the (R)-configured alcohol with high enantiomeric excess. The choice of substituents on the TarB-X ligand, such as hydrogen, nitro, chloro, or trifluoromethyl groups, allows for fine-tuning of the electronic and steric properties, further enhancing the selectivity and reaction rate. Understanding this mechanistic nuance is crucial for R&D teams aiming to replicate or scale this chemistry, as it highlights the importance of ligand integrity and reaction temperature control in maintaining high optical purity throughout the synthesis.
Following the asymmetric reduction, the control of impurities becomes paramount, particularly in the one-pot esterification and amination steps. The process utilizes acetic anhydride for esterification in the presence of a base and a catalyst like DMAP or tetrabutylammonium salts, which activates the alcohol for nucleophilic attack. The subsequent addition of dimethylamine solution facilitates the nucleophilic substitution of the acetate group. A key advantage of this mechanism is that the reaction conditions are mild enough to prevent racemization of the chiral center, which is a common risk in amine synthesis involving alpha-chiral carbons. The absence of intermediate isolation means that potential impurities from the reduction step are carried forward but are effectively managed or removed during the final workup, which typically involves extraction and washing. This integrated approach ensures that the final product meets stringent purity specifications required for use in catalytic applications, minimizing the risk of catalyst poisoning in downstream hydrogenation reactions.
How to Synthesize (R)-1-ferrocenyl ethyl dimethylamine Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and temperature management to ensure optimal yield and stereochemistry. The process begins with the preparation of the chiral reducing agent in situ, followed by the controlled addition of the acetylferrocene substrate. Once the reduction is complete, the reaction mixture is directly treated with acylating agents and amines without intermediate purification, leveraging the compatibility of the reagents to drive the transformation to completion. Detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are essential for reproducibility and are outlined in the technical guide below for process engineers.
- Asymmetric reduction of acetylferrocene using TarB-X chiral ligand and borohydride reductant at controlled temperatures.
- Direct one-pot esterification of the resulting alcohol using acetic anhydride and base without isolation.
- In-situ amination using dimethylamine solution to yield the final chiral amine product with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers significant strategic advantages for procurement managers and supply chain heads looking to optimize their sourcing of chiral intermediates. The elimination of the resolution step fundamentally alters the cost structure of the manufacturing process by removing the need for expensive chiral resolving agents and the associated loss of material. This efficiency gain translates directly into a more competitive pricing model for the final product, allowing buyers to secure high-quality materials without the premium typically associated with resolved chiral compounds. Furthermore, the use of readily available and inexpensive raw materials like acetylferrocene and sodium borohydride ensures that the supply chain is not vulnerable to the volatility of exotic reagent markets. This stability is crucial for long-term production planning and risk management in the pharmaceutical and fine chemical sectors.
- Cost Reduction in Manufacturing: The streamlined one-pot methodology significantly reduces operational expenditures by minimizing the number of unit operations required. By combining esterification and amination into a single vessel, manufacturers save substantially on solvent usage, energy consumption for heating and cooling cycles, and labor costs associated with multiple isolation steps. The removal of the resolution process also means that the effective yield per batch is drastically increased, lowering the cost per kilogram of the active intermediate. These cumulative savings allow for a more aggressive pricing strategy while maintaining healthy margins, making the final ligand products more accessible for downstream applications in drug manufacturing.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetic anhydride, dimethylamine, and common borohydrides ensures a robust and resilient supply chain. Unlike processes that depend on specialized, single-source catalysts or complex chiral auxiliaries, this method utilizes reagents that are widely produced and stocked by multiple global suppliers. This diversification of the raw material base reduces the risk of supply disruptions and allows for greater flexibility in procurement negotiations. Additionally, the mild reaction conditions reduce the wear and tear on production equipment, leading to higher asset availability and more consistent delivery schedules for customers relying on just-in-time inventory models.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily manageable in large-scale reactors without requiring extreme pressures or temperatures. The reduction in solvent usage and the elimination of waste streams associated with resolution by-products contribute to a lower environmental footprint, aligning with increasingly strict global regulations on chemical manufacturing. This green chemistry profile not only simplifies regulatory compliance but also enhances the corporate sustainability credentials of the supply chain. For supply chain heads, this means a future-proof sourcing strategy that mitigates the risk of regulatory shutdowns and aligns with the environmental goals of major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: What is the primary advantage of this synthesis route over traditional resolution methods?
A: The primary advantage is the elimination of the resolution step, which traditionally results in a maximum theoretical yield of 50%. This asymmetric synthesis route achieves high yields and high enantiomeric excess (ee value) directly, significantly improving material efficiency and reducing waste generation associated with discarding the unwanted enantiomer.
Q: How does the one-pot esterification and amination process impact production costs?
A: By combining esterification and amination into a single pot without intermediate isolation or purification, the process drastically reduces solvent consumption, energy usage for drying and distillation, and labor hours. This streamlining of operations leads to substantial operational cost savings and a shorter overall production cycle time.
Q: Are the reagents used in this process suitable for large-scale industrial application?
A: Yes, the process utilizes readily available and inexpensive reagents such as acetylferrocene, sodium borohydride, and acetic anhydride. The reaction conditions are mild (0°C to 30°C), avoiding the need for extreme high-pressure or high-temperature equipment, which makes the technology highly scalable and safe for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-1-ferrocenyl ethyl dimethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation pharmaceuticals and advanced materials. Our team of expert chemists has extensively evaluated the synthesis route described in patent CN103755748A and possesses the technical capability to scale this diverse pathway from 100 kgs to 100 MT/annual commercial production. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, ensuring that every batch of (R)-1-ferrocenyl ethyl dimethylamine performs consistently in your catalytic applications. Our infrastructure is designed to handle complex organometallic chemistry safely and efficiently, providing you with a secure and reliable source for your key building blocks.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this advanced synthesis technology into your production portfolio. Let us be your partner in driving innovation and efficiency in your chemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
